Abstract
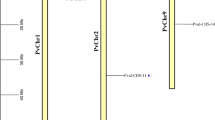
Lophopyrum elongatum is a facultative halophyte related to wheat. Eleven unique clones corresponding to genes showing enhanced mRNA accumulation in the early stages of salt stress were previously isolated from a L. elongatum salt-stressed-root cDNA library. The chromosomal distribution of genes complementary to these clones in several genomes of the tribe Triticeae and their copy number in the L. elongatum and wheat genomes are reported. Genes complementary to clones pESI4, pESI14, pESI15, pESI28, and pESI32 were found in homoeologous group 5, those complementary to pESI18 and pESI35 in homoeologous group 6, and those complementary to pESI47, pESI48, pESI3, and pESI2 in homoeologous groups 1, 3, 4, and 7, respectively. The genes are present in a single copy per genome in L. elongatum with the exception of those complementary to pESI2 and pESI18 which are present in at least two and five copies, respectively. Since similar copy numbers per genome were found in wheat (except for pESI2), the ability of L. elongatum to accumulate higher mRNA levels than wheat in response to salt shock apears to have evolved by changes in the regulation of these genes.
Similar content being viewed by others
References
Anderson JA, Ogihara Y, Sorells ME, Tanksley SD (1992) Development of a chromosomal arm map for wheat based on RFLP markers. Theor Appl Genet 83:1035–1043
Bennett MD (1972) Nuclear DNA content and minimum generation time in herbaceous plants. Proc R Soc Lond Ser B 181:109–135
Chandler PM, Walker-Simmons M, King RW, Crouch M, Close TJ (1988) Expression of ABA-inducible genes in waterstressed cereal seedlings (abstract). J Cell Biochem suppl 12C:143
Close TJ, Chandler PM (1990) Cereal dehydrins: serology, gene mapping and potential functional roles. Aust J Plant Physiol 17:333–344
Dvořák J (1979) Metaphase-I pairing frequencies of individual Argopyron elongatum chromosome arms with Triticum chromosomes. Can J Genet Cytol 21:243–254
Dvořák J (1980) Homoeology between Agropyron elongatum chromosomes and Triticum aestivum chromosomes. Can J Genet Cytol 26:237–259
Dvořák J, Chen KC (1984) Phylogenetic relationships between chromosomes of wheat and chromosome 2E of Elytrigia elongata. Can J Genet Cytol 26:128–132
Dvořák J, Ross K (1986) Expression of tolerance of Na+, K+, Mg2+, Cl-, and SO 42 - ions and sea water in the amphiploid of Triticum aestivum x Elytrigia elongata. Crop Sci 26:658–660
Dvořák J, McGuire PE, Cassidy B (1988) Apparent sources of the A genomes of wheats inferred from polymorphism in abundance and restriction fragment length of repeated nucleotide sequences. Genome 30:680–689
Dvořák J, Resta P, Kota RS (1990) Molecular evidence on the origin of wheat chromosomes 4A and 4B. Genome 33:30–39
Dvořák J, Epstein E, Galvez A, Gulick PJ, Omielan JA (1992) Genetics of the physiological and molecular mechanisms associated with the salt tolerance in wheat and Lophopyrum elongatum. Proc Int Conf on Agricultural Management in Salt-affected Areas, Agadir, Morocco, April 26–May 3, 1991, pp 125–136
Galvez AF, Gulick PJ, Dvořák J (1993) Characterization of the early stages of genetic salt-stress responses in salt-tolerant Lophopyrum elongatum; salt-sensitive wheat and their amphiploid. Plant Phys 103:257–265
Gulick PJ, Dvořák J (1987) Gene induction and repression by salt treatment in roots of the salinity-sensitive Chinese Spring wheat and the salinity-tolerant Chinese Spring x Elytrigia elongata amphiploid. Proc Natl Acad Sci USA 84:99–103
Gulick PJ, Dvořák J (1990) Selective enrichment of cDNAs from salt-stress-induced genes in the wheatgrass, Lophopyrum elongatum, by the formamide-phenol emulsion reassociation technique. Gene 95:173–177
Gulick PJ, Dvořák J (1992) Coordinate gene response to salt stress in Lophopyum elongatum. Plant Physiol 100:1384–1388
Hart GE, Tuleen NA (1983a) Chromosomal locations of eleven Elytrigia elongata (=Agropyron elongatum) isozyme structural genes. Genet Res 41:181–202
Hart GE, Tuleen NA (1983b) Introducing and characterization of alien genetic material. In: Tanksley SD, Orton TJ (ed) Isozymes in plant genetics and breeding. Elsevier Scientific, Amsterdam, pp 35–55
Heun M, Kennedy AE, Anderson JA, Lapitan NLV, Sorrells ME, Tanksley SD (1991) Construction of a restriction fragment length polymorphism map for barley (Hordeum vulgare). Genome 34:437–447
Islam AKMR, Shepherd KW, Sparrow DHB (1981) Isolation and characterization of euplasmic wheat-barley chromosome addition lines. Heredity 46:161–174
McGuire PE, Dvořák J (1981) High salt-tolerance potential in wheatgrasses. Crop Sci 21:702–705
Mundy J, Chua NH (1988) Abscisic acid and water stress induce the expression of a novel rice gene. EMBO J 7:2279–2286
Omielan JA, Epstein E, Dvořák J (1991) Salt tolerance and ionic relations of wheat as affected by individual chromosomes of salt-tolerant Lophopyrum elongatum. Genome 34:961–974
Piatkowski D, Schneider K, Salamini F, Bartels D (1990) Characterization of five abscisic acid-responsive cDNA clones isolated from the desiccation-tolerant plant Craterostigma plantagineum and their relationship to other water-stress genes. Plant Physiol 94:1682–1688
Sears ER, Sears LMS (1979) The telocentric chromosomes of common wheat. Proc 5th Int Wheat Genetic Symp, vol 2, pp 389–407
Tuleen NA, Hart GE (1988) Isolation and characterization of wheat-Elytrigia elongata chromosome 3E and 5E addition and substitution lines. Genome 30:519–524
Author information
Authors and Affiliations
Additional information
Communicated by G. E. Hart
Rights and permissions
About this article
Cite this article
Dubcovsky, J., Galvez, A.F. & Dvořák, J. Comparison of the genetic organization of the early salt-stress-response gene system in salt-tolerant Lophopyrum elongatum and salt-sensitive wheat. Theoret. Appl. Genetics 87, 957–964 (1994). https://doi.org/10.1007/BF00225790
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00225790