Abstract
Background
Behçet's disease (BD) is a chronic, multi-systemic, recurrent condition that affects the vascular, ocular, mucocutaneous, and central nervous systems. The diagnosis of this disease depends on its clinical features, which are similar to those observed in several diseases, such as Parkinson’s disease, pemphigus vulgaris, systemic lupus erythematosus, Crohn ҆s disease, and Sjӧgren’s syndrome. Lysosome-mediated autophagy is a catabolic, cytoprotective mechanism that maintains cell homeostasis by degrading undesired long-lived proteins and recycling nutrients. The aim of this study was to evaluate the correlations between some autophagy-related genes (ATG5, ATG7, ATG12, LC3b, mTOR) and the pathogenesis and immunopathology of BD. The expression levels of the genes were evaluated by quantitative polymerase chain reaction (qPCR) in 101 individuals that are classified into two groups. Group 1: (n = 71) BD patients, Group 2: (n = 30) healthy controls.
Results
Patients with BD had lower mRNA expression levels of ATG5 and mTOR and higher levels of LC3b mRNA than the controls. No significant differences in the levels of both ATG7 and ATG12 were observed between the two groups. According to the area under the curve analysis, LC3b was considered the best candidate biomarker among the selected markers for the diagnosis of BD. The mRNA expression of ATG5 was significantly correlated with patient age and the presence of oral ulcers. The mRNA expression of ATG7 was significantly associated with age and the presence of erythema nodosum and vascular lesions, whereas that of LC3b was significantly correlated with the presence of pustules.
Conclusion
These findings indicated that elevated levels of LC3b were strongly associated with BD. Likewise, the levels of ATG5 and ATG7 were associated with the complications and outcomes of this disease. Additional assessments of the mRNA expression levels of these autophagy-related genes might prove beneficial in diagnosing this autoimmune disorder.
Similar content being viewed by others
Introduction
Behçet’s disease (BD, #OMIM 109650) is an uncommon systemic disease of unknown etiology, primarily defined by recurring oral ulcer attacks (60%–90%) that occur at least three times in 12 months. This symptom is considered as the earliest manifestation of the disease. Additionally, genital ulcers (48%–85%), ocular lesions (45%–90%), neurological manifestations (2.3%–38.5%), and other systemic manifestations may develop over several years [1]. The 1994 International Chapel Hill Consensus Conference, which was updated in 2012, classified BD as variable vessel vasculitis (VVV) because the vasculitis associated with this disease does not involve any predominant vessel; this disease can affect any size or type (arteries, veins, and capillaries) of the blood vessel [2]. BD often leads to serious systemic problems, such as blindness, and the main causes of mortality are associated with neurological and cardiovascular complications [3]. The prevalence of BD differs worldwide, with an estimated ratio of 3.6 per 100,000 in the Egyptian population [4]. Moreover, organs affected BD is made more difficult by occurring of vessels thrombosis of any type. In male patients with active disease thrombosis is prevalent and is a major root of death and morbidity. Hyperactivation of neutrophils has been demonstrated in patients with BD, possibly due to the presence of the chromosome 6 human leukocyte antigen (HLA)-B51 allele [5].
Mucocutaneous involvement is prevalent in BD. In the current study, four patients had lost sight. The prevalence of oral ulcers in BD is estimated to range between 97 and 100%. Oral ulcers make it difficult to chew, speak, and drink, thereby decreasing the patient's quality of life [6]. Approximately 90% of the patients in the present study developed oral ulcers. As reported by an earlier study, the oral ulcers are considered as the majority of BD symptoms (98.2%), followed by genital ulcers (62.4%), ocular involvements (53.2%), erythema nodosum (53.2%), acniform lesions (51.8%), arthritis (38.6%), gastrointestinal symptoms (25.1%), neurogenic disorders (9.0%) and vascular involvements (8.1%). In addition, it has been previously observed how HLA and disease symptoms correlate (41.5% with HLA-B*51 and 24.1% with HLA-A*26) [7]. Acneiform lesions are the most common type of skin lesions that occur primarily on the trunk and extremities of patients with BD. Lesions found on other parts of the body other than the face were considered to be more specific for BD [8]. Arthritis and genital ulcers are more common in females, whereas ocular manifestations, acneiform lesions, and HLA-B*51 are more common in males [7].
Various genetic and environmental factors are involved in the initiation and development of this disease, but the most robustly associated risk factor is the HLA-B51 allele [9]. Four main supporting criteria for evidence genetic influence on BD susceptibility include instances of familial aggregation, unusual geographical distribution, association with HLA class I antigens (HLA-B51) and gene polymorphisms that affect immunological response [9]. Several genetic studies have shown that HLA-B is significantly correlated with cytokine production [10].
Several clinical criteria are used to diagnose BD owing to the lack of a specific diagnostic test [11]. However, it is difficult to reach a diagnosis in some cases because of the time it takes for the typical symptoms to manifest. In addition to the signs and symptoms, the diagnosis of this disease is made based on the positive clinical criteria commonly known as the “International Clinical Criteria for Behçet’s Disease.” Recurrent mouth sores are generally necessary for a diagnosis of BD and are the ideal benchmark for clinical diagnosis. Moreover, recurring at least two of additional signs as eye inflammation, skin and genital sores confirm the diagnosis of BD [12]. The pathergy test is considered as one of the major criteria, according to the “International Study Group for Behçet’s Disease” [13]. Blobner first identified the pathergy reaction, which appears as an erythematous papule or pustule at the injection site 24–48 h after an intradermal puncture, in 1937 [14]. The pathergy test reveals skin hyperreactivity and is used as an additional test in BD diagnosis. The pathergy test is currently not standardized, and the most popular procedure is the intradermal insertion of a 20–22 gauge needle at a 45-degree angle into at least two separate places on the forearm's avascular area [15]. A positive result of this test suggests that a minor injury is causing the immune system to overreact. Hence, the pathergy reaction test is not specific because it can give positive results with other conditions. Moreover, a positive pathergy phenomenon is only seen in small percentage of patients with BD. Therefore, there is an urgent need to search for efficient and specific diagnostic tests for this disease [16, 17].
