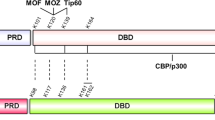
Abstract
Acetylation has been shown to be an important posttranslational modification (PTM) of both histone and nonhistone proteins with particular implications in cell signaling and transcriptional regulation of gene expression. Many studies have already demonstrated that SIRT1 is able to deacetylate histones and lead to gene silencing. It can also regulate the function of tumor suppressors including FOXO proteins and p53 by deacetylation. Here, we describe three experimental approaches for studying the modulation of the acetylation status of some of the known downstream targets of SIRT1.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Shore D, Squire M, Nasmyth KA (1984) Characterization of two genes required for the position-effect control of yeast mating-type genes. EMBO J 3(12):2817–2823
Kaeberlein M, McVey M, Guarente L (1999) The SIR2/3/4 complex and SIR2 alone promote longevity in Saccharomyces cerevisiae by two different mechanisms. Genes Dev 13(19):2570–2580
Liszt G et al (2005) Mouse Sir2 homolog SIRT6 is a nuclear ADP-ribosyltransferase. J Biol Chem 280(22):21313–21320
Vaquero A et al (2004) Human SirT1 interacts with histone H1 and promotes formation of facultative heterochromatin. Mol Cell 16(1):93–105
Luo J et al (2000) Deacetylation of p53 modulates its effect on cell growth and apoptosis. Nature 408(6810):377–381
Brunet A et al (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science 303(5666):2011–2015
Motta MC et al (2004) Mammalian SIRT1 represses forkhead transcription factors. Cell 116(4):551–563
Huffman DM et al (2007) SIRT1 is significantly elevated in mouse and human prostate cancer. Cancer Res 67(14):6612–6618
Eades G et al (2011) miR-200a regulates SIRT1 expression and epithelial to mesenchymal transition (EMT)-like transformation in mammary epithelial cells. J Biol Chem 286(29):25992–26002
Stunkel W et al (2007) Function of the SIRT1 protein deacetylase in cancer. Biotechnol J 2(11):1360–1368
Bradbury CA et al (2005) Histone deacetylases in acute myeloid leukaemia show a distinctive pattern of expression that changes selectively in response to deacetylase inhibitors. Leukemia 19(10):1751–1759
Vaziri H et al (2001) hSIR2(SIRT1) functions as an NAD-dependent p53 deacetylase. Cell 107(2):149–159
Luo J et al (2001) Negative control of p53 by Sir2alpha promotes cell survival under stress. Cell 107(2):137–148
Lain S et al (2008) Discovery, in vivo activity, and mechanism of action of a small-molecule p53 activator. Cancer Cell 13(5):454–463
Peck B et al (2010) SIRT inhibitors induce cell death and p53 acetylation through targeting both SIRT1 and SIRT2. Mol Cancer Ther 9(4):844–855
Wang RH et al (2008) Impaired DNA damage response, genome instability, and tumorigenesis in SIRT1 mutant mice. Cancer Cell 14(4):312–323
Herranz D et al (2010) Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nat Commun 1:3
Wang RH et al (2008) Interplay among BRCA1, SIRT1, and Survivin during BRCA1-associated tumorigenesis. Mol Cell 32(1):11–20
Brunet A et al (2004) Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Sci Aging Knowl Environ 2004(8):2
Khongkow M et al (2013) SIRT6 modulates paclitaxel and epirubicin resistance and survival in breast cancer. Carcinogenesis 34(7):1476–1486
Zhang T, Kraus WL (2010) SIRT1-dependent regulation of chromatin and transcription: linking NAD+ metabolism and signaling to the control of cellular functions. Biochim Biophys Acta 1804(8):1666–1675
Yuan J et al (2009) Histone H3-K56 acetylation is important for genomic stability in mammals. Cell Cycle 8(11):1747–1753
Daitoku H et al (2004) Silent information regulator 2 potentiates Foxo1-mediated transcription through its deacetylase activity. PNAS 101(27):10042–10047
Lin Z, Fang D (2013) The roles of SIRT1 in cancer. Genes Cancer 421(2):384–388
Bouras T et al (2005) SIRT1 deacetylation and repression of p300 involves lysine residues 1020/1024 within the cell cycle regulatory domain 1. J Biol Chem 280(11):10264–10276
