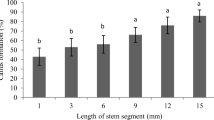
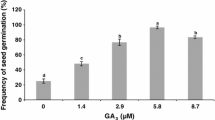
Abstract
In order to know the genetic differences of de- and redifferentiation capacities, seven Brassica species (B. campestris, B. nigra, B. oleracea, B. hirta, B. carinata, B. juncea and B. napus) were cultured in vitro, and their response to the medium supplemented with various combinations of auxin and cytokinin hormones was compared. Important factors for callus initiation were shown to be auxin and species. Calli were induced most frequently in Murashige and Skoog (MS) medium with 1.0 mg/l 2,4-dichlorophenoxyacetic acid (2,4-D), whereas α-naphthaleneacetic acid (NAA) induced preferentially roots at a concentration of 2 to 5 mg/l. Callus-, root- and shoot-forming capacities from explanted cotyledon tissues were significantly different among the seven Brassica species. Calli derived from cotyledons and hypocotyls of seven species were transferred to MS media with 20g/l sucrose, 0 to 0.1 mg/l NAA and 0 to 4 mg/l kinetin to compare their regeneration capacities. Among the seven species tested, B. napus (2n=4x=38, genome AACC) had the highest shoot forming capacity (20.0%). Other amphiploid species, B. carinata (2n=4x=34, BBCC) and B. juncea (2n=4x=36, AABB) formed shoots at low frequencies (2.8% and 1.2%, respectively). A diploid species, B. oleracea (2n=2x 18, CC) also showed high shoot formation (10.2% on average). This result suggests that the gene(s) controlling shoot formation may be localized in the C genome. Differences were also found among varieties and cultivars within a species. One of the cultivars, Siberian kale (B. oleracea var. acephala) gave about 50% shoot formation. This kale was shown cytologically to be an autotetraploid (2n=4x=36, CCCC), thus probably possessing a double set of the shoot-forming gene(s).
Similar content being viewed by others
References
Bajaj YPS, Nitsch P (1975) In vitro propagation of red cabbage (Brassica oleracea L. var. capitata). J Exp Bot 26: 883–890
Baroncelli S, Buiatti M, Bennici A (1973) Genetics of growth and differentiation in vitro of Brassica oleracea var. botrytis. I. Differences between 6 inbred lines. Z Pflanzenzuchtg 70: 99–107
Buiatti M, Baroncelli S, Bennici A (1974) Genetics of growth and differentiation in vitro of Brassica oleracea var. botrytis. IV. Genotype-hormone interactions. Z Pflanzenzuchtg 73: 298–302
Dietert MF, Barron SA, Yoder OC (1982) Effects of genotypes on in vitro culture in the genus Brassica. Plant Sci Lett 26: 233–240
Evans DA, Sharp WR, Flick CE (1981) Plant regeneration from cell cultures. Hort Rev 3: 214–314
Evans DA, Sharp WR, Flick CE (1981) Growth and behaviour of cell culture: embryogenesis and organogenesis. In: Thorpe TA (ed) Plant Tissue Culture: Method and Application in Agriculture. Academic Press, New York, pp 45–113
Frankenberger EA, Hasegawa PM, Tigchelaar EC (1981) Influence of environment and developmental state on the shoot-forming capacity of tomato genotypes. Z Pflanzenphysiol 102: 221–232
Green CE (1977) Prospects for crop improvement in the field of cell culture. Hort Sci 12: 131–134
Horak J, Lustinec J, Mesicek J, Kaminek M, Polackova D (1975) Regeneration of diploid and polyploid plants from the stem pith explants of diploid marrow stem kale (Brassica oleracea L.). Ann Bot 39: 571–577
Kartha KK, Michayluk MR, Kao KN, Gamborg OL, Constabel F (1974) Callus formation and plant regeneration from mesophyll protoplasts of rape plants (Brassica napus cv. Zephyr). Plant Sci Lett 3: 265–271
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15: 473–497
Ogura H, Tsuji S (1977) Differential responses of Nicotiana tabacum L. and its putative progenitors to de- and redifferentiation. Z Pflanzenphysiol 83: 419–426
Reinert J, Bajaj YPS, Zbell B (1977) Aspects of organization-organogenesis, embryogenesis, cytodifferentiation. In: Street HE (ed) Plant Tissue and Cell Culture. 2nd ed, Univ of California Press, Berkeley, Los Angeles, pp 389–427
Schenk HR (1981) Plant regeneration from protoplasts of Brassica oleracea. Cruciferae Newsl 6: 23–24
Schenk HR, Robbelen G (1982) Somatic hybrids by fusion of protoplasts from Brassica oleracea and B. campestris. Z Pflanzenzuchtg 89: 278–288
Thomas E, Hoffmann F, Potrykus I, Wenzel G (1976) Protoplast regeneration and stem embryogenesis of haploid androgenetic rape. Mol Gen Genet 145: 245–248
U N (1935) Genomic analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jap J Bot 7: 389–452
Xu Z, Davey MR, Cocking EC (1982) Plant regeneration from root protoplasts of Brassica. Plant Sci Lett 24: 117–121
Yeomann MacLod (1977) Tissue (callus) cultures-techniques. In: Street HE (ed) Plant Tissue and Cell Culture. 2nd ed, Univ of California Press, Berkeley, Los Angeles, pp 31–59
Zee SY, Hui LH (1977) In vitro plant regeneration from hypocotyl and cotyledons of chinese kale (Brassica alboglabra Bailey). Z Pflanzenphysiol 82: 440–445
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Murata, M., Orton, T.J. Callus initiation and regeneration capacities in Brassica species. Plant Cell Tiss Organ Cult 11, 111–123 (1987). https://doi.org/10.1007/BF00041844
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00041844