BD has long been considered as an autoimmune disorder with unknown clear causes, but it shares many clinical symptoms with other autoinflammatory diseases. BD aetiology has traditionally been attributed to immune system disorders. Both adaptive and innate immune cells are implicated in disease pathogenesis [18]. The loss of normal immunological control, along with the activation and migration of neutrophils to the inflammatory lesion site, is thought to play a major role in the pathogenesis of BD. Patients with BD show hallmark depletion in the natural killer (NK) cells in the peripheral blood, a shift that corresponded to the presence of the disease, accompanied by a reduction in the cytokine IL-10 and regulatory T cells (Tregs). Conversely, over production of tumor necrosis factor (TNF)-α, interferon (IFN)-γ, interleukin (IL)-8 and IL-17 level that correlated with BD activity [19].
Autophagy (autophagocytosis) is a natural, intracellular regulated mechanism characterized by the removal of aggregated proteins, infectious organisms, and damaged organelles via lysosomes, and hence plays a multitude of physiological and pathological functions [20]. Macroautophagy, microautophagy, and chaperon-mediated autophagy are the three most prevalent types of autophagy [21]. Autophagy pathways and their mediators have a crucial function in the immune system and inflammatory process; they may help prevent autoimmune disorders and inflammatory conditions. Hence, autophagy dysfunction can contribute to the pathogenesis of several autoimmune diseases [22].
Autophagy is carried out by autophagy-related genes (ATG). Genetic screening performed on the budding yeast Saccharomyces cerevisiae led to the identification of the first genes involved in autophagy [23]. Numerous genes, such as the immune-related GTPase family M protein (IRGM), ATG2A, ATG5, ATG7, ATG12, ATG16 like 1 (ATG16L1), leucine-rich repeat kinase 2), death-associated protein [24], mechanistic target of rapamycin (mTOR), regulatory associated protein of mTOR (RAPTOR), rapamycin insensitive companion of mTOR (RICTOR), and microtubule-associated protein light chain 3b (LC3b), have been examined in BD [10]. Nonetheless, there is a lack of research concerning these genes in Egyptian patients with BD.
Atg5 is a crucial protein found in the phagophoric membrane of the autophagic vesicle [25]. It is activated by Atg7 to form a complex with Atg12 and Atg16L1. Atg7 is required for the formation and expansion of autophagosomes [26, 27]. Atg12 is activated by Atg7, following which it conjugates with Atg5; this Atg12-Atg5 conjugate forms a complex with Atg16L [28]. The LC3-1 C-terminus conjugates with the Atg12-Atg5-Atg16L complex and is associated with the phagophore membranes to form LC3-II. The Atg12-Atg5-Atg16L complex separates from the autophagosome following the development of the autophagosome [29]. LC3-B was thought to be involved in the regulation of the assembly and disassembly of microtubules [30]. LC3 is a key protein in the autophagy pathway and is involved in the selection of the autophagy substrate and biogenesis of the autophagosome. LC3 is the most widely used marker of autophagosomes [31]. mTOR, also known as the mechanistic target of rapamycin and FK506-binding protein 12-rapamycin-associated protein 1 (FRAP1), is a kinase encoded by the mTOR gene in humans [32]. mTOR forms connections with other proteins and is a key element of the two different protein complexes, mTOR complex 1 (mTORC1), and mTOR complex 2 (mTORC2), which control many biological activities [33]. Active mTORC1 is located on lysosomes, and mTOR inhibition occurs by damaging the lysosomal membrane through various exogenous or endogenous agents [34]. However, mTOR suppression during the damage of lysosomes promotes autophagy's reaction, resulting in lysophagy, which eliminates the destroyed lysosomes [35].
A study by Adeeb et al. [36] identified some probable diagnostic biomarkers, such as the HLA-B*51, chemokines CCR1 and CCR3, cytokines TNF, ILs, and IFNs, T lymphocytes, inflammasomes, autoantibodies, and fecal calprotectin. In addition, hypovitaminosis D has been linked to various inflammatory diseases, including BD. Vitamin D has immunosuppressive effects and is recommended as a therapeutic tool for many autoimmune disorders [37]. According to Do et al. [38], vitamin D reduces the expression of toll-like receptor (TLR) in the monocytes of BD patients; additionally, TLR-2 and TLR-4, which are crucial for the etiology of BD, are responsible for inducing inflammation. Thus, vitamin D may be useful as a therapy option for patients with this type of inflammation. However, despite the appearance of these candidate markers, there is currently insufficient widespread data to justify their adoption and incorporation into the most recent classification standards [38].
The present study was conducted to screen a panel of differentially expressed autophagy-related genes (ATG5, ATG7, ATG12, LC3b, mTOR) that have been postulated to play a role in the immunopathology and pathogenesis of BD. Additionally, correlations between the clinical heterogeneity and prognosis of the disease and the selected specific autophagy markers were determined.
Subjects and methods
Patients
This prospective study comprised 101 subjects (71 BD patients and 30 healthy controls) recruited from the Internal Medicine inpatient ward and Rheumatology and Clinical Immunology outpatient clinics at the Internal Medicine Department, Kasr Elainy, School of Medicine, Cairo University Hospitals. Written informed consents were signed from all the patients, family members or legal guardians, as appropriate according to the organization of ethical guidelines. The aim, methods and duration of the study were fully explained to all participants. The anonymity and confidentiality of the patient's data were ensured by coding the datasheets. Informed consent for the use of the detailed medical history, demographic, and medication profile information was obtained from the subjects.
The study was approved by the Institutional Bioethics Committee of the National Research Center (Registration number: 19047) and conducted in accordance with the ethical guidelines of the Declaration of Helsinki [39].
BD was diagnosed by an expert rheumatologist based on the “International Clinical Criteria for Behçet’s Disease.” Exclusion criteria: (1) BD mimics diseases e.g.: vasculitis, (2) patients with other autoimmune diseases, (3) diabetic patients, (4) patients with rheumatic diseases, (5) patients with current infections such as HCV, (6) malignancies and (7) endocrine diseases were also excluded.
Drugs and treatment protocol
Demographic and clinical information were collected from the records. The currently used medications such as disease-modifying anti-rheumatic drugs (DMARDs), Colchicine, Azathioprine, Prednisolone, Rituximab and Etanercept and others such as anticoagulants (Warfarin or Marivan), Cyclophosphamide (Endoxan) were also recorded.