Qiang L et al (2012) Brown remodeling of white adipose tissue by SIRT1-dependent deacetylation of Ppary. Cell 150(3):620–632
Pestell R et al (2013) Ppary deacetylation by SIRT1 determines breast tumour lipid synthesis and growth. Cancer Res 73:2-06-02
Yeung F et al (2004) Modulation of NF-κB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBOJ 23(12):2369–2380
Pickard A, Wong PP, McCance DJ (2010) Acetylation of Rb by PCAF is required for nuclear localization and keratinocyte differentiation. J Cell Sci 123:3718–3726
Menssen A et al (2012) The c-MYC oncoprotein, the NAMPT enzyme, the SIRT1-inhibitor DBC1, and the SIRT1-inhibitor DBC1, and the SIRT1 deacetylase form a positive feedback loop. PNAS 109(4):187–196
Bharathy N, Taneja R (2012) Methylation muscles into transcription factor silencing. Transcription 3(5):215–220
Zhao X et al (2005) Regulation of MEF2 by histone deacetylase 4- and SIRT1 deacetylase-mediated lysine modifications. Mol Cell Biol 25(19):8456–8464
Cheng HL et al (2003) Developmental defects and p53 hyperacetylation in Sir2 homolog (SIRT1)-deficient mice. PNAS 100(19):10794–10799
Dehennaut V et al (2012) Molecular dissection of the interaction between HIC1 and SIRT1. Biochem Biophys Res Commun 421(2):384–388
Cohen HY et al (2004) Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Sci Express 10(1126):1–4
Fan W, Luo J (2010) SIRT1 regulates UV-induced DNA repair through deacetylating XPA. Mol Cell 39(2):247–258
Yuan Z et al (2007) SIRT1 regulates the function of the Nijmegen breakage syndrome protein. Mol Cell 27(1):149–162
Westerheide SD et al (2009) Stress-inducible regulation of heat shock factor 1 by the deacetylase SIRT1. Science 323(5917):1063–1066
Tiberi L et al (2012) BCL6 controls neurogenesis through SIRT1-dependent epigenetic repression of selective notch targets. Nat Neurosci 15(12):1627–1635
Inoue Y et al (2007) Smad3 is acetylated by p300/CBP to regulate its transactivation activity. Oncogene 26:500–508
Chen Y et al (2012) Quantitative acetylome analysis reveals the roles of SIRT1 in regulating diverse substrates and cellular pathways. Am Soc Biochem Mol Biol 11(10):1048–1062
Nakagawa T, Guarente L (2011) Sirtuins at a glance. J Cell Sci 124:833–838
Ikenoue T, Inoki K, Zhao B (2008) PTEN acetylation modulates its interaction with PDZ domain. Cancer Res 68:6908–6912
Montie HL, Pestell RG, Merry DE (2011) SIRT1 modulates aggregation and toxicity through deacetylation of the androgen receptor in cell models of SBMA. J Neurosci 21(48):17425–17436
Akieda-Asai S et al (2010) SIRT1 regulates thyroid-stimulating hormone release by enhancing PIP5Kγ activity through deacetylation of specific lysine residues in mammals. PLoS One 5(7)
Chen IY et al (2006) Histone H2A.z is essential for cardiac myocyte hypertrophy but opposed by silent information regulator 2alpha. J Biol Chem 281(8):19369–19377
Yu J, Auwerx J (2010) Protein deacetylation by SIRT1: an emerging key post-translational modification in metabolic regulation. Pharmacol Res 62(1):35–41
Peng L et al (2011) SIRT1 deacetylates the DNA Methyltransferase 1 (DNMT1) protein and alters its activities. Mol Cell Biol 31:4720–4734
Hallows WC, Lee S, Denu JM (2006) Sirtuins deacetylate and activate mammalian acetyl-CoA synthetases. PNAS 103(27):10230–10235
Fusco S, Maulucci G, Pani G (2012) Sirt1: Def-eating senescence? Cell Cycle 11(22):4135–4146
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer Science+Business Media New York
About this protocol
Cite this protocol
Gomes, A.R. et al. (2016). Sirtuin1 (SIRT1) in the Acetylation of Downstream Target Proteins. In: Sarkar, S. (eds) Histone Deacetylases. Methods in Molecular Biology, vol 1436. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-3667-0_12
Download citation
DOI: https://doi.org/10.1007/978-1-4939-3667-0_12
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-3665-6
Online ISBN: 978-1-4939-3667-0
eBook Packages: Springer Protocols