Abstract
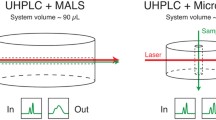
We present a review of high-throughput techniques for the characterization and quality control of proteins in the course of purification, evaluation, and formulation, based on static and dynamic light scattering. Multi-angle static light scattering (MALS) in combination with rapid, low-volume UHPLC size exclusion chromatography is effective in characterizing key biophysical properties, while dynamic light scattering (DLS) in high-throughput microwell-plate format provides large quantities of data in a short time to screen many conditions, excipients, cell lines, or candidate biotherapeutics.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Liu T, Chu B (2015) Light-scattering by proteins. In: Somasundaran P (ed) Encyclopedia of surface and colloid science. CRC, Boca Raton
Folta-Stogniew EJ (2009) Macromolecular interactions: light scattering. In: Encyclopedia of life sciences. Wiley, Hoboken
PJ W (1993) Light scattering and the absolute characterization of macromolecules. Anal Chim Acta 272:1–40
Zhao H, Brown PH, Schuck P (2011) On the distribution of protein refractive index increments. Biophys J 100(9):2309–2317
Some D, Kenrick S (2012) Characterization of protein-protein interactions via static and dynamic light scattering. In: Cai J (ed) Protein interactions. In Tech, Rijeka
Wen J, Arakawa T, Philo JS (1996) Size-exclusion chromatography with on-line light-scattering, absorbance, and refractive index detectors for studying proteins and their interactions. Anal Biochem 240(2):155–166
Gimpl K, Klement J, Keller S (2016) Characterising protein/detergent complexes by triple-detection size-exclusion chromatography. Biol Proced Online 18(1):4
Rebolj K, Pahovnik D, Žagar E (2012) Characterization of a protein conjugate using an asymmetrical-flow field-flow fractionation and a size-exclusion chromatography with multi-detection system. Anal Chem 84(17):7374–7383
Bouvier ESP, Koza SM (2014) Advances in size-exclusion separations of proteins and polymers by UHPLC. TrAC Trends Anal Chem 63:85–94
Philo JS (2009) A critical review of methods for size characterization of non-particulate protein aggregates. Curr Pharm Biotechnol 10(4):359–372
He F et al (2010) High-throughput dynamic light scattering method for measuring viscosity of concentrated protein solutions. Anal Biochem 399(1):141–143
Menzen T, Friess W (2014) Temperature-ramped studies on the aggregation, unfolding, and interaction of a therapeutic monoclonal antibody. J Pharm Sci 103(2):445–455
Lehermayr C et al (2011) Assessment of net charge and protein–protein interactions of different monoclonal antibodies. J Pharm Sci 100(7):2551–2562
Kenrick S, Some D (2018) The diffusion interaction parameter kd as an indicator of colloidal and thermal stability. Available from http://www.wyatt.com/files/literature/app-notes/dls-plate/WP5004-diffusion-interaction-parameter-for-colloidal-and-thermal-stability.pdf
He F et al (2011) High-throughput assessment of thermal and colloidal stability parameters for monoclonal antibody formulations. J Pharm Sci 100(12):5126–5141
Austerberry JI et al (2017) The effect of charge mutations on the stability and aggregation of a human single chain Fv fragment. Eur J Pharm Biopharm 115:18–30
Connolly BD et al (2012) Weak interactions govern the viscosity of concentrated antibody solutions: high-throughput analysis using the diffusion interaction parameter. Biophys J 103(1):69–78
Dear BJ et al (2017) Contrasting the influence of cationic amino acids on the viscosity and stability of a highly concentrated monoclonal antibody. Pharm Res 34(1):193–207
He F et al (2011) Screening of monoclonal antibody formulations based on high-throughput thermostability and viscosity measurements: design of experiment and statistical analysis. J Pharm Sci 100(4):1330–1340
Luo H et al (2017) Liquid-liquid phase separation causes high turbidity and pressure during low pH elution process in Protein A chromatography. J Chromatogr A 1488:57–67
Some D (2014) Protein quality control in SPR and BLI high-throughput screening studies. Available from http://www.wyatt.com/files/literature/white-papers/protein-quality-control-high-throughput-screening-studies.pdf
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Some, D., Razinkov, V. (2019). High-Throughput Analytical Light Scattering for Protein Quality Control and Characterization. In: Vincentelli, R. (eds) High-Throughput Protein Production and Purification. Methods in Molecular Biology, vol 2025. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9624-7_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9624-7_16
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9623-0
Online ISBN: 978-1-4939-9624-7
eBook Packages: Springer Protocols