Clinical investigation and diagnosis
Detailed information about the medical history and medication profile was obtained. Ophthalmological fundus and slit lamp examinations were performed for those presenting with ocular manifestations. All the participants were subjected to laboratory assessments, including hemoglobin (Hb; mmol/l), white blood cell count (WBC; 109/l), platelet count (plt; 10.9/l), erythrocyte sedimentation rate (ESR; mm/s), and immunological profiles for rheumatoid factor (RF), C-reactive protein (CRP), and antinuclear antibody (ANA). The relative expression levels of the autophagy markers were determined by quantitative polymerase chain reaction (qPCR)
Relative expression of autophagy markers by quantitative polymerase chain reaction (qPCR)
Five milliliters of blood were collected in EDTA tubes by vein puncture, and the total RNA was extracted using a QIAamp RNA blood Mini Kit (Cat. No. 52304; Qiagen, USA) [40]. The total RNA was reverse transcribed into first-strand complementary DNA (cDNA) using a High-Capacity cDNA Reverse Transcription Kit (Cat. No. 4368813, Applied Biosystems, USA) [41]. The relative mRNA expression levels of ATG5, ATG7, ATG12, LC3b, and mTOR were measured by qPCR using the Roche real-time PCR system (light cycler 480, product no. 05015278001; Roche life science, UK) and the SYBR Green master mix (Cat. No. K0252; Thermo Scientific, USA) [42].
Sequence of primers for genes is illustrated in Table 1. The values were normalized based on the expression level of the endogenous housekeeping gene (β-actin), which was used as a denominator. The relative changes in gene expression between patient and healthy control groups were determined using the light cycler 480 real-time PCR system software 1.5.0 SP3. The thermal cycling was performed using a three-step cycling protocol: Initial step (95 ºC, 10 min for 1 cycle), denaturation step (95 ºC, 15 s for 40 cycles), annealing step (59 ºC, 30 s for 40 cycles), and finally extension step (72 ºC, 30 s for 40 cycles).
Statistical analysis
The results were statistically analyzed using the Minitab 17.1.0.0 software for windows (Minitab Inc., 2013, Pennsylvania, USA). Continuous data were presented as mean and standard deviation, whereas categorical data were described as frequency counts and percentages. The normality of the data was examined using the Shapiro–Wilk test. Comparisons between two continuous groups were performed using the independent t-test or Mann–Whitney test, and those between two or more categorical groups were analyzed using the Chi-square test.
The accuracy of the mRNA expression of ATG5, ATG7, ATG12, mTOR, and LC3b was assessed using the receiver operating characteristics (ROC) curve analysis, assuming that the null hypothesis of the area under ROC (AUROC) was 0.5 (power, 80%; confidence interval [CI], 95%, and BD prevalence, 1%) [43].
A sample size calculated with minimum total number of 90 using MedCalc (MedCalc Software v.13, Ostend, Belgium) software for windows. The quality of calculated AUROC was as follows: (0.90–1 = excellent), (0.80–0.90 = good), (0.70–0.80 = fair), (0.60–0.70 = poor) and (0.50–0.60 = fail). General linear model (GLM) used to estimate the association between clinical features of patients and relative mRNA expression of examined genes. All tests were two sided, p considered significant if < 0.05.
Results
Patient demographics
A total of 101 subjects aged (10–65) years were included in this study. All 71 patients consented for detailed medical history, demographic, and medication profile examination. A summary of the clinical characteristics of all participants are represented in Table 2. The 71 patients presented with a mean Hb of 7.94 ± 1.3, WBC of 7.1 ± 2.99, platelet count of 296.7 ± 237.8, and ESR of 5.9*10−4 ± 5.2*10−4. Furthermore, 31 (43.66%) patients were positive for CRP. The immunological profiles for RF and ANA showed negative results in all 71 patients.
The mRNA expression levels of the autophagy-related genes
To examine the autophagy-related genes expression in patients with BD, we used qPCR technique to investigate the levels of mRNA expression of ATG5, ATG7, ATG12, LC3b and mTOR genes as shown in Table 3 and Fig. 1. The expression levels of ATG5 and mTOR were significantly decreased (p = 0.004 and 0.01, respectively), whereas that of LC3b was significantly upregulated (p < 0.001) in the BD patients when compared to those in the healthy controls. No significant differences in the expression levels of ATG7 and ATG12 were observed between the two groups (p = 0.24 and 0.41, respectively) Table 3.
Association between the expression levels of the autophagy-related genes and the clinical parameters of patients with BD
The clinical features of the 71 patients with BD and the association of each marker with various clinical criteria are examined and shown in Table 4. The mRNA expression level of ATG5 was significantly associated with patient age (mean = 35.3; p = 0.001) and the incidence of an oral ulcer (90.1%; p = 0.01). Likewise, the expression level of ATG7 was significantly associated with patient age (p = 0.001) and the presence of erythema nodosum (8.4%; p = 0.02) and vascular lesions (25.3%; p = 0.04). LC3b expression was significantly associated with the presence of pustules (25.4%; p = 0.03).
Association between autophagy markers and BD diagnosis
The ROC curve and cutoff values for the selected autophagy markers were calculated to identify the predictive marker for BD Table 5 and Fig. 2. The accuracies of the expression levels of ATG5 and mTOR were demonstrated by the AUC values (68% and 66%, respectively; p = 0.004 and 0.01, respectively). LC3b expression showed accuracy with an AUC of 88% (p < 0.001). A cutoff point of > 0.024 for the LC3b expression proved most useful for diagnosing BD, with a sensitivity and specificity of 80%. Likewise, a cut point of < 0.0034 showed a sensitivity of 39% and specificity of 90% for ATG5, while that of < 0.0081 showed a sensitivity of 54% and specificity of 90% for mTOR expression. Despite the increase in the specificity for the expression levels of ATG5 and mTOR, their sensitivity values were low. Alternatively, the increase in both the sensitivity and specificity for LC3b expression indicated its significant association with BD.
Discussion
BD is a persistent inflammatory multi-systemic condition that affects numerous organs and has broad clinical manifestations involving the mucocutaneous, genital, ophthalmic, vascular, and gastrointestinal systems. The symptoms are more pronounced in those with chronic inflammation and endothelial dysfunction [5]. BD is a form of chronic vasculitis that affects vessels of all sizes and is defined by the presence of recurrent oral ulcers, genital ulcers, and ophthalmic issues (particularly uveitis), thus forming the triple-symptom complex [44]. These manifestations are caused by chronic inflammation due to the infiltration of neutrophils and lymphocytes [45].
BD is equally prevalent among males and females in some geographic regions and more prevalent among males in others [46]. BD is classified as a systemic vasculitis associated with significant morbidity and mortality, particularly with an early age onset in males [47]. In addition, a study by Kural-Seyahi et al. [48] demonstrated that the disease has a more severe course and higher mortality among male patients. Moreover, Maldini et al. [49] stated that both male gender and the presence of the HLA-B51 allele are consistently associated with a severe disease course and poor prognosis in BD. Likewise, HLA-B51 was more common among men with BD [49]. Male patients are more likely to be affected at a younger age, have a more severe uveitis, present with worse visual acuity, and suffer vision loss over time [50].
