Abstract
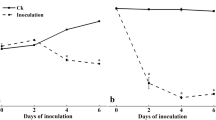
The amounts of oxalic acid and polygalacturonase (PG) produced in the tissues of cowpea (Vigna unguiculata Walp.) plants infected byPythium aphanidermatum were higher in vars. IT81D-1020 and VITA 5 than in vars. IT82E-32 and TVX3236. Oxalic acid accumulated early at the infective stage of the disease but its production decreased as the plants became older or as the disease developed. At the peak of oxalic acid and PG production (8–10 days after infection) the pH of the tissue fell from 7.2 to 3.1 and thereafter rose to 4.3 (within 18 days). It seems, therefore, that the combination of oxalic acid and PG and the accompanying reduced pH of infected tissue play an important role in the pathogenesis of susceptible varieties of cowpea byP. aphanidermatum. In the two varieties found to be resistant in this study, oxalic acid and PG production were lower than in the susceptible varieties and the pH did not fall as low. Early accumulation of oxalic acid in cowpea tissue during pathogenesis may be a useful tool for monitoring disease severity, and hence susceptibility or resistance toP. aphanidermatum, when there is a compatible interaction between host and pathogen.
Similar content being viewed by others
References
Andrew, J.C. and Viser, E.T. (1951) The oxalic acid content of some common foods.FdRes. 16:306–312.
Anon. (1980) Pests, Diseases, Resistance and Protection in Cowpeas. UTA, Ibadan, Nigeria.
Bateman, D.F. (1963) The “macerating enzyme” ofRhizoctonia solarii.Phytopathology53:1178–1186.
Bateman, D.F. (1964) An induced mechanism of tissue resistance to polygalacturonase inRhizoctonia-infected hypocotyls of bean.Phytopathology 54:438–445.
Bateman, D.F. and Beer, S.V. (1965) Simultaneous production and synergistic action ofoxalic acid and polygalacturonase during pathogenesis bySclerotium rolfsii.Phytopathology 55:204–211.
Domsh, K.H. and Anderson, T. (1980) Compendium of Soil Fungi. Vol. 1, pp. 678–682.
Echandi, E. and Walker, J.C. (1957) Pectolytic enzymes produced bySclerotiniasclerotiorum.Phytopathology 47:303–306.
Faboya, O., Ikotun, T. and Fatoki, O.S. (1983) Production of oxalic acid by some fungi ininfected tubers.Z. allg. Microbiol. 24:621–624.
Hancock, J.G. (1967) Hemicellulose degradation in sunflower hypocotyls infected withSclerotinia sclerotiorum.Phytopathology 57:203–206.
Ikotun, T. (1984) Cell wall degrading enzymes produced byPenicillium oxalicum Curie andThorn.Mycopath. Mycol. appi. 88:15–21.
Lumsden, R.D. (1969)Sclerotinia sclerotorium infection of bean and the production ofcellulase.Phytopathology 59:653–657.
Ogundana, S.K. (1971) Studies on pythium wet rot of cowpea in Nigeria.Afr. J. PL Prot.2:71–74.
Onuorah, P.E. (1973) Pythium seed decay and stem rot of cowpea(Vigna sinensis [Linn.]Savi) in southern Nigeria.Pl. Soil 39:187–191.
Pierce, W.C. and Haenisch, E.L. (1954) Quantitative Analysis. John Wiley, New York, NY.p. 520.
Singh, S.R. and Allen, D.T. (1979) Cowpea Pests and Diseases. Manual series. UTA, Ibadan,Nigeria.
Williams, R.J. and Ayanaba, A. (1975) Increased incidence ofPythium stem rot in cowpeastreated with benomyl and related fungicides.Phytopathology 65:217–218.
Winstead, N.N. and McCombs, C.L. (1961) Pectinolytic and cellulolytic enzyme productionbyPythium aphanidermatum.Phytopathology 51:270–273.
Zaumeyer, W.J. and Thomas, H.R. (1962) Bean Diseases. How to Control Them. HandbkU.S. Dep. Agric. 25.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koleosho, B., Ikotun, T. & Faboya, O. The role of oxalic acid and polygalacturonase in the pathogenicity ofPythium aphanidermatum on different cowpea varieties. Phytoparasitica 15, 317–323 (1987). https://doi.org/10.1007/BF02979547
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02979547