Abstract
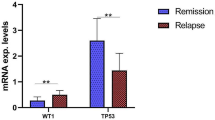
We have previously shown the clinical usefulness of Wilms’ tumor 1 gene (WT1) mRNA expression in peripheral blood (PB) as a minimal residual disease (MRD) monitoring marker in 191 acute myeloid leukemia (AML) patients using the WT1 mRNA assay kit “Otsuka” (Otsuka Pharmaceutical Co., Ltd.; “former kit”). In contrast, the usefulness of WT1 mRNA expression in bone marrow (BM) has been investigated in only a limited number of subjects using former kit. Following that previous study, a next-generation kit, WT1 mRNA assay kit II “Otsuka” (Otsuka Pharmaceutical Co., Ltd.; “new kit”) has been newly developed. In the present study, we aimed to evaluate the performance of the new kit and to investigate the clinical usefulness of WT1 mRNA expression in BM. The PB and BM were collected on the same day from 164 blood disease patients, including 118 AML patients. WT1 mRNA expression was determined using the new and former kits and the values obtained were compared. The performance of new kit was shown to be equivalent to that of former kit. As reported in PB, WT1 mRNA expression in BM was found to be a useful marker for monitoring disease status as well as for a diagnosis of early stage relapse in AML patients.




Similar content being viewed by others
References
Call KM, Glaser T, Ito CY, Buckler AJ, Pelletier J, Haber DA, et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms’ tumor locus. Cell. 1990;60:509–20.
Gessler M, Poustka A, Cavenee W, Neve RL, Orkin SH, Bruns GA. Homozygous deletion in Wilms tumors of a zinc-finger gene identified by chromosome jumping. Nature. 1990;343:774–8.
Rauscher FJ 3rd. The WT1 Wilms tumor gene product: a developmentally regulated transcription factor in the kidney that functions as a tumor suppressor. FASEB J. 1993;7:896–903.
Inoue K, Sugiyama H, Ogawa H, Nakagawa M, Yamagami T, Miwa H, et al. WT1 as a new prognostic factor and a new marker for the detection of minimal residual disease in acute leukemia. Blood. 1994;84:3071–9.
Oji Y, Ogawa H, Tamaki H, Oka Y, Tsuboi A, Kim EH, et al. Expression of the Wilms’ tumor gene WT1 in solid tumors and its involvement in tumor cell growth. Jpn J Cancer Res. 1999;90:194–204.
Sugiyama H. Wilms’ tumor gene WT1: its oncogenic function and clinical application. Int J Hematol. 2001;73:177–87.
Inoue K, Ogawa H, Yamagami T, Soma T, Tani Y, Tatekawa T, et al. Long-term follow-up of minimal residual disease in leukemia patients by monitoring WT1 (Wilms tumor gene) expression levels. Blood. 1996;88:2267–78.
Miyawaki S, Emi N, Mitani K, Oyashiki K, Kitamura K, Morishita T. Clinical course of the disease and the level of WT1 mRNA in patients with acute myeloid leukemia (AML): joint research by 23 institutions in Japan. Jpn J Clin Hematol. 2005;46(12):1279–87.
Trka J, Kalinová M, Hrusák O, Zuna J, Krejcí O, Madzo J, et al. Real-time quantitative PCR detection of WT1 gene expression in children with AML: prognostic significance, correlation with disease status and residual disease detection by flow cytometry. Leukemia. 2002;16(7):1381–9.
Cilloni D, Gottardi E, De Micheli D, Serra A, Volpe G, Messa F, et al. Quantitative assessment of WT1 expression by real time quantitative PCR may be a useful tool for monitoring minimal residual disease in acute leukemia patients. Leukemia. 2002;16(10):2115–21.
Tamaki H, Mishima M, Kawakami M, Tsuboi A, Kim EH, Hosen N, et al. Monitoring minimal residual disease in leukemia using real-time quantitative polymerase chain reaction for Wilms tumor gene (WT1). Int J Hematol. 2003;78(4):349–56.
Østergaard M, Olesen LH, Hasle H, Kjeldsen E, Hokland P. WT1 gene expression: an excellent tool for monitoring minimal residual disease in 70 % of acute myeloid leukaemia patients—results from a single-centre study. Br J Haematol. 2004;125:590–600.
Weisser M, Kern W, Rauhut S, Schoch C, Hiddemann W, Haferlach T, et al. Prognostic impact of RT-PCR-based quantification of WT1 gene expression during MRD monitoring of acute myeloid leukemia. Leukemia. 2005;19:1416–23.
Kitamura K, Nakano Y, Watamoto K, Koga D, Naoe T. Significance of monitoring WT1 mRNA levels of peripheral blood and bone marrow in acute myeloid leukemia. Jpn J Clin Hematol. 2010;51(12):1748–55.
Cilloni D, Renneville A, Hermitte F, Hills RK, Daly S, Jovanovic JV, et al. Real-time quantitative polymerase chain reaction detection of minimal residual disease by standardized WTl assay to enhance risk stratification in acute myeloid leukemia: a European leukemia net study. J Clin Oncol. 2009;27(31):5195–201.
Miyawaki S, Hatsumi N, Tamaki T, Naoe T, Ozawa K, Kitamura K, et al. Prognostic potential of detection of WT1 mRNA level in peripheral blood in adult acute myeloid leukemia. Leuk Lymphoma. 2010;51(10):1855–61.
Ommen HB, Nyvold CG, Braendstrup K, Andersen BL, Ommen IB, Hasle H, et al. Relapse prediction in acute myeloid leukaemia patients in complete remission using WT1 as a molecular marker: development of a mathematical model to predict time from molecular to clinical relapse and define optimal sampling intervals. Br J Haematol. 2008;141(6):782–91.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This study was funded by Otsuka Pharmaceutical Co., Ltd. K.K. received research funding from Otsuka during the conduct of the study. T. Nishiyama, S.M., K.M., K.S., H.M., M.O., H.O., K. Imai, H.K., Y.Y., S.C., T.H., Y.M., Y.H., J.T., Y. N., M.K. and Y.U. received research funding from Otsuka during the conduct of the study. K. Ishiyama and T. Naoe received research funding and personal fees from Otsuka during the conduct of the study. H.S. received personal fees from Otsuka during the conduct of the study. D.K. is employee of Otsuka. F.T. declares no conflict of interest.
About this article
Cite this article
Kitamura, K., Nishiyama, T., Ishiyama, K. et al. Clinical usefulness of WT1 mRNA expression in bone marrow detected by a new WT1 mRNA assay kit for monitoring acute myeloid leukemia: a comparison with expression of WT1 mRNA in peripheral blood. Int J Hematol 103, 53–62 (2016). https://doi.org/10.1007/s12185-015-1882-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-015-1882-1