BD is diagnosed based on clinical features, which are commonly encountered in other autoinflammatory diseases such as (Pemphigus vulgaris, Sjӧgren syndrome, Parkinson disease, Systemic lupus erythematosus, and Crohn’s Disease) [51]. This could lead to an inaccurate diagnosis; hence, the identification of reliable blood markers for a more accurate diagnosis is crucial.
Autophagy is a lysosome-mediated cytoprotective catabolic mechanism that upholds cellular homeostasis by the degradation and reuse of the unwanted intracellular constituents of long-lived proteins and recycling nutrients. These biological processes are performed by autophagy proteins via several pathways that affect the functions of the immune system, especially the production of B and T lymphocytes [24]. Several studies have suggested strong correlations between BD and the autophagy-related inflammatory and immune features [52–54]. The dysregulation of some autophagy-related genes has been examined in BD [10, 24].
The present study was conducted to screen a panel of differentially expressed genes related to autophagy (ATG5, ATG7, ATG12, LC3b, mTOR) to ascertain their use as markers for the diagnosis of BD in the Egyptian population. The expression of LC3b was upregulated and significantly correlated with the diagnosis of BD. Although the expression levels of ATG5 and mTOR were significantly decreased in the BD patients, they demonstrated low sensitivity. No significant differences in the expression levels of ATG7 and ATG12 were observed in the current study. These data were partially in agreement with the quantitative data of a previous study [10], wherein the mRNA expression levels of all ATGs, including ATG5, ATG7, ATG12, LC3b, mTOR, RAPTOR, and RICTOR, were downregulated in M1 macrophages from BD patients when compared to those in healthy individuals. On the other hand, higher levels of ATG5 mRNA in BD patients compared to healthy controls [24]. The mRNA expression levels in the present study confirm the correlation between the inflammatory complications of BD and the disruption of the autophagy process. A similar finding was reported in another study done by Liang et al. [55] which demonstrated that macroautophagy played a role in Parkinson’s disease, a neurodegenerative disorder that shared its autoimmune features with BD; the authors observed abnormal expression levels for ATG5, ATG7, ATG12, and LC3b.
Several triggers, including food deprivation [56], hypoxia [57], oxidative stress [58], pathogen infection [59], and endoplasmic reticulum stress [60], can trigger the evolutionarily conserved process of autophagy through different signaling pathways.
Apoptosis can be prevented by the mammalian target of rapamycin (mTOR), which can also encourage cell proliferation in the presence of nutrients and cytokines [61]; while stress and food deprivation prevent mTOR from activating several molecular complexes, comprising the transmembrane protein complex, the PI3K complex, the unc-51-like kinase (ULK) complex, and two ubiquitin-like protein conjugation systems (Atg12 and LC3), to initiate autophagy [62].
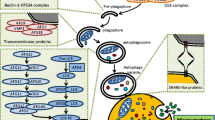
Assembly of the ULK complex triggers the start of autophagy by phosphorylating AMBRA1 to activate the PI3K complex. Membrane nucleation is regulated by class III PI3K and Beclin-1. In order to fundamentally prevent the premature fusion of the vesicles and lysosomes, the Atg5- Atg12-Atg16 complex is drawn to the pre-autophagosomal structure (PAS). Here, it makes contact with the phagophore's outer membrane [63].
The second ubiquitin-like system enhances the interaction between phosphatidylethanolamine (PE) and Atg8 / microtubule-associated protein 1 light chain 3 (LC3). A pathogen ingested by LC3 is destroyed and degraded at a higher rate because of its high affinity for the lysosome, while coupled to the phagosome [64]. LC3 is processed into LC3II via Atg4, Atg7, and Atg3 [65] and is necessary for the expansion and completion of the autophagic membrane and is present on its outer and inner surfaces of it. The Atg5-Atg12-Atg16 complex gets out of autophagosome. Atg9 is necessary for the formation of the intraluminal vesicles and the localized acidification within the autolysosome [66]. It is translocated to the autophagosome formation site, where it provides for the phagophore membrane [67], following which the autophagosome merges with the lysosome to form the autophagosome [68], Fig. 3.
The inflammatory complications of BD may be interpreted by the downregulation of the ATG5 gene, which inhibits the formation of autophagosomes in mammalian cells or mice, and enhances the inflammatory reaction [57]. These inflammatory reactions may occur due to the hypersensitivity of T cells against multiple antigens. Following T-cell activation, the monocytes are activated through CD40-CD154 interactions and various T-cell-derived cytokines (IFN-γ and TNF-α) to produce IL-12, which enhances the shift to Th1 responses. Consequently, this abnormal T-cell activation leads to neutrophil activation, which can be triggered by cytokines such as IL-8, IL-17, IFN-γ, and TNF-α. Also, circulating proteins (C3, C4, C5, IgA, and Haptoglobin) were increased [69]. Atg5 and Atg16L1-deficient macrophages produce significant amounts of proinflammatory cytokines after lipopolysaccharide stimulation [70]. One study reported a direct association between low ATG5 levels and autoimmunity; the genome-wide association study showed that many autoimmune diseases, such as multiple myeloma, SLE, rheumatoid arthritis, and systemic sclerosis, were correlated with low ATG5 levels [71]. Alternatively, another study reported that the upregulation of ATG5 was associated with blood pressure, insulin sensitivity, and glucose homeostasis [72].
Similar to the findings of the current study, environmental cues, including starvation, were reported to suppress mTOR and repress autophagy in another study [73]. Furthermore, a shortage of nutrients following mTOR inhibition resulted in reduced biosynthesis and increased autophagy in BD patients [74]. mTOR stimulates anabolic cellular processes that result in growth. Even though autophagy is a fundamental catabolic function in the cell [75, 76]. The conserved serine/threonine kinase mTOR belongs to the family of protein kinases called phosphatidylinositol 3-kinase of and is a downstream effector of PI3K/AKT pathway [77].
Inflammation and immunity are intricately correlated with the autophagy pathway and its proteins [78]; autophagy-related proteins play a part in the activation and inhibition of inflammatory and immunological responses. Moreover, inflammatory and immune signals are involved in the activation and inhibition of autophagy, Fig. 4. Therefore, the pathophysiology of many infectious diseases and inflammatory disorders may be caused by deficiencies in autophagy, which might be caused by mutations in the autophagy gene.
