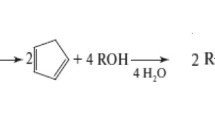
Abstract
We have studied the reaction of 1,1-dichloro-2,2-difluoro-, chlorotrifluoro-, and tetrafluoro-ethylenes and also hexafluoropropylene withcis,trans andtrans,trans isomers of conjugated methyl octadecadienoate. Reactions gave 50-82% yields of cycloaddition products. The pre-ponderance of products with a cyclobutane struc-ture and a double bonda,β to the cyclobutane ring was shown by IR and NMR spectra.
Reactions were carried out in diluent with hy-droquinone inhibitor at autogenous pressure and a temperature of 200C for 5 hr. The distilled adducts are colorless liquids with viscosities rang-ing from 11 to 19 centistokes at 100F. These products and their hydrogenated derivatives ex-hibit low pour points (down to -76F) and may be useful as low-temperature plasticizers or lubricant additives.
Similar content being viewed by others
References
Bartlett, P. D., L. K. Montgomery and B. Scidel, J. Am. Chem. Soc.86, 616–622 (1964).
Coffman, D. D., P. L. Barrick, R. D. Cramer and M. S. Raasch, —Ibid.71, 490–496 (1949).
Danzig, M. J., J. L O’Donnell, E. W. Bell, J. C. Cowan and H. M. Tetter, JAOCS34, 136–138 (1957).
Friedrich, J. P., E. W. Bell and R. E. Beal, —Ibid.39, 420 (1962).
Friedrich, J. P., E. W. Bell and L. E. Gast, —Ibid.42, 643–645 (1965).
Montgomery, L. K., K. Schueller and P. D. Bartlett, J. Am. Chem. Soc.86, 622–628 (1964).
Roberts, J. D., and C. M. Shants, Org. Reactions12, 1–56 (1962).
Schneider, W. J., L. E. Gast and H. M. Teeter, JAOCS41, 605–606 (1964).
Trutnorsky, H., Mikrochlm. Acta1963, 499.
Author information
Authors and Affiliations
Additional information
N. Utiliz. Ees. and Dev. Div., ARS, USDA.
About this article
Cite this article
Bell, E.W., Friedrich, J.P., Gast, L.E. et al. 1,2-Cycloaddition of Haloalkenes to Conjugated Fatty Esters. J Am Oil Chem Soc 45, 388–392 (1968). https://doi.org/10.1007/BF02667117
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02667117