Abstract
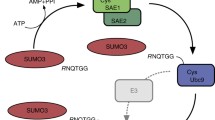
In humans cells three SUMO paralogues (SUMO-1, SUMO-2 and SUMO-3) and six SUMO specific proteases (SENP1-SENP3 and SENP5-SENP7) are expressed. Together the SUMO proteases perform three distinct functions. They: (1) process the immature pro-SUMO proteins into the active forms, (2) remove SUMO molecules conjugated to protein targets, and (3) depolymerise SUMO conjugated within polymeric chains. By regulating these processes the SENPs play a crucial role in regulating the sumoylation state of target proteins in cells, and therefore are academically and pharmacologically interesting enzymes. Gel-based techniques for SENP analysis are well established and can be used for many applications, but their laborious methodology makes them cumbersome tools for kinetic analysis or inhibitor screening. Therefore in vitro FRET-based assays have been developed to test the three major functions of the SENPs. These use fluorescent protein fusions of the SUMOs, and together facilitate high-throughput, real-time analysis of the three major SUMO protease activities.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Seeler, J. S. and Dejean, A. (2003) Nuclear and unclear functions of SUMO. Nat. Rev. Mol. Cell. Biol. 4, 690–699.
Saitoh, H. and Hinchey, J. (2000) Functional heterogeneity of small ubiquitin-related protein modifiers SUMO-1 versus SUMO-2/3. J. Biol. Chem. 275, 6252–6258.
Tatham, M. H., Jaffray, E., Vaughan, O. A., Desterro, J. M., Botting, C. H., Nai-smith, J. H. and Hay, R. T. (2001) Polymeric chains of SUMO-2 and SUMO-3 are conjugated to protein substrates by SAE1/SAE2 and Ubc9. J. Biol. Chem. 276, 35368–35374.
Ayaydin, F. and Dasso, M. (2004) Distinct in vivo dynamics of vertebrate SUMO para-logues. Mol. Biol. Cell 15, 5208–5218.
Hay, R. T. (2005) SUMO: a history of modification. Mol. Cell 18, 1–12.
Hay, R. T. (2007) SUMO-specific proteases: a twist in the tail. Trends Cell Biol. 17, 370– 376.
Li, S. J. and Hochstrasser, M. (1999) A new protease required for cell-cycle progression in yeast. Nature 398, 246–251.
Takahashi, Y., Mizoi, J., Toh, E. A. and Kikuchi, Y. (2000) Yeast Ulp1, an Smt3-Specific Protease, Associates with Nucleop-orins. J. Biochem. (Tokyo) 128, 723–725.
Li, S. J. and Hochstrasser, M. (2000) The yeast ULP2 (SMT4) gene encodes a novel protease specific for the ubiquitin-like Smt3 protein. Mol. Cell. Biol. 20, 2367–2377.
Bylebyl, G. R., Belichenko, I. and Johnson, E. S. (2003) The SUMO isopeptidase Ulp2 prevents accumulation of SUMO chains in yeast. J. Biol. Chem. 278, 44113–44120.
Melchior, F., Schergaut, M. and Pichler, A. (2003) SUMO: ligases, isopeptidases and nuclear pores. Trends. Biochem. Sci. 28, 612–618.
Reverter, D. and Lima, C. D. (2004) A basis for SUMO protease specificity pro- vided by analysis of human Senp2 and a Senp2-SUMO complex. Structure (Camb) 12, 1519–1531.
Shen, L. N., Dong, C., Liu, H., Naismith, J. H. and Hay, R. T. (2006) The structure of SENP1-SUMO-2 complex suggests a structural basis for discrimination between SUMO paralogues during processing. Bio-chem. J. 397, 279–288.
Arnold, J. J., Bernal, A., Uche, U., Sterner, D. E., Butt, T. R., Cameron, C. E. and Mattern, M. R. (2006) Small ubiquitin-like modifying protein isopeptidase assay based on poliovirus RNA polymerase activity. Anal. Biochem. 350, 214–221.
Bossis, G., Chmielarska, K., Gartner, U., Pichler, A., Stieger, E. and Melchior, F. (2005) A fluorescence resonance energy transfer-based assay to study SUMO modification in solution. Methods Enzymol. 398, 20–32.
Martin, S. F., Hattersley, N., Samuel, I. D., Hay, R. T. and Tatham, M. H. (2007) A fluorescence-resonance-energy-transfer-based protease activity assay and its use to monitor paralog-specific small ubiquitin-like modifier processing. Anal. Biochem. 363, 83–90.
Mikolajczyk, J., Drag, M., Bekes, M., Cao, J. T., Ronai, Z. and Salvesen, G. S. (2007) Small ubiquitin-related modifier (SUMO)-specific proteases: profiling the specificities and activities of human SENPs. J. Biol. Chem. 282, 26217–26224.
Wilkinson, K. D., Gan-Erdene, T. and Komlli, N. (2005) Derivatization of the C-Terminus of Ubiquitin and Ubiquitin-like Proteins Using Intein Chemistry: Methods and Uses. Methods Enzymol. 399, 37–51.
Shen, L., Tatham, M. H., Dong, C., Zagorska, A., Naismith, J. H. and Hay, R. T. (2006) SUMO protease SENP1 induces isomerization of the scissile peptide bond. Nat. Struct. Mol. Biol. 13, 1069–1077.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Humana Press, a part of Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Tatham, M.H., Hay, R.T. (2009). FRET-Based In Vitro Assays for the Analysis of SUMO Protease Activities. In: Ulrich, H.D. (eds) SUMO Protocols. METHODS IN MOLECULAR BIOLOGY™, vol 497. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-59745-566-4_17
Download citation
DOI: https://doi.org/10.1007/978-1-59745-566-4_17
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-934115-80-0
Online ISBN: 978-1-59745-566-4
eBook Packages: Springer Protocols