The two different mTORC1/2 complexes and how autophagy is regulated by them. DEPTOR and PIKK are components shared by mTOR complexes, whereas PRAS40 and RAPTOR are special to mTORC1 and RICTOR and mSIN1 are limited to mTORC2. When mTORC1 phosphorylates the MiT-TFE factors, ATG13 and ULK1, autophagy cannot be positively regulated. Subsequently, Beclin-1, GFAP, and VDAC1 are phosphorylated by mTORC2 to prevent them from also inducing autophagy. The subcellular location of FOXO proteins is altered by mTORC2-mediated phosphorylation, which has a variety of effects on the control of autophagy
The energy depletion could activate adenosine monophosphate (AMP) protein kinase and further stimulate the mTOR substrate complex to amplify the formation of autophagosomes [79]. mTOR has a core function in controlling metabolic programs and is essential for linking metabolism with immune functions [80]. The metabolic apparatus involved in food uptake and glycolysis must be upregulated for T cells to function properly. Thus, mTOR inhibition contributes to the inhibition of T-cell function [81]. Studies have indicated that the activity of mTOR may play a crucial role in incorporating immunological microenvironment cues to control the differentiation of helper cells [82].
LC3 is considered as an autophagy factor that exists in a soluble (LC3-1) or lipidated (LC3-II) form [83]. LC3-1 is transformed into LC3-II and degraded after the fusion of the lysosomal autophagosomes. Moreover, LC3 is a crucial protein in the autophagy pathway, where it plays a role in the selection of the autophagy substrate and biogenesis of the autophagosome [84]. LC3 has been extensively utilized to track the number of autophagosomes and the autophagic activity [85]. Therefore, the quantity of LC3 is associated with the number of autophagosomes and is considered as a predictor of autophagic behavior [30]. However, increased LC3 expression cannot reliably represent increased autophagic activity. It may also suggest a decrease in autophagic function due to fusion blocking following an increase in the number of autophagosomes [34], which may explain the high expression level of LC3b in the current study. A previous study stated that LC3 is a major way of autophagy in phagocytes such as dendritic cells (DCs) and macrophages to remove intracellular pathogens; thus, autophagy involves LC3 and pathogens in single-membrane phagosomes [86].
Moreover, it is clear that in BD there is an ongoing autoimmune cascade resulting from signals from infected cells that the host releases. This immune reaction would override any outside influences. On a favorable genetic landscape, T cells and other antigen-presenting cells would direct the cycle to continue. Uncontrolled adaptive responses would be activated and allowed to continue as a persistent pathogenic presence via autoantigens that activate the dendritic T cells and B cells [87]. Both adaptive and innate immune systems are stimulated with a proinflammatory and Th1-type cytokine profile in BD. BD might be accompanied by a genetic mutation that affects an adhesion molecule, a proinflammatory cytokine/chemokine, a transcription factor, or a regulatory component and predispose to early or more intense neutrophil and T-cell responses. This concept, which also explains the “pathergy” or “skin urate” tests, is represented by the increased neutrophil response to urate crystals [88].
In the current study, associations between the expression levels of the autophagy genes and the clinical characteristics of the BD patients were examined at baseline. The mRNA expression level of ATG5 was significantly associated with patient age and the incidence of oral ulcers, whereas that of ATG7 was significantly associated with patient age and the presence of erythema nodosum and vascular lesions. However, LC3b showed a significant association with the presence of pustules. One study reported that ATG5-deficient mice, particularly in the neural cells, may develop gradual impairment of motor function with increased cytoplasmic inclusion bodies in the neurons [89]. In another study, mice lacking the ATG5 gene died within one day after giving birth [57]. Furthermore, elevated expression levels of several ATG genes were found to be significantly linked to a high patient survival rate, thereby indicating the potential of Atg proteins as useful prognostic markers [90].
Conclusion
The present study showed that LC3b expression might prove as an accurate and reliable diagnostic marker for patients with BD. The expression of this gene was correlated with the presence of pustules, whereas low levels of ATG5 were associated with the presence of oral ulcers in patients with BD. Nonetheless, additional national and international collaborative efforts using larger sample sizes are warranted to conduct genetic and/or molecular studies to confirm the findings of this study and identify the most reliable marker for BD.
Availability for data and materials
The datasets used and / or analysed during the current study are available from the corresponding author on reasonable request.
Change history
12 December 2022
A Correction to this paper has been published: https://doi.org/10.1186/s43042-022-00376-9
Abbreviations
- BD:
-
Behçet's disease
- ATG:
-
Autophagy-related gene
- Atg:
-
Autophagy-related protein
- LC3:
-
Light chain 3
- mTOR:
-
Mammalian target of rapamycin
- VVV:
-
Variable vessel vasculitis
- HLA-B51:
-
Human leukocyte antigen
- RAPTOR:
-
Regulatory associated protein of mTOR
- RICTOR:
-
Rapamycin-insensitive companion of mTOR
- mTORC1:
-
MTOR complex 1
- mTORC2:
-
MTOR complex 2
- TLR:
-
Toll-like receptor
- ROC:
-
Receiver operating characteristic
- AUC:
-
Area under a curve
References
McHugh J (2021) Different phenotypes identified for Behçet syndrome. Nat Rev Rheumatol 17:188. https://doi.org/10.1038/s41584-021-00587-1
Jennette JC (2013) Overview of the 2012 revised International Chapel Hill Consensus Conference nomenclature of vasculitides. Clin Exp Nephrol 17:603–606. https://doi.org/10.1007/s10157-013-0869-6
Greco A, De Virgilio A, Ralli M, Ciofalo A, Mancini P, Attanasio G et al (2018) Behçet’s disease: New insights into pathophysiology, clinical features and treatment options. Autoimmun Rev 17:567–575. https://doi.org/10.1016/j.autrev.2017.12.006
Gheita TA, El-Latif EA, El-Gazzar II, Samy N, Hammam N, Abdel Noor RA et al (2019) Behçet’s disease in Egypt: a multicenter nationwide study on 1526 adult patients and review of the literature. Clin Rheumatol 38:2565–2575. https://doi.org/10.1007/s10067-019-04570-w
Emmi G, Becatti M, Bettiol A, Hatemi G, Prisco D, Fiorillo C (2019) Behçet’s syndrome as a model of thrombo-inflammation: the role of neutrophils. Front Immunol 10:1085. https://doi.org/10.3389/fimmu.2019.01085
Taylor J, Glenny AM, Walsh T, Brocklehurst P, Riley P, Gorodkin R et al (2014) Interventions for the management of oral ulcers in Behçet’s disease. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD011018.pub2
Saito R, Nishimura K, Mukoyama H, Nakamura Y, Nagamoto T, Akashi K, et al. (2017) THU0571 The clinical features of 223 behcet’s disease patients in Japan. In: Annals of Rheumatic Diseases BMJ 2017; vol 422. pp 1–422. https://doi.org/10.1136/annrheumdis-2017-eular.4216.
Scherrer MAR, Rocha VB, Garcia LC (2017) Behçet´s disease: Review with emphasis on dermatological aspects. An Bras Dermatol 92:452–464. https://doi.org/10.1590/abd1806-4841.20177359
Tong B, Liu X, Xiao J, Su G (2019) Immunopathogenesis of Behcet’s disease. Front Immunol 10:1–12. https://doi.org/10.3389/fimmu.2019.00665
Palizgir MT, Akhtari M, Shahram F, Mostafaei S, Akhlaghi M, Sobhani S et al (2018) Downregulation of autophagy-related genes in macrophages from patients with Behcet’s disease. CRESCENT J Med Biol Sci 5:14–20
Yildiz M, Koker O, Adrovic A, Sahin S, Barut K, Kasapcopur O (2020) Pediatric Behçet’s disease - clinical aspects and current concepts. Eur J Rheumatol 7:38–47. https://doi.org/10.5152/eurjrheum.2019.19121
Diagnosis of Behcet’s Disease | American Behcets Disease Association, (n.d.). https://www.behcets.com/basics-of-behcets/diagnosis/ (accessed June 18, 2021).
International Study Group for Behçet’s Disease (1990) Criteria for diagnosis of Behcet’s disease. Lancet 335:1078–1080. https://doi.org/10.1016/0140-6736(90)92643-V
Yildizhan IK, Boyvat A (2020) Diagnostic sensitivity of different applications of pathergy test for behçet’s disease. Arch Rheumatol 35(1):29
Erdem C, Gürler A, Kundakçı N (1987) Behçet hastalarında üç aşamalı paterji testi ile elde edilen sonuçlar. Lepr Mec 18:73–84
Ergun T (2021) Pathergy Phenomenon. Front Med 8:639404. https://doi.org/10.3389/fmed.2021.639404
Kutlubay Z, Tüzün Y, Wolf R (2015) The pathergy test as a diagnostic tool. Skinmed 15:97–104
Hahn HJ, Kwak SG, Kim DK, Kim JY (2021) Association of Behçet disease with psoriasis and psoriatic arthritis. Sci Rep 11:1–10. https://doi.org/10.1038/s41598-021-81972-4
Hasan MS, Ryan PL, Bergmeier LA, Fortune F (2017) Circulating NK cells and their subsets in Behçet’s disease. Clin Exp Immunol 188:311–322. https://doi.org/10.1111/cei.12939
Graef M (2020) Recent advances in the understanding of autophagosome biogenesis. F1000Research 9:1–7. https://doi.org/10.12688/f1000research.22111.1
Kobayashi S (2015) Choose delicately and reuse adequately: the newly revealed process of autophagy. Biol Pharm Bull 38:1098–1103. https://doi.org/10.1248/bpb.b15-00096
Levine B, Mizushima N, Virgin HW (2011) Autophagy in immunity and inflammation. Nature 469:323–335. https://doi.org/10.1038/nature09782
Harding TM, Morano KA, Scott SV, Klionsky DJ (1995) Isolation and characterization of yeast mutants in the cytoplasm to vacuole protein targeting pathway. J Cell Biol 131:591–602. https://doi.org/10.1083/jcb.131.3.591
Zheng M, Yu H, Zhang L, Li H, Liu Y, Kijlstra A et al (2015) Association of ATG5 gene polymorphisms with behçet’s disease and ATG10 gene polymorphisms with VKH syndrome in a chinese han population Investig. Ophthalmol Vis Sci 56:8280–8287. https://doi.org/10.1167/iovs.15-18035
Yang L, Liu C, Zhao W, He C, Ding J, Dai R et al (2018) Impaired autophagy in intestinal epithelial cells alters gut microbiota and host immune responses. Appl Environ Microbiol 84(18):e00880-e918
Juhász G, Érdi B, Sass M, Neufeld TP (2007) Atg7-dependent autophagy promotes neuronal health, stress tolerance, and longevity but is dispensable for metamorphosis in Drosophila. Genes Dev 21:3061–3066. https://doi.org/10.1101/gad.1600707
Pattison JS, Osinska H, Robbins J (2011) Atg7 induces basal autophagy and rescues autophagic deficiency in CryABR120G cardiomyocytes. Circ Res 109:151–160. https://doi.org/10.1161/CIRCRESAHA.110.237339
Sakoh-Nakatogawa M, Matoba K, Asai E, Kirisako H, Ishii J, Noda NN et al (2013) Atg12-Atg5 conjugate enhances E2 activity of Atg3 by rearranging its catalytic site. Nat Struct Mol Biol 20:433–439. https://doi.org/10.1038/nsmb.2527
Otomo C, Metlagel Z, Takaesu G, Otomo T (2013) Structure of the human ATG12∼ATG5 conjugate required for LC3 lipidation in autophagy. Nat Struct Mol Biol 20:59–66. https://doi.org/10.1038/nsmb.2431
Brier LW, Ge L, Stjepanovic G, Thelen AM, Hurley JH, Schekman R (2019) Regulation of LC3 lipidation by the autophagyspecific class III phosphatidylinositol-3 kinase complex. Mol Biol Cell 30:1098–1107. https://doi.org/10.1091/mbc.E18-11-0743
Klionsky DJ, Abdel-Aziz AK, Abdelfatah S, Abdellatif M, Abdoli A, Abel S et al (2021) Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition). Autophagy. https://doi.org/10.1080/15548627.2020.1797280
Sabatini DM, Erdjument-Bromage H, Lui M, Tempst P, Snyder SH (1994) RAFT1: a mammalian protein that binds to FKBP12 in a rapamycin-dependent fashion and is homologous to yeast TORs. Cell 78:35–43. https://doi.org/10.1016/0092-8674(94)90570-3
Lipton JO, Sahin M (2014) The neurology of mTOR. Neuron 84:275–291. https://doi.org/10.1016/j.neuron.2014.09.034
Jia J, Abudu YP, Claude-Taupin A, Gu Y, Kumar S, Choi SW et al (2018) Galectins control mTOR in response to endomembrane damage. Mol Cell 70:120-135.e8. https://doi.org/10.1016/j.molcel.2018.03.009
Hasegawa J, Maejima I, Iwamoto R, Yoshimori T (2015) Selective autophagy: lysophagy. Methods 75:128–132. https://doi.org/10.1016/j.ymeth.2014.12.014
Adeeb F, Khan MU, Stack AG, Fraser AD (2017) Etiology, immunopathogenesis and biomarkers in Behçet’s disease. In: Behcet’s Dis. InTech 2017. https://doi.org/10.5772/intechopen.68342
Arnson Y, Amital H, Shoenfeld Y (2007) Vitamin D and autoimmunity: new aetiological and therapeutic considerations. Ann Rheum Dis 66:1137–1142. https://doi.org/10.1136/ard.2007.069831
Do JE, Kwon SY, Park S, Lee ES (2008) Effects of vitamin D on expression of Toll-like receptors of monocytes from patients with Behçet’s disease. Rheumatology 47:840–848. https://doi.org/10.1093/rheumatology/ken109
Association WM (2013) World medical association declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA J Am Med Assoc 310:2191–2194. https://doi.org/10.1001/jama.2013.281053
Häntzsch M, Tolios A, Beutner F, Nagel D, Thiery J, Teupser D et al (2014) Comparison of whole blood RNA preservation tubes and novel generation RNA extraction kits for analysis of mRNA and miRNA profiles. PLoS ONE. https://doi.org/10.1371/journal.pone.0113298
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622. https://doi.org/10.1373/clinchem.2008.112797
Longo MC, Berninger MS, Hartley JL (1990) Use of uracil DNA glycosylase to control carry-over contamination in polymerase chain reactions. Gene 93:125–128. https://doi.org/10.1016/0378-1119(90)90145-H
Leonardo NM, McNeil J (2015) Behcet’s disease: Is there geographical variation. A review far from the silk road. Int J Rheumatol. https://doi.org/10.1155/2015/945262
Fernández-Ávila DG, Rincón-Riaño DN, Bernal-Macías S, Gutiérrez Dávila JM, Rosselli D (2020) Prevalence and demographic characteristics of Behcet disease in Colombia: data from the national health registry 2012–2016. Rheumatol Int 40:17–20. https://doi.org/10.1007/s00296-019-04466-7
Shadmanfar S, Masoumi M, Davatchi F, Shahram F, Akhlaghi M, Faezi ST et al (2021) Correlation of clinical signs and symptoms of Behçet’s disease with platelet-to-lymphocyte ratio (PLR) and neutrophil-to-lymphocyte ratio (NLR). Immunol Res 1:3–12. https://doi.org/10.1007/s12026-021-09194-4
Dalvi SR, Yildirim R, Yazici Y (2012) Behçet’s syndrome. Drugs 72:2223–2241
Mendes D, Correia M, Barbedo M, Vario T, Mota M, Goncalves O, Valente J (2009) Behçet’s disease—a contemporary review. J Autoimmune 32(3–4):178–188
Kural-Seyahi E, Fresko I, Seyahi N, Ӧzyazgan Y, Mat C, Hamuryudan V, Yurdakul S, Yazici H (2003) The long term mortality and morbidity of Behçet’s syndrome: a 2-decade outcome survey of 387 patients followed of a dedicated center. Medicine (Baltimore) 82(1):60–76
Maldini C, Lavalley MP, Cheminant M, de Menthon M, Mahr A (2012) Relationships of HLA-B51 or B5 genotype with Behçet’s disease clinical characteristics: systemic review and meta-analysis of observational studies. Rheumatology 51(5):887–900
Tugal-Tukan I (2009) Behçet’s uveitis. Middle East Afr J Opthalmology 16(4):219–224
Saccucci M, Di Carlo G, Bossù M, Giovarruscio F, Salucci A, Polimeni A (2018) Autoimmune diseases and their manifestations on oral cavity: diagnosis and clinical management. J Immunol Res. https://doi.org/10.1155/2018/6061825
Conway KL, Kuballa P, Khor B, Zhang M, Shi HN, Virgin HW et al (2013) ATG5 regulates plasma cell differentiation. Autophagy 9:528–537. https://doi.org/10.4161/auto.23484
Le Pham D, Kim SH, Losol P, Yang EM, Shin YS, Ye YM et al (2016) Association of autophagy related gene polymorphisms with neutrophilic airway inflammation in adult asthma. Korean J Intern Med 31:375–385. https://doi.org/10.3904/kjim.2014.390
van Beek N, Klionsky DJ, Reggiori F (2018) Genetic aberrations in macroautophagy genes leading to diseases. Biochim Biophys Acta Mol Cell Res 1865:803–816. https://doi.org/10.1016/j.bbamcr.2018.03.002
Liang L, Zhou Q, Feng L (2021) Decreased microRNA-155 in Behcet’s disease leads to defective control of autophagy thereby stimulating excessive proinflammatory cytokine production. Arthritis Res Ther. https://doi.org/10.1186/s13075-021-02517-8
Joy S, Agrawal P, Singh A, Sagar BKC, Manjithaya R, Surolia N (2018) Basal and starvation-induced autophagy mediates parasite survival during intraerythrocytic stages of Plasmodium falciparum. Cell Death Discov 4(1):1–13
Yang Z, Goronzy JJ, Weyand CM (2015) Autophagy in autoimmune disease. J Mol Med 93:707–717. https://doi.org/10.1007/s00109-015-1297-8
Li S, Hong M, Tan H-Y, Wang N, Feng Y (2016) Insights into the role and interdependence of oxidative stress and inflammation in liver diseases. Oxid Med Cell Longev 2016:1–21
Ahmad L, Mostowy S, Sancho-Shimizu V (2018) Autophagy-virus interplay: from cell biology to human disease. Front Cell Dev Biol 6:155
Lee WS, Yoo WH, Chae HJ (2015) ER stress and autophagy. Curr Mol Med 15(8):735–745
Kamada Y, Yoshino K-I, Kondo C, Kawamata T, Oshiro N, Yonezawa K et al (2010) Tor directly controls the Atg1 kinase complex to regulate autophagy. Mol Cell Biol 30(4):1049–1058
Mercer TJ, Gubas A, Tooze SA (2018) A molecular perspective of mammalian autophagosome biogenesis. J Biol Chem 293(15):5386–5395
Kaur J, Debnath J (2015) Autophagy at the crossroads of catabolism and anabolism. Nat Rev Mol Cell Biol 16(8):461–472
Herb M, Gluschko A, Schramm M (2020) LC3-associated phagocytosis - The highway to hell for phagocytosed microbes. Semin Cell Dev Biol 101:68–76
Glick D, Barth S, Macleod KF (2010) Autophagy: cellular and molecular mechanisms. J Pathol 221(1):3–12
Bader CA, Shandala T, Ng YS, Johnson IRD, Brooks DA (2015) Atg9 is required for intraluminal vesicles in amphisomes and autolysosomes. Biol Open 4(11):1345–1355
Mari M, Griffith J, Rieter E, Krishnappa L, Klionsky DJ, Reggiori F (2010) An Atg9-containing compartment that functions in the early steps of autophagosome biogenesis. J Cell Biol 190(6):1005–1022
Bednarczyk M, Zmarzły N, Grabarek B, Mazurek U, Muc-Wierzgoń M (2018) Genes involved in the regulation of different types of autophagy and their participation in cancer pathogenesis. Oncotarget 9(76):34413
