Abstract
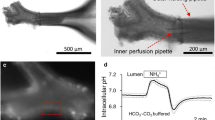
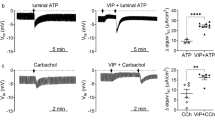
The importance of impaired HCO3 secretion in the pathophysiology of the pancreas of cystic fibrosis (CF) patients has been well documented and known for many years (1). Studies in 1990s from the Welsh (2) and Boucher (3) laboratories suggested that HCO3 secretion may also be impaired in the airways of CF patients. Perhaps because Cl- secretion has been assumed to be of higher importance and thus received the greater attention, the transcellular mechanisms of HCO3 secretion remain poorly understood and underinvestigated. Studies from our own laboratory (4) and the laboratories of Wine and Widdicombe (5) have now established that the human airway serous cell line, Calu-3 cells, secrete HCO3 and not Cl-, in response to a cAMP-mediated secretory agonist. Serous cells are the most abundant cell type of the submucosal glands (6) and are the predominate site of CFTR expression in the airways (7,8). Thus, a better understanding of the transport mechanisms of serous cells is of critical importance in establishing how mutations in CFTR lead to submucosal gland and airways pathophysiology. The focus of this chapter is to provide a description of the methods used to study Calu-3 cell monolayers grown on permeable supports by the short-circuit current (ISC) technique. In addition, methods for ion flux studies and ion substitution studies of short-circuited monolayers are described along with background information necessary to interpret the experimental results specifically as they relate to the net secretion of HCO3 . To introduce the subject of HCO3 secretion, we provide a brief description of the mechanisms of transepithelial HCO3 secretion as they are presently understood.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Quinton, P. M. (1999) Physiological basis of cystic fibrosis: a historical perspective. Physiol. Rev. 79, S3–S22.
Smith, J. J. and Welsh, M. J. (1992) cAMP stimulates bicarbonate secretion across normal, but not cystic fibrosis airway epithelia. J. Clin. Invest. 89, 1148–1153.
Willumsen, N. J. and Boucher, R. C. (1992) Intracellular pH and its relationship to regulation of ion transport in normal and cystic fibrosis human nasal epithelia. J. Physiol. (Lond) 455, 247–269.
Devor, D. C., Singh, A. K., Lambert, L. C., DeLuca, A., Frizzell, R. A., and Bridges, R. J. (1999) Bicarbonate and chloride secretion in Calu-3 human airway epithelial cells. J. Gen. Physiol. 113, 743–760.
Lee, M. C., Penland, C. M., Widdicombe, J. H., and Wine, J. J. (1998) Evidence that Calu-3 human airway cells secrete bicarbonate. Am. J. Physiol. 274, L450–L453.
Basbaum, C. B., Jany, B., and Finkbeiner, W. E. (1990) The serous cell. Annu. Rev. Physiol. 52, 97–113.
Puchelle, E., Gaillard, D., Ploton, D., Hinnrasky, J., Fuchey, C., Boutterin, M. C., Jacquot, J., Dreyer, D., Pavirani, A., and Dalemans, W. (1992) Differential localization of the cystic fibrosis transmembrane conductance regulator in normal and cystic fibrosis airway epithelium. Am. J. Respir. Cell Mol. Biol. 7, 485–491.
Engelhardt, J. F., Zepeda, M., Cohn, J. A., Yankaskas, J. R., and Wilson, J. M (1994) Expression of the cystic fibrosis gene in adult human lung. J. Clin. Invest. 93, 737–749.
Stetson, D. L., Beauwens, R., Palmisano, J., Mitchell, P. P., and Steinmetz, P. R. (1985) A double-membrane model for urinary bicarbonate secretion. Am. J. Physiol. 249, F546–F552.
Novak, I. and Greger, R. (1988). Properties of the luminal membrane of isolated perfused rat pancreatic ducts. Pfluegers Arch Eur. J. Physiol. 411, 546–553.
Ishiguro, H., Steward, M. C., Wilson, R. W., and Case, R. M. (1996) Bicarbonate secretion in interlobular ducts from guinea-pig pancreas. J. Physiol. (Lond) 495, 179–191.
Ishiguro, H., Steward, M. C., Lindsay, A. R., and Case, R. M. (1996) Accumulation of intracellular HCO3-by Na(+)-HCO3-cotransport in interlobular ducts from guinea-pig pancreas. J. Physiol. (Lond) 495, 169–178.
Abuladze, N., Lee, I., Newman, D., Hwang, J., Boorer, K., Pushkin, A., and Kurtz, I. (1998) Molecular cloning, chromosomal localization, tissue distribution, and functional expression of the human pancreatic sodium bicarbonate cotransporter. J. Biol. Chem. 273, 17,689–17,695.
Peters, K. W., Gangopadhyay, N. N., Devor, D. C., Watkins, S. C., Frizzell, R. A.,and Bridges, R. J. (1999). Ped. Pulmonol. S19, 193.
Marino, C. R., Jeanes, V., Boron, W. F., and Schmitt, B. M. (1999) Expression and distribution of the Na(+)-HCO(-)(3) cotransporter in human pancreas. Am. J. Physiol. 277, G487–G494.
Romero, M. F., Hediger, M. A., Boulpaep, E. L., and Boron, W. F. (1997) Expression cloning and characterization of a renal electrogenic Na+/HCO3-cotransporter. Nature 387, 409–413.
Gray, M. A., Pollard, C. E., Harris, A., Coleman, L., Greenwell, J. R., and Argent, B. E. (1990) Anion selectivity and block of the small-conductance chloride channel on pancreatic duct cells. Am. J. Physiol. 259, C752–C761.
Linsdell, P., Tabcharani, J. A., and Hanrahan, J. W. (1997) Multi-Ion mechanism for ion permeation and block in the cystic fibrosis transmembrane conductance regulator chloride channel. J. Gen. Physiol. 110, 365–377.
Illek, B., Tam, A. W., Fischer, H., and Machen, T. E. (1999). Anion selectivity of apical membrane conductance of Calu 3 human airway epithelium. Pflugers Arch. 437, 812–822.
Hug, M., Frizzell, R. A., and Bridges, R. J. (2000) Forskolin-stimulated Calu-3 cells have a very high apical membrane conductance with important implications for the mechanisms of anion secretion. Pediatric Pulmonology S20, 209.
Wills, N. K., Reuss, L., and Lewis, S. A. (eds.) (1996) Epithelial Transport. Chapman & Hall, London, 1996.
Ussing, H. H. and Zerahn, K. (1999) Active transport of sodium as the source of electric current in the short-circuited isolated frog skin. Reprinted from Acta Physiol. Scand. 23, 110-127. J. Am. Soc. Nephrol. 20, 2056–2065.
Guba, M., Kuhn, M., Forssmann, W. G., Classen, M., Gregor, M., and Seidler, U. (1996) Guanylin strongly stimulates rat duodenal HCO3-secretion: proposed mechanism and comparison with other secretagogues. Gastroenterology 111, 1558–1568.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Humana Press Inc.
About this protocol
Cite this protocol
Bridges, R.J. (2002). Transepithelial Measurements of Bicarbonate Secretion in Calu-3 Cells. In: Skach, W.R. (eds) Cystic Fibrosis Methods and Protocols. Methods in Molecular Medicine™, vol 70. Humana Press. https://doi.org/10.1385/1-59259-187-6:111
Download citation
DOI: https://doi.org/10.1385/1-59259-187-6:111
Publisher Name: Humana Press
Print ISBN: 978-0-89603-897-4
Online ISBN: 978-1-59259-187-9
eBook Packages: Springer Protocols