Abstract
Background
Emerging fungal pathogens pose important threats to global public health. The World Health Organization has responded to the rising threat of traditionally neglected fungal infections by developing a Fungal Priority Pathogens List (FPPL). Taking the highest-ranked fungal pathogen in the FPPL, Cryptococcus neoformans, as a paradigm, we review progress made over the past two decades on its global burden, its clinical manifestation and management of cryptococcal infection, and its antifungal resistance. The purpose of this review is to drive research efforts to improve future diagnoses, therapies, and interventions associated with fungal infections.
Methods
We first reviewed trends in the global burden of HIV-associated cryptococcal infection, mainly based on a series of systematic studies. We next conducted scoping reviews in accordance with the guidelines described in the Preferred Reporting Items for Systematic Reviews and Meta-analyses extension for Scoping Reviews using PubMed and ScienceDirect with the keyword Cryptococcus neoformans to identify case reports of cryptococcal infections published since 2000. We then reviewed recent updates on the diagnosis and antifungal treatment of cryptococcal infections. Finally, we summarized knowledge regarding the resistance and tolerance of C. neoformans to approved antifungal drugs.
Results
There has been a general reduction in the estimated global burden of HIV-associated cryptococcal meningitis since 2009, probably due to improvements in highly active antiretroviral therapies. However, cryptococcal meningitis still accounts for 19% of AIDS-related deaths annually. The incidences of CM in Europe and North America and the Latin America region have increased by approximately two-fold since 2009, while other regions showed either reduced or stable numbers of cases. Unfortunately, diagnostic and treatment options for cryptococcal infections are limited, and emerging antifungal resistance exacerbates the public health burden.
Conclusion
The rising threat of C. neoformans is compounded by accumulating evidence for its ability to infect immunocompetent individuals and the emergence of antifungal-resistant variants. Emphasis should be placed on further understanding the mechanisms of pathogenicity and of antifungal resistance and tolerance. The development of novel management strategies through the identification of new drug targets and the discovery and optimization of new and existing diagnostics and therapeutics are key to reducing the health burden.
Similar content being viewed by others
Background
Emerging fungal pathogens and infections pose increasing threats to global public health. People most at risk of invasive fungal disease (IFD) are those with a compromised immune system, due to HIV infection, chemotherapy and immunotherapy for cancer, solid organ transplantation, or other factors. In addition, people with underlying diseases including diabetes mellitus, liver or kidney disease, chronic obstructive pulmonary disease, and viral respiratory tract infections, have been newly identified as an at-risk population.
Viral and bacterial infections tend to be the pathogens that receive the most attention, especially for their potential to cause pandemics; accordingly, human fungal pathogens and IFDs have long been underrecognized. In recent years, however, this perception has changed rapidly, and there is growing concern about the threats posed by fungi to animals and humans. