Abstract
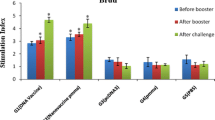
In the present study, we evaluated induced immune responses following DNA vaccine containing cocktail or fusion of LeIF, LACK and TSA genes or each gene alone. Mice were injected with 100 µg of each plasmid containing the gene of insert, plasmid DNA alone as the first control group or phosphate buffer saline as the second control group. Then, cellular and humoral responses, lesion size were measured for all groups. All vaccinated mice induced Th1 immune responses against Leishmania characterized by higher IFN-γ and IgG2a levels compared with control groups (p < 0.05). In addition, IFN-γ levels increased in groups immunized with fusion and cocktail vaccines in comparison with LACK (p < 0.001) and LeIF (p < 0.01) groups after challenge. In addition, fusion and cocktail groups produced higher IgG2a values than groups vaccinated with a gene alone (p < 0.05). Lesion progression delayed for all immunized groups compared with control groups from 5th week post-infection (p < 0.05). Mean lesion size decreased in immunized mice with fusion DNA than three groups vaccinated with one gene alone (p < 0.05). While, lesion size decreased significantly in cocktail recipient group than LeIF recipient group (p < 0.05). There was no difference in lesion size between fusion and cocktail groups. Overall, immunized mice with cocktail and fusion vaccines showed stronger Th1 response by production of higher IFN-γ and IgG2a and showed smaller mean lesion size. Therefore, use of multiple antigens can improve induced immune responses by DNA vaccination.





Similar content being viewed by others
References
Abdoli A, Maspi N, Ghaffarifar F (2017) Wound healing in cutaneous leishmaniasis: a double edged sword of IL-10 and TGF-β. Comp Immunol Microbiol Infect Dis 51:15–26
Ahmed SB, Touihri L, Chtourou Y et al (2009) DNA based vaccination with a cocktail of plasmids encoding immunodominant Leishmania (Leishmania) major antigens confers full protection in BALB/c mice. Vaccine 27:99–106
Alvar J, Canavate C, Gutierrez-Solar B et al (1997) Leishmania and human immunodeficiency virus coinfection: the first 10 years. Clin Microbiol Rev 10:298–319
Alvar J, Vélez ID, Bern C et al (2012) Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 7:e35671
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Campos-Neto A, Webb JR, Greeson K et al (2002) Vaccination with plasmid DNA encoding TSA/LmSTI1 leishmanial fusion proteins confers protection against Leishmania major infection in susceptible BALB/c mice. Infect Immun 70:2828–2836
Carneiro FP, De Magalhaes AV, De Jesus Abreu Almeida Couto M et al (2009) Foxp3 expression in lesions of the different clinical forms of American tegumentary leishmaniasis. Parasite Immunol 31:646–651
Coelho EA, Tavares CA, Carvalho FA et al (2003) Immune responses induced by the Leishmania (Leishmania) donovani A2 antigen, but not by the LACK antigen, are protective against experimental Leishmania (Leishmania) amazonensis infection. Infect Immun 71:3988–3994
Coler RN, Skeiky YAW, Bernards K et al (2002) Immunization with a polyprotein vaccine consisting of the T-cell antigens thiol-specific antioxidant, Leishmania major stress-inducible protein 1, and Leishmania elongation initiation factor protects against leishmaniasis. Infect Immun 70:4215–4225
Coler RN, Goto Y, Bogatzki L et al (2007) Leish-111f, a recombinant polyprotein vaccine that protects against visceral Leishmaniasis by elicitation of CD4+ T cells. Infect Immun 75:4648–4654
Das S, Freier A, Boussoffara T et al (2014) Modular multiantigen T cell epitope-enriched DNA vaccine against human leishmaniasis. Sci Transl Med 6:234ra56
Dunning N (2009) Leishmania vaccines: from leishmanization to the era of DNA technology. Biosci Horizons 2:73–82
Ghaffarifar F, Jorjani O, Sharifi Z et al (2013) Enhancement of immune response induced by DNA vaccine cocktail expressing complete LACK and TSA genes against Leishmania major. APMIS 121:290–298
Gonzalez U, Pinart M, Rengifo-Pardo M et al (2009) Interventions for American cutaneous and mucocutaneous leishmaniasis. Cochrane Database Syst Rev 2:CD004834
Goto Y, Bhatia A, Raman VS et al (2011) KSAC, the first defined polyprotein vaccine candidate for visceral leishmaniasis. Clin Vaccine Immunol 18:1118–1124
Gurunathan S, Sacks DL, Brown DR et al (1997) Vaccination with DNA encoding the immunodominant LACK parasite antigen confers protective immunity to mice infected with Leishmania major. J Exp Med 186:1137–1147
Gurunathan S, Klinman DM, Seder RA (2000) DNA vaccines: immunology, application, and optimization. Annu Rev Immunol 18:927–974
Heinzel FP, Sadick MD, Holaday BJ et al (1989) Reciprocal expression of interferon gamma or interleukin 4 during the resolution or progression of murine leishmaniasis. Evidence for expansion of distinct helper T cell subsets. J Exp Med 169:59–72
Hepburn NC (2000) Cutaneous leishmaniasis. Clin Exp Dermatol 25:363–370
Hezarjaribi HZ, Ghaffarifar F, Dalimi A et al (2014) Evaluation of protective effect of IL-22 and IL-12 on cutaneous leishmaniasis in BALB/c mice. Asian Pac J Trop Med 7:940–945
Ivory C, Chadee K (2004) DNA vaccines: designing strategies against parasitic infections. Genet Vaccines Ther 2:17
Jorjani O, Ghaffarifar F, Sharifi Z et al (2012) Cloning and expression of recombinant plasmid containing LACK gene of Leishmania major (MHRO/IR/75/ER) in CHO cells. ISESCO J Sci Technol 8:37–43
Katara GK, Ansari NA, Verma S et al (2011) Foxp3 and IL-10 expression correlates with parasite burden in lesional tissues of post kala azar dermal leishmaniasis (PKDL) patients. PLoS Negl Trop Dis 5:e1171
Kedzierski L, Zhu Y, Handman E (2006) Leishmania vaccines: progress and problems. Parasitology 133(Suppl):S87–112
Kedzierski L, Sakthianandeswaren A, Curtis JM et al (2009) Leishmaniasis: current treatment and prospects for new drugs and vaccines. Curr Med Chem 16:599–614
Kemp K (2000) Cytokine-producing T cell subsets in human leishmaniasis. Arch Immunol Ther Exp 48:173–176
Khamesipour A, Dowlati Y, Asilian A et al (2005) Leishmanization: use of an old method for evaluation of candidate vaccines against leishmaniasis. Vaccine 23:3642–3648
Kutzler MA, Weiner DB (2008) DNA vaccines: ready for prime time? Nat Rev Genet 9:776–788
Laguna F (2003) Treatment of leishmaniasis in HIV-positive patients. Ann Trop Med Parasitol 97(Suppl 1):135–142
Liu MA (2011) DNA vaccines: an historical perspective and view to the future. Immunol Rev 239:62–84
Locksley RM, Scott P (1991) Helper T-cell subsets in mouse leishmaniasis: induction, expansion and effector function. Immunol Today 12:A58–A61
Lu S, Wang S, Grimes-Serrano JM (2008) Current progress of DNA vaccine studies in humans. Expert Rev Vaccines 7:175–191
Maspi N, Ghaffarifar F, Sharifi Z et al (2015) Cloning and constructing a plasmid encoding leishmania eukaryotic initiation factor gene of Leishmania major fused with green fluorescent protein gene as a vaccine candidate. West Indian Med J 65:256–259
Maspi N, Abdoli A, Ghaffarifar F (2016a) Pro- and anti-inflammatory cytokines in cutaneous leishmaniasis: a review. Pathog Glob Health 110:247–260
Maspi N, Ghaffarifar F, Sharifi Z et al (2016b) Codelivery of DNA vaccination encoding LeIF gene and IL-12 increases protection against Leishmania major infection in BALB/c mice. Parasite Immunol 38:228–235
Maspi N, Ghaffarifar F, Sharifi Z et al (2017) Immunogenicity and efficacy of a bivalent DNA vaccine containing LeIF and TSA genes against murine cutaneous leishmaniasis. APMIS 125:249–258
McLachlan JB, Catron DM, Moon JJ et al (2009) Dendritic cell antigen presentation drives simultaneous cytokine production by effector and regulatory T cells in inflamed skin. Immunity 30:277–288
Miura R, Kooriyama T, Yoneda M et al (2015) Efficacy of recombinant canine distemper virus expressing Leishmania antigen against Leishmania challenge in dogs. PLoS Negl Trop Dis 9:e0003914
Nadim A, Javadian E, Tahvildar-Bidruni G et al (1983) Effectiveness of leishmanization in the control of cutaneous leishmaniasis. Bull Soc Pathol Exot Filiales 76:377–383
Okwor I, Uzonna J (2009) Vaccines and vaccination strategies against human cutaneous leishmaniasis. Hum Vaccines 5:291–301
Premenko-Lanier M, Rota PA, Rhodes GH et al (2004) Protection against challenge with measles virus (MV) in infant macaques by an MV DNA vaccine administered in the presence of neutralizing antibody. J Infect Dis 189:2064–2071
Rabello A, Orsini M, Disch J (2003) Leishmania/HIV co-infection in Brazil: an appraisal. Ann Trop Med Parasitol 97(Suppl 1):17–28
Reithinger R, Dujardin JC, Louzir H et al (2007) Cutaneous leishmaniasis. Lancet Infect Dis 7:581–596
Sacks D, Noben-Trauth N (2002) The immunology of susceptibility and resistance to Leishmania major in mice. Nat Rev Immunol 2:845–858
Salman SM, Rubeiz NG, Kibbi AG (1999) Cutaneous leishmaniasis: clinical features and diagnosis. Clin Dermatol 17:291–296
Scott P, Novais FO (2016) Cutaneous leishmaniasis: immune responses in protection and pathogenesis. Nat Rev Immunol 16:581–592
Tabatabaie F, Ghaffarifar F, Dalimi A et al (2007) Cloning and sequencing of Leishmania major thiol-specific-antioxidant antigen (TSA) gene. Iran J Parasitol 2:30–41
Tabatabaie F, Mahdavi M, Faezi S et al (2014) Th1 platform immune responses against Leishmania major induced by thiol-specific antioxidant-based DNA vaccines. Jundishapur J Microbiol 7:e8974
Talat H, Attarwala S, Saleem M (2014) Cutaneous Leishmaniasis with HIV. J Coll Phys Surg Pak 24(Suppl 2):S93–S95
WHO (2010) New technical report series on the control of leishmaniasis. WHO, Geneva, pp 49–64
Williams JA (2013) Vector design for improved DNA vaccine efficacy, safety and production. Vaccines 1:225–249
Acknowledgements
This research was financially supported by Faculty of Medical Sciences, Tarbiat Modares University, and Tehran, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical consideration
All procedures including maintenance, animal handling and blood sample collection were approved by Medical Ethics Committee of Tarbiat Modares University of Iran (ID: 52/5101 dated March 16, 2013).
About this article
Cite this article
Maspi, N., Ghaffarifar, F., Sharifi, Z. et al. Comparative Assessment of Induced Immune Responses Following Intramuscular Immunization with Fusion and Cocktail of LeIF, LACK and TSA Genes Against Cutaneous Leishmaniasis in BALB/c Mice. Arch. Immunol. Ther. Exp. 66, 55–64 (2018). https://doi.org/10.1007/s00005-017-0484-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00005-017-0484-4