Abstract
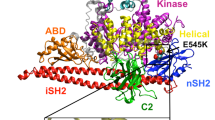
New developments regarding the structure and in vivo dynamics of protein kinase B (PKB/Akt) have been recently exposed. Here, we specifically review how the use of multi-disciplinary approaches has resulted in reaching the recent progress made to relate the quaternary structure of PKB to its in vivo function. Using X-ray crystallography, the structure of PKB pleckstrin homology (PH) and kinase domains was determined separately. The molecular mechanisms involved in (a) the binding of the phosphoinositides to the PH domain and (b) the activation of the kinase with the rearrangement of the catalytic site and substrate binding were determined. In vitro, nuclear magnetic resonance and circular dychroism studies gave complementary information on the interaction of the PH domain with the phosphoinositides. However, the molecular nature and the function of the interactions between the PKB domains could not be deduced from the X-ray data since the full-length PKB has not been crystallised. In vitro, dynamic information on the inter-domain conformational changes related to PKB activation states emerged with the use of tandem mass spectrometry. Cell imaging and Förster resonance energy transfer provided in vivo dynamics. Molecular modelling and dynamic simulations in conjunction with mutagenesis and biochemical analysis were used to investigate the complex interactions between the PKB domains in vivo and understand at the molecular level how it linked to its activity. The compilation of the information obtained on the 3-D structure and the spatiotemporal dynamics of this widely studied oncogene could be applied to the study of other proteins. This inter-disciplinary approach led to a more profound understanding of PKB complex activation mechanism in vivo that will shed light onto new ideas and possibilities for modulating its activity.





Similar content being viewed by others
References
Alcor D, Calleja V, Larijani B (2009) Revealing signaling in single cells by single- and two-photon fluorescence lifetime imaging microscopy. Methods Mol Biol 462:307
Alessi DR, Andjelkovic M, Caudwell B (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. Embo J 15(23):6541
Alessi DR, Deak M, Casamayor A (1997) 3-Phosphoinositide-dependent protein kinase-1 (PDK1): structural and functional homology with the Drosophila DSTPK61 kinase. Curr Biol 7(10):776
Ananthanarayanan B, Fosbrink M, Rahdar M (2007) Live-cell molecular analysis of Akt activation reveals roles for activation loop phosphorylation. J Biol Chem 282(50):366334
Ananthanarayanan B, Ni Q, Zhang J (2005) Signal propagation from membrane messengers to nuclear effectors revealed by reporters of phosphoinositide dynamics and Akt activity. Proc Natl Acad Sci U S A 102(42):15081
Andjelkovic M, Alessi DR, Meier R (1997) Role of translocation in the activation and function of protein kinase B. J Biol Chem 272(50):31515
Andjelkovic M, Jakubowicz T, Cron P (1996) Activation and phosphorylation of a pleckstrin homology domain containing protein kinase (RAC-PK/PKB) promoted by serum and protein phosphatase inhibitors. Proc Natl Acad Sci U S A 93(12):5699
Auguin D, Barthe P, Auge-Senegas MT (2004) Solution structure and backbone dynamics of the pleckstrin homology domain of the human protein kinase B (PKB/Akt). Interaction with inositol phosphates. J Biomol NMR 28(2):137
Barnett SF, Bilodeau MT, Lindsley CW (2005a) The Akt/PKB family of protein kinases: a review of small molecule inhibitors and progress towards target validation. Curr Top Med Chem 5(2):109
Barnett SF, Defeo-Jones D, Fu S (2005b) Identification and characterization of pleckstrin-homology-domain-dependent and isoenzyme-specific Akt inhibitors. Biochem J 385(Pt 2):399
Bellacosa A, Chan TO, Ahmed NN (1998) Akt activation by growth factors is a multiple-step process: the role of the PH domain. Oncogene 17(3):313
Brazil DP, Hemmings BA (2001) Ten years of protein kinase B signalling: a hard Akt to follow. Trends Biochem Sci 26(11):657
