Abstract
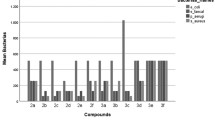
Thein vitro antifungal activity of several N2-phenyl-3(2H)-isothiazolones substituted at C4 of the phenyl moiety with heterocyclic nucleus or groups of different physico-chemical properties against four human pathogenic fungi was determined by broth macrodilution method; results were compared with those obtained with itraconazole and ketoconazole. These isothiazolones showed moderate to high activity against some or all tested strains and in comparison with the reference drugs, 5-chloro-2-(4-nitrophenyl)isothiazol-3-one (1g), 5-chloro-2-phenylisothiazol-3-one (1c), 4-[4-(5-chloro-3-oxo-3H-isothiazol-2-yl)phenyl]-1,4-dihydrotriazol-5-one (1s) and 2-(4-nitrophenyl)isothiazol-3-one (2g) againstAspergillus niger, 5-chloro-2-(4-nitrophenyl)isothiazol-3-one (1g) and 4-[4-(5-chloro-3-oxo-3H-isothiazol-2-yl)phenyl]piperazine-1-carboxamide (1q) againstTrichophyton mentagrophytes had comparable activity, compounds1g and2g showing higher activity againstMicrosporum canis. Antifungal activity was favored by the presence of chlorine at C5 of the isothiazolone and/or the presence of nitro group or heterocyclic nucleus at C4 of the phenyl ring and proper hydrophilicity of the molecule.
Similar content being viewed by others
Abbreviations
- MIC(s):
-
minimal inhibitory concentration(s)
- Mops:
-
3-morpholinopropanesulfonic acid
References
Jantová S., Ovádeková R., Letašiová S., Špirková K., Stankovský Š.: Antimicrobial activity of some substituted triazoloquinazolines.Folia Microbiol. 50, 90–94 (2005).
Ji H., Zhang W., Zhou Y., Zhang M., Zhu J., Song Y., Lü J., Zhu J.: A three-dimensional model of lanosterol 14α-demethylase ofCandida albicans and its interaction with azole antifungals.J.Med.Chem. 43, 2493–2505 (2000).
Khalaj A., Adibpour N., Shahverdi A.R., Daneshtalab M.: Synthesis and antibacterial activity of 2-(4-substituted phenyl)-3(2H)-isothiazolones.Eur.J.Med.Chem. 43, 699–705 (2004).
Lewis S.N., Miller G.A., Law A.B.: Pesticidal substituted 3-isothiazolinones.French Pat. 1 555 416 (1969);Chem.Abstr. 72, 111 459n (1970).
Lewis S.N., Miller G.A.: Biocidal acyl derivatives of oxo- and oxyisothiazoles.US Pat. 3 544 580 (1970);Chem.Abstr. 76, 34 242q (1972).
Lewis S.N., Miller G.A.: 3-Hydroxyisothiazole and 4-isothiazolinone-3-on derivatives.US Pat. 3 706 757 (1972);Chem.Abstr. 78, 84 399hq (1973).
Lewis S.N., Miller G.A., Law A.B.: 3-Isothiazolones.US Pat. 3 761 488 (1973).
Lewis S.N., Miller G.A.: Derivatives of 3-isothiazolones with active halogen compounds.US Pat. 3 835 150 (1974);Chem.Abstr. 81, 152 212f (1974).
Lewis S.N., Miller G.A., Law A.B.: 3-Isothiazolones as biocides.US Pat. 4 105 431 (1978);Chem.Abstr. 90, 54 934j (1979).
Lewis S.N., Miller G.A., Law A.B.: Cosmetic formulation comprising 3-isothiazolones.US Pat. 4 265 899 (1981).
Messick C.R., Pendland S.L., Moshirfar M., Fiscella R.G., Losendahl K.J., Schriever C.A., Schreckenberger P.C.:In-vitro activity of polyhexamethylene biguanide (PHMB) against fungal isolates associated with infective keratitis.J.Antimicrob.Chemother. 44, 297 (1999).
Sádaba B., García-Quetglas E., Azanza J.R.: Relación entre e structura y función en los azoles.Rev.Esp.Quimioterap. 17, 71–78 (2004).
Yoshida Y., Aoyama Y.: Interaction of azole antifungal agents with cytochrome P-45014DM purified fromSaccharomyces cerevisiae microsomes.Biochem.Pharmacol. 36, 229–235 (1987).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adibpour, N., Khalaj, A., Rezaee, S. et al. In Vitro antifungal activity of 2-(4-substituted phenyl)-3(2H)-isothiazolones. Folia Microbiol 52, 573–576 (2007). https://doi.org/10.1007/BF02932185
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02932185