Abstract
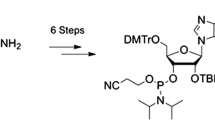
Pyrrolidinyl PNA with an α-/β-dipeptide backbone consisting of alternating nucleobase-modified d-proline and (1S,2S)-2-aminocyclopentanecarboxylic acid (also known as acpcPNA) is a class of conformationally constrained PNA that shows exceptional DNA hybridization properties including very high specificity and the inability to form self-pairing hybrids. In this chapter, details of the syntheses of acpcPNA as well as its monomers and a protocol for site-specific labeling with a fluorescent dye via click chemistry are reported.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kumar VA, Ganesh KN (2005) Conformationally constrained PNA analogues: structural evolution toward DNA/RNA binding selectivity. Acc Chem Res 38:404–412
Pokorski JK, Witschi MA, Purnell BL, Appella DH (2004) (S,S)-trans-Cyclopentane-constrained peptide nucleic acids. A general backbone modification that improves binding affinity and sequence specificity. J Am Chem Soc 126:15067–15073
Govindaraju T, Kumar VA, Ganesh KN (2005) (SR/RS)-Cyclohexanyl PNAs: conformationally preorganized PNA analogues with unprecedented preference for duplex formation with RNA. J Am Chem Soc 127:4144–4145
Dragulescu-Andrasi A, Rapireddy S, Frezza BM, Gayathri C, Gil RR, Ly DH (2006) A simple γ-backbone modification preorganizes peptide nucleic acid into a helical structure. J Am Chem Soc 128:10258–10267
Vilaivan T (2015) Pyrrolidinyl PNA with α/β-dipeptide backbone: from development to applications. Acc Chem Res 48:1645–1656
Vilaivan T, Srisuwannaket C (2006) Hybridization of pyrrolidinyl peptide nucleic acids and DNA: selectivity, base-pairing specificity, and direction of binding. Org Lett 8:1897–1900
Vilaivan C, Srisuwannaket C, Ananthanawat C, Suparpprom C, Kawakami J, Yamaguchi Y, Tanaka Y, Vilaivan T (2011) Pyrrolidinyl peptide nucleic acid with α/β-peptide backbone: a conformationally constrained PNA with unusual hybridization properties. Artificial DNA PNA XNA 2:50–59
Lohse J, Dahl O, Nielsen PE (1999) Double duplex invasion by peptide nucleic acid: a general principle for sequence-specific targeting of double-stranded DNA. Proc Natl Acad Sci U S A 96:11804–11808
Bohländer PR, Vilaivan T, Wagenknecht H-A (2015) Strand displacement and duplex invasion into double-stranded DNA by pyrrolidinyl peptide nucleic acids. Org Biomol Chem 13:9223–9230
Ditmangklo B, Boonlua C, Suparpprom C, Vilaivan T (2013) Reductive alkylation and sequential reductive alkylation-click chemistry for on-solid-support modification of pyrrolidinyl peptide nucleic acid. Bioconjug Chem 24:614–625
Vilaivan T (2018) Fluoreogenic PNA probes. Beilstein J Org Chem 14:253–281
Hövelmann F, Seitz O (2016) DNA stains as surrogate nucleobases in fluorogenic hybridization probes. Acc Chem Res 49:714–723
Reenabthue N, Boonlua C, Vilaivan C, Vilaivan T, Suparpprom C (2011) 3-Aminopyrrolidine-4-carboxylic acid as versatile handle for internal labeling of pyrrolidinyl PNA. Bioorg Med Chem Lett 21:6465–6469
Lowe G, Vilaivan T (1997) Amino acids bearing nucleobases for the synthesis of novel peptide nucleic acids. J Chem Soc Perkin Trans 1 1997:539–546
Lowe G, Vilaivan T (1997) Dipeptides bearing nucleobases for the synthesis of novel peptide nucleic acids. J Chem Soc Perkin Trans 1 1997:547–554
Cruickshank KA, Jiricny J, Reese CB (1984) The benzoylation of uracil and thymine. Tetrahedron 25:681–684
LePlae PR, Umezawa N, Lee H-S, Gellman SH (2001) An efficient route to either enantiomer of trans-2-aminocyclopentanecarboxylic acid. J Org Chem 66:5629–5632
Lee HS, LePlae PR, Porter EA, Gellman SH (2001) An efficient route to either enantiomer of orthogonally protected trans-3-aminopyrrolidine-4-carboxylic acid. J Org Chem 66:3597–3599
Blake J, Willson CD, Rapoport H (1964) 3-Pyrrolidinones by intramolecular condensation. J Am Chem Soc 86:5293–5299
Yotapan N, Charoenpakdee C, Wathanathavorn P, Ditmangklo B, Wagenknecht H-A, Vilaivan T (2014) Synthesis and optical properties of pyrrolidinyl peptide nucleic acid carrying a clicked Nile red label. Beilstein J Org Chem 10:2166–2174
Ditmangklo B, Taechalertpaisarn J, Siriwong K, Vilaivan T (2019) Clickable styryl dyes for fluorescence labeling of pyrrolidinyl PNA probes for the detection of base mutations in DNA. Org Biomol Chem 17:9712–9725
Bates RW, Dewey MR (2009) A formal synthesis of swainsonine by gold-catalyzed allene cyclization. Org Lett 11:3706–3708
Robinson DS, Greenstein JP (1952) Sterioisomers of hydroxyproline. J Biol Chem 195:383–388
Peterson ML, Vince R (1991) Synthesis and biological evaluation of 4-purinylpyrrolidine nucleosides. J Med Chem 34:787–2797
Lee K-H, Ko K-Y (2006) Catalytic oxidation of benzophenone hydrazone with alumina-supported KMnO4 under oxygen atmosphere. Bull Kor Chem Soc 27:185–186
Miller JB (1959) Preparation of crystalline diphenyldiazomethane. J Org Chem 24:560–561
Obkircher M, Stähelin C, Dick F (2008) Formation of Fmoc-β-alanine during Fmoc-protections with Fmoc-OSu. J Pept Sci 14:763–766
Egholm M, Nielsen PE, Buchardt O, Berg RH (1992) Recognition of guanine and adenine in DNA by cytosine and thymine containing peptide nucleic acids (PNA). J Am Chem Soc 114:9677–9678
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Ditmangklo, B., Muangkaew, P., Supabowornsathit, K., Vilaivan, T. (2020). Synthesis of Pyrrolidinyl PNA and Its Site-Specific Labeling at Internal Positions by Click Chemistry. In: Nielsen, P. (eds) Peptide Nucleic Acids. Methods in Molecular Biology, vol 2105. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0243-0_3
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0243-0_3
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0242-3
Online ISBN: 978-1-0716-0243-0
eBook Packages: Springer Protocols