Brugge J, Hung MC, Mills GB (2007) A new mutational AKTivation in the PI3K pathway. Cancer Cell 12(2):104
Calleja V, Alcor D, Laguerre M (2007) Intramolecular and intermolecular interactions of protein kinase B define its activation in vivo. PLoS Biol 5(4):e95
Calleja V, Ameer-Beg SM, Vojnovic B (2003) Monitoring conformational changes of proteins in cells by fluorescence lifetime imaging microscopy. Biochem J 372(Pt 1):33
Calleja V, Laguerre M, Parker PJ (2009) Role of a novel PH-kinase domain interface in PKB/Akt regulation: structural mechanism for allosteric inhibition. PLoS Biol 7(1):e17
Cameron AJ, De Rycker M, Calleja V (2007) Protein kinases, from B to C. Biochem Soc Trans 35(Pt 5):1013
Chan TO, Rittenhouse SE, Tsichlis PN (1999) AKT/PKB and other D3 phosphoinositide-regulated kinases: kinase activation by phosphoinositide-dependent phosphorylation. Annu Rev Biochem 68:965
Chen R, Kim O, Yang J (2001) Regulation of Akt/PKB activation by tyrosine phosphorylation. J Biol Chem 276(34):31858
Cicenas J (2008) The potential role of Akt phosphorylation in human cancers. Int J Biol Markers 23(1):1
Collins BJ, Deak M, Arthur JS (2003) In vivo role of the PIF-binding docking site of PDK1 defined by knock-in mutation. EMBO J 22(16):4202
Collins BJ, Deak M, Murray-Tait V (2005) In vivo role of the phosphate groove of PDK1 defined by knockin mutation. J Cell Sci 118(Pt 21):5023
Conus NM, Hannan KM, Cristiano BE (2002) Direct identification of tyrosine 474 as a regulatory phosphorylation site for the Akt protein kinase. J Biol Chem 277(41):38021
Feng J, Park J, Cron P (2004) Identification of a PKB/Akt hydrophobic motif Ser-473 kinase as DNA-dependent protein kinase. J Biol Chem 279(39):41189
Ferguson KM, Kavran JM, Sankaran VG (2000) Structural basis for discrimination of 3-phosphoinositides by pleckstrin homology domains. Mol Cell 6(2):373
Gambhir A, Hangyas-Mihalyne G, Zaitseva I (2004) Electrostatic sequestration of PIP2 on phospholipid membranes by basic/aromatic regions of proteins. Biophys J 86(4):2188
Gao T, Furnari F, Newton AC (2005) PHLPP: a phosphatase that directly dephosphorylates Akt, promotes apoptosis, and suppresses tumor growth. Mol Cell 18(1):13
Gold MG, Barford D, Komander D (2006) Lining the pockets of kinases and phosphatases. Curr Opin Struct Biol 16(6):693
Green CJ, Goransson O, Kular GS et al (2008) Use of Akti and a drug-resistant mutant validates a critical role for PKB/Akt in the insulin-dependent regulation of glucose and system A amino acid uptake. J Biol Chem 283:27653–27667
Hauge C, Antal TL, Hirschberg D (2007) Mechanism for activation of the growth factor-activated AGC kinases by turn motif phosphorylation. EMBO J 26(9):2251
Huang BX, Kim HY (2006) Interdomain conformational changes in Akt activation revealed by chemical cross-linking and tandem mass spectrometry. Mol Cell Proteomics 5(6):1045
Huang X, Begley M, Morgenstern KA (2003) Crystal structure of an inactive Akt2 kinase domain. Structure 11(1):21
Ikenoue T, Inoki K, Yang Q (2008) Essential function of TORC2 in PKC and Akt turn motif phosphorylation, maturation and signalling. EMBO J 27(14):1919
Jeffrey PD, Russo AA, Polyak K (1995) Mechanism of CDK activation revealed by the structure of a cyclinA–CDK2 complex. Nature 376(6538):313
Kannan N, Haste N, Taylor SS (2007) The hallmark of AGC kinase functional divergence is its C-terminal tail, a cis-acting regulatory module. Proc Natl Acad Sci U S A 104(4):1272
Kim C, Xuong NH, Taylor SS (2005) Crystal structure of a complex between the catalytic and regulatory (RIalpha) subunits of PKA. Science 307(5710):690
Knighton DR, Zheng JH, Ten Eyck LF (1991) Crystal structure of the catalytic subunit of cyclic adenosine monophosphate-dependent protein kinase. Science 253(5018):407
Kunkel MT, Ni Q, Tsien RY (2005) Spatio-temporal dynamics of protein kinase B/Akt signaling revealed by a genetically encoded fluorescent reporter. J Biol Chem 280(7):5581
Laguerre M, Saux M, Dubost J-P (1997) MLPP: a program for the calculation of molecular lipophilicity in proteins. Pharm. Sci 3:217
