Abstract
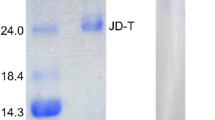
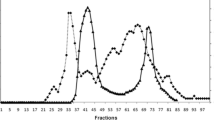
Two trypsin-like enzymes (TLEs) were purified from North Pacific krill (Euphausia pacifica) by ammonium sulfate precipitation, ion-exchange and gel-filtration chromatography. The purified enzymes were identified as trypsins by LC-ESI-MS/MS analysis. The relative molecular mass of TLE I and TLE II were 33 and 32.3 kDa, respectively, with isoelectric points of 4.5 and 4.3, respectively. The TLEs showed excellent thermal stable in the crude extract and the purified TLEs were active over a wide pH (6.0–11.0) and temperature (10–70°C) range. Compared with trypsins from other organisms, the purified TLEs had physiological efficiencies of 1.6–6.7-fold. The difference in Arg, Ile and Asp content might explain why E. pacifica TLEs have good thermal stability and physiological efficiency.



Similar content being viewed by others
References
Anheller JE, Hellgren L, Karlstam B et al (1989) Biochemical and biological profile of a new enzyme preparation from Antarctic krill (E. superba) suitable for debriment of ulcerative lesions. Arch Dermatol Res 281:105–110
Britton KL, Baker PJ, Borges KM et al (1995) Insights into thermal stability from a comparison of the glutamate dehydrogenases from Pyrococcus furiosus and Thernococcus litoralis. Eur J Biochem 229:688–695
Bustos RO, Romo CR, Healy MG (1999) Purification of Trypsin-like enzymes from Antarctic krill processing wastewater. Proc Biochem 35:327–333
Candiano G, Bruschi M, Musante L et al (2004) Blue silver: a very sensitive colloidal Coomassie G-250 staining for proteome analysis. Electrophoresis 25:1327–1333
Erlanger BE, Kokowsky N, Cohen W (1961) The preparation and properties of two new chromogenic substrates of trypsin. Arch Biochem Biophys 95:271–278
Johnston D, Hermans JM, Yellowlees D (1995) Isolation and Characterization of a Trypsin from the Slipper Lobster, Thenus orientalis (Lund). Arch Biochem Biophys 324(1):35–40
Kishimura H, Klomklao S, Benjakul S et al (2007) Characteristics of trypsin from the pyloric ceca of walleye pollock (Theragra chalcogramma). Food Chem. DOI 10.1016/j.foodchem.2007.05.056
Leiros HKS, Willassen NP, Smalâs AO (1999) Residue determinants and sequence analysis of cold-adapted trypsins. Extremophiles 3:205–219
Mekkes J, Le Poole I, Das Pranab K et al (1998) Efficient debridement of necrotic wounds using proteolytic enzymes derived from Antarctic krill: a double blind, placebo-controlled study in a standardized animal wound model. Wound Repair Regen 6(1):50–57
Nakagawa Y, Endo Y, Taki K (2002) Contributions of heterotrophic and autotrophic prey to the diet of Euphausia pacifica Hansen in Sanriku waters off northeastern Japan. Polar Biosci 15:52–65
Nicol S, Endo Y (1997) Krill fisheries of the world. FAO Fish Tech Pap T367
Osnes KK, Mohr V (1985a) Peptide hydrolases of Antarctic krill, Euphausia superba. Comp Biochem Phys B 82:559–606
Osnes KK, Mohr V (1985b) On the purification and characterization of three serine-type peptide hydrolases from Antarctic krill, Euphausia superba. Comp Biochem Phys B 82:607–619
Sainz JC, García-Carreňo FL, Hernández-Cortés P (2004) Penaeus vannamei isotrypsins: purification and characterization. Comp Biochem Phys B 138:155–162
Shevchenko A, Jensen ON, Podtelejnikov AV et al (1996) Linking genome and proteome by mass spectrometry: large-scale identification of yeast proteins from two-dimensional gels. Proc Natl Acad Sci USA 93(25):14440–14445
Suzuki T, Shibata N (1990). The utilization of Antarctic krill for human food. Food Rev Int 6:119–147
Acknowledgements
This work was supported by a grant from the National High Technology Research and Development Program (863 program) of China (No. 2001AA625020) and Department of Science and Technology of Shandong Province. We thank Dr. Mingyu Shao, Guangxu Liu and Yaning Sun for paper writing and revising.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Z., Jiang, G., Xiang, P. et al. Purification and characterization of trypsin-like enzymes from North Pacific krill (Euphausia pacifica). Biotechnol Lett 30, 67–72 (2008). https://doi.org/10.1007/s10529-007-9511-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-007-9511-6