Abstract
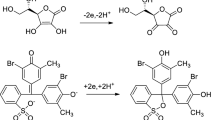
A fluorophotometric method for the determination of hydrogen peroxide (H2O2) using fluorescin was developed. This method was based on the oxidative reaction of fluorescin, a colorless, non-fluorescent lactoid fluorescein, by H2O2 to give highly fluorescein fluorescence emission. In the determination of H2O2, the calibration curve exhibited linearity over the H2O2 concentration range of 1.5–310 ng mL−1 at an emission wavelength of 525 nm with an excitation of 500 nm and with relative standard deviations (n = 6) of 2.51%, 2.48%, and 1.31% for 3.1 ng mL−1, 30.8 ng mL−1, and for 308 ng mL−1 of H2O2, respectively. The detection limit for H2O2 was 1.9 ng mL−1 six blank determinations was performed (ρ = 6). This proposed method was applied to detection of other reactive oxygen species and nitrogen species (ROS/RNS) such as singlet oxygen (1O2), hydroxyl radical (•OH), peroxynitrite (ONOO−) etc., and it was possible to detect them with a high sensitivity. In addition, this proposed method was applied to the recovery tests of H2O2 in calf serum, human saliva, rain water, and wheat noodles; the results were satisfactory.



Similar content being viewed by others
References
Ohshima H, Tatemichi M, Sawa T (2003) Chemical basis of inflammation-induced carcinogenesis. Arch Biochem Biophys 417:3–11. doi:10.1016/S0003-9861(03)00283-2
Shah AM, Channon KM (2004) Free radicals and redox signalling in cardiovascular disease. Heart 904:86
Kojo S (2005) Vitamine 79:334–337
Eunah Yu, E.B.M.K.I.Y.S.Y (2001) Development for the analysis of reactive oxygen species using capillary electrophoresis with laser-induced fluorescence detection. J Microcolumn Sep 13:327–331. doi:10.1002/mcs.10014
Gabbita SP, Robinson KA, Stewart CA, Floyd RA, Hensley K (2000) Redox Regulatory Mechanisms of Cellular Signal Transduction. Arch Biochem Biophys 376:1–13. doi:10.1006/abbi.1999.1685
Aruoma OI, Halliwell B (1998) Molecular biology of free radicals in human diseases. OICA International
Balaban RS, Nemoto S, Finkel T (2005) Mitochondria, oxidants, and aging. Cell February 25(120):483–495
Beckman KB, Ames BN (1998) The free radical theory of aging matures. Physiol Rev 78:547–581
Yamashiro N, Uchida S, Satoh Y, Morishima Y, Yokoyama H, Satoh T, Sugama J, Yamada R (2004) Determination of hydrogen peroxide in water by chemiluminescence detection. J Nucl Sci Technol 41:890–897. doi:10.3327/jnst.41.890
Zappacosta B, Persichilli S, Mormile F, Minucci A, Russo A, Giardina B, De Sole P (2001) A fast chemiluminescent method for H2O2 measurement in exhaled breath condensate. Clin Chim Acta 310:187–191. doi:10.1016/S0009-8981(01) 00571-X
Chai XS, Hou QX, Luo Q, Zhu JY (2004) Rapid determination of hydrogen peroxide in the wood pulp bleaching streams by a dual-wavelength spectroscopic method. Anal Chim Acta 507:285–288. doi:10.1016/j.aca.2003.11.036
Deiana L, Carru C, Pes G, Tadolini B (1999) Spectrophotometric measurement of hydroperoxides at increased sensitivity by oxidation of Fe2+ in the presence of xylenol orange. Free Radic Res 31:237–244. doi:10.1080/10715769900300801
Vieira IC (1998) Flow injection spectrophotometric determination of hydrogen eroxide using a crude extract of zucchini (Cucurbita pepo) as a source of peroxidase. Analyst (Lond) 123:1809–1812. doi:10.1039/a803478h
Amer J, Goldfarb A, Fibach E (2004) Flow cytometric analysis of the oxidative status of normal and thalassemic red blood cells. Cytom Part A 60A:73–80
Amer J, Goldfarb A, Fibach E (2003) Flow cytometric measurement of reactive oxygen species production by normal and thalassaemic red blood cells. Eur J Haematol 70:84–90. doi:10.1034/j.1600-0609.2003.00011.x
Chang MCY, Pralle A, Isacoff EY, Chang CJ (2004) A Selective, cell-permeable optical probe for hydrogen peroxide in living cells. J Am Chem Soc 126:15392. doi:10.1021/ja0441716
Lazrus AL, Kok GL, Gitlin SN, Lind JA, McLaren SE (1985) Automated fluorimetric method for hydrogen peroxide in atmospheric precipitation. Anal Chem 57:917–922. doi:10.1021/ac00281a031