Lasserre R, Guo XJ, Conchonaud F (2008) Raft nanodomains contribute to Akt/PKB plasma membrane recruitment and activation. Nat Chem Biol 4(9):538
Lindsley CW, Zhao Z, Leister WH (2005) Allosteric Akt (PKB) inhibitors: discovery and SAR of isozyme selective inhibitors. Bioorg Med Chem Lett 15(3):761
Manning BD, Cantley LC (2007) AKT/PKB signaling: navigating downstream. Cell 129(7):1261
Meier R, Thelen M, Hemmings BA (1998) Inactivation and dephosphorylation of protein kinase Balpha (PKBalpha) promoted by hyperosmotic stress. EMBO J 17(24):7294
Milburn CC, Deak M, Kelly SM (2003) Binding of phosphatidylinositol 3,4,5-trisphosphate to the pleckstrin homology domain of protein kinase B induces a conformational change. Biochem J 375(Pt 3):531
Mora A, Komander D, van Aalten DM (2004) PDK1, the master regulator of AGC kinase signal transduction. Semin Cell Dev Biol 15(2):161
Newton AC (2003) Regulation of the ABC kinases by phosphorylation: protein kinase C as a paradigm. Biochem J 370(Pt 2):361
Nolen B, Taylor S, Ghosh G (2004) Regulation of protein kinases; controlling activity through activation segment conformation. Mol Cell 15(5):661
Obata T, Yaffe MB, Leparc GG (2000) Peptide and protein library screening defines optimal substrate motifs for AKT/PKB. J Biol Chem 275(46):36108
Parsons DW, Wang TL, Samuels Y (2005) Colorectal cancer: mutations in a signalling pathway. Nature 436(7052):792
Pellicena P, Kuriyan J (2006) Protein–protein interactions in the allosteric regulation of protein kinases. Curr Opin Struct Biol 16(6):702
Sable CL, Filippa N, Filloux C (1998) Involvement of the pleckstrin homology domain in the insulin-stimulated activation of protein kinase B. J Biol Chem 273(45):29600
Sarbassov DD, Guertin DA, Ali SM (2005) Phosphorylation and regulation of Akt/PKB by the rictor–mTOR complex. Science 307(5712):1098
Sasaki K, Sato M, Umezawa Y (2003) Fluorescent indicators for Akt/protein kinase B and dynamics of Akt activity visualized in living cells. J Biol Chem 278(33):30945
Scheid MP, Woodgett JR (2001) PKB/AKT: functional insights from genetic models. Nat Rev Mol Cell Biol 2(10):760
Shi Z, Resing KA, Ahn NG (2006) Networks for the allosteric control of protein kinases. Curr Opin Struct Biol 16(6):686
Stephens L, Anderson K, Stokoe D (1998) Protein kinase B kinases that mediate phosphatidylinositol 3,4,5-trisphosphate-dependent activation of protein kinase B. Science 279(5351):710
Testa JR, Tsichlis PN (2005) AKT signaling in normal and malignant cells. Oncogene 24(50):7391
Thomas CC, Deak M, Alessi DR (2002) High-resolution structure of the pleckstrin homology domain of protein kinase b/akt bound to phosphatidylinositol (3,4,5)-trisphosphate. Curr Biol 12(14):1256
Toker A, Newton AC (2000) Akt/protein kinase B is regulated by autophosphorylation at the hypothetical PDK-2 site. J Biol Chem 275(12):8271
Tokunaga E, Oki E, Egashira A (2008) Deregulation of the Akt pathway in human cancer. Curr Cancer Drug Targets 8(1):27
Yang J, Cron P, Good VM (2002a) Crystal structure of an activated Akt/protein kinase B ternary complex with GSK3-peptide and AMP-PNP. Nat Struct Biol 9(12):940
Yang J, Cron P, Thompson V (2002b) Molecular mechanism for the regulation of protein kinase B/Akt by hydrophobic motif phosphorylation. Mol Cell 9(6):1227
Zhang J, Allen MD (2007) FRET-based biosensors for protein kinases: illuminating the kinome. Mol Biosyst 3(11):759
Zhang L, Lee KC, Bhojani MS (2007) Molecular imaging of Akt kinase activity. Nat Med 13(9):1114
Zhao Z, Leister WH, Robinson RG (2005) Discovery of 2,3,5-trisubstituted pyridine derivatives as potent Akt1 and Akt2 dual inhibitors. Bioorg Med Chem Lett 15(4):905
Acknowledgements
We would like to thank Dr. Richard Byrne for the critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Calleja, V., Laguerre, M. & Larijani, B. 3-D structure and dynamics of protein kinase B—new mechanism for the allosteric regulation of an AGC kinase. J Chem Biol 2, 11–25 (2009). https://doi.org/10.1007/s12154-009-0016-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12154-009-0016-8