Türsen Ü (2012) Pathophysiology of the Behçet’s disease. Pathol Res Int 2012:11. https://doi.org/10.1155/2012/493015
Saitoh T, Fujita N, Jang MH, Uematsu S, Yang BG, Satoh T et al (2008) Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456:264–268. https://doi.org/10.1038/nature07383
Mitchell JS, Li N, Weinhold N, Försti A, Ali M, Van Duin M et al (2016) Genome-wide association study identifies multiple susceptibility loci for multiple myeloma. Nat Commun 7:22. https://doi.org/10.1038/ncomms12050
Keller CW, Lünemann JD (2017) Autophagy and autophagy-related proteins in CNS autoimmunity. Front Immunol 8:165. https://doi.org/10.3389/fimmu.2017.00165
Saxton RA, Sabatini DM (2017) mTOR signaling in growth, metabolism, and disease. Cell 168:960–976. https://doi.org/10.1016/j.cell.2017.02.004
Yu L, McPhee CK, Zheng L, Mardones GA, Rong Y, Peng J et al (2010) Termination of autophagy and reformation of lysosomes regulated by Mtor. Nature 465:942–946. https://doi.org/10.1038/nature09076
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6(4):463–477
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147(4):728–741
Mitra A, Luna JI, Marusina AI, Merleev A, Kundu-Raychaudhuri S, Fiorentino D et al (2015) Dual mTOR inhibition is required to prevent TGF-β-mediated fibrosis: implications for scleroderma. J Invest Dermatol 135(11):2873
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132(1):27–42
Holczer M, Hajdú B, Lőrincz T, Szarka A, Bánhegyi G, Kapuy O (2019) A double negative feedback loop between mTORC1 and AMPK kinases guarantees precise autophagy induction upon cellular stress. Int J Mol Sci. https://doi.org/10.3390/ijms20225543
Jones RG, Thompson CB (2007) Revving the engine: signal transduction fuels T cell activation. Immunity 27:173–178. https://doi.org/10.1016/j.immuni.2007.07.008
Powell JD, Pollizzi KN, Heikamp EB, Horton MR (2012) Regulation of immune responses by mTOR. Annu Rev Immunol 30:39–68. https://doi.org/10.1146/annurev-immunol-020711-075024
Murphy KM, Stockinger B (2010) Effector T cell plasticity: Flexibility in the face of changing circumstances. Nat Immunol 11:674–680. https://doi.org/10.1038/ni.1899
Delorme-Axford E, Klionsky DJ (2020) The LC3-conjugation machinery specifies cargo loading and secretion of extracellular vesicles. Autophagy 16:1169–1171. https://doi.org/10.1080/15548627.2020.1760057
Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A et al (2016) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 12(1):1–222
Rogov V, Dötsch V, Johansen T, Kirkin V (2014) Interactions between autophagy receptors and ubiquitin-like proteins form the molecular basis for selective autophagy. Mol Cell 53(2):167–178
Yin H, Wu H, Chen Y, Zhang J, Zheng M, Chen G et al (2018) The therapeutic and pathogenic role of autophagy in autoimmune diseases. Front Immunol. https://doi.org/10.3389/fimmu.2018.01512
Zeidan MJ, Saadoun D, Garrido M, Klatzmann D, Six A, Cacoub P (2016) Behçet’s disease physiopathology: a contemporary review. Autoimmun Highlights 7:4. https://doi.org/10.1007/s13317-016-0074-1
Direskeneli H, editor (2003) Behçet's disease: from innate to adaptive immunity. In: Arthritis Research & Therapy, Springer
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R et al (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889. https://doi.org/10.1038/nature04724
Bortnik S, Gorski SM (2017) Clinical applications of autophagy proteins in cancer: from potential targets to biomarkers. Int J Mol Sci. https://doi.org/10.3390/ijms18071496
Acknowledgements
I thank all of my research team; Dr. Eman, dr. Rania, dr. Abeer and dr. Shimaa. Also I thank my father and my mother for caring my baby girl, Mariam to help me continue my study.
Funding
The study was done on my personal account.
Author information
Authors and Affiliations
Contributions
All authors have read and approved the final version of the manuscript. Dr. E.M.S.: is the head of the supervision. Dr. A.R.L.A. and Mrs. D.N.S.: contributed to samples collection from patients in Kasr Alainy school of Medicine; Cairo university. Dr. A.R.L. in addition contributed to full clinical diagnosis of all patients. Dr. A.R. and Mrs. D.N.S.: contributed to conducting practical tests of molecular biology of our study. Dr. R.H.M.: contributed to conducting statistical tests of our study, and with Mrs. D.N.S.: writing and revise the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Since the study was done on humans, the approval of Ethics Committee was taken from the National Research Center, Medical Research Ethics Committee, and final ethical approval number: 19047, on May 2, 2019. This was according to the relevant Egyptian laws and with Helsinki Declaration, good medical and laboratory practice (GCP and GLP) guidelines as well as World Health Organization rules regarding the Ethics of scientific research.
Consent for publication
All authors permitted publication and written license was received.
Compting interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised: we have added the initial name for author Doaa N. Saleh.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saleh, D.N., Ramadan, A., Mohammed, R.H. et al. Autophagy-related genes in Egyptian patients with Behçet's disease. Egypt J Med Hum Genet 23, 155 (2022). https://doi.org/10.1186/s43042-022-00367-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43042-022-00367-w