Liu Z, Cai R, Mao L, Huang H, Ma W (1999) Highly sensitive spectrofluorimetric determination of hydrogen peroxide with β-cyclodextrin?hemin as catalyst. Analyst (Lond) 124:173–176. doi:10.1039/a807027j
Maeda H, Fukuyasu Y, Yoshida S, Fukuda M, Saeki K, Matsuno H, Yamauchi Y, Yoshida K, Hirata K, Miyamoto K (2004) Fluorescent probes for hydrogen peroxide based on a non-oxidative mechanism. Angew Chem Int Ed 43:2389–2391. doi:10.1002/anie.200452381
Maeda H, Yamamoto K, Nomura Y, Kohno I, Hafsi L, Ueda N, Yoshida S, Fukuda M, Fukuyasu Y, Yamauchi Y (2005) A design of fluorescent probes for superoxide based on a nonredox mechanism. J Am Chem Soc 127:68–69. doi:10.1021/ja047018k
Odo J, Matsumoto K, Shinmoto E, Hatae Y, Shiozaki A (2004) Spectrofluorometric determination of hydrogen peroxide based on oxidative catalytic reactions of p-Hydroxyphenyl derivatives with metal complexes of Thiacalix[4] arenetetrasulfonate on a modified anion-exchanger. Anal Sci 20:707–710. doi:10.2116/analsci.20.707
Setsukinai K, Urano Y, Kakinuma K, Majima HJ, Nagano T (2003) Development of novel fluorescence probes that can reliably detect reactive oxygen species and distinguish specific species. J Biochem 278:3170–3175
Mori I, Takasaki K, Fujita Y, Matsuo T (1998) Selective and sensitive fluorometric determinations of cobalt (II) and hydrogen peroxide with fluorescein-hydrazide. Talanta 47:631–637. doi:10.1016/S0039-9140(98) 00118-0
Nakahara R, Fujimoto T, Doi M, Morita K, Yamaguchi T, Fujita Y (2008) Fluorophotometric determination of hydrogen peroxide and other reactive oxygen species with fluorescein hydrazide (FH) and its crystal structure. Chem Pharm Bull (Tokyo) 56:977–981. doi:10.1248/cpb.56.977
Choi MF, Hawkins P (1995) A novel oxygen and/or carbon dioxide-sensitive optical transducer. Talanta 42:483–492. doi:10.1016/0039-9140(95) 01436-F
McHedlov-Petrossyan NO, Rubtsov MI, Lukatskaya LL (1992) Ionization and Tautomerism of chloro-derivatives of fluorescein in water and aqueous acetone. Dyes Pigments 18:179–198. doi:10.1016/0143-7208(92) 87002-I
Anthoni U, Christophersen C, Nielsen PH, Pschl A, Schaumburg K (1995) Structure of red and orange fluorescein. Struct Chem 6:161–165. doi:10.1007/BF02286443
Mori I, Fujita Y, Fujita K, Nakahashi Y, Tanaka T, Ishihara S (1988) Highly sensitive spectrophotometric determination of cobalt using o-hydroxyhydroquinonephthalein and hydrogen peroxide in the presence of mixed surfactants. Anal Bioanal Chem 330:619–623
Mori I, Fujita Y, Toyoda M, Hamada M, Akagi M (1992) Simple fluorophotometric determination of cobalt (II) with p-hydroxy-2-anilinopyridine and hydrogen peroxide. Anal Bioanal Chem 343:902–904
Mori I, Fujita Y, Toyoda M, Kato K, Yoshida N, Akagi M (1991) Fluorimetric determination of hydrogen peroxide by use of the fluorescence reaction between N-(4'-hydroxyphenyl)-N-(4-methylquinolinyl) amine and Cobalt (II) in the presence of trimethyl stearylammoniumchloride. Talanta 38:683–686. doi:10.1016/0039-9140(91) 80157-U
Mori I, Fujita Y, Toyoda M, Kubo S (1992) Alternative spectrophotometric determination of niobium and tantalum with o-hydroxyhydroquinonephthalein in cationic surfactant micellar media. Anal Bioanal Chem 342:80–82
Nonova D, Stoyanov K (1982) Extraction-spectrophotometric determination of Copper(II) with 4-(2-Pyridylazo) Resorcinol and a long-chain quaternary ammonium salt. Anal Chim Acta 38:321–328. doi:10.1016/S0003-2670(01) 85316-2
Aubry JM, Bouttemy S (1997) Preparative oxidation of organic compounds in microemulsions with singlet oxygen generated chemically by the sodium molybdate/hydrogen peroxide system. J Am Chem Soc 119:5286–5294. doi:10.1021/ja9644079
Acknowledgements
This study was supported by a Grant-in-Aid for High Technology Research from Ministry of Education, Science, Sports and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nakahara, R., Kashitani, S., Hayakawa, K. et al. Fluorophotometric Determination of Hydrogen Peroxide with Fluorescin in the Presence of Cobalt (II) and Reaction Against Other Reactive Oxygen Species. J Fluoresc 19, 769–775 (2009). https://doi.org/10.1007/s10895-009-0473-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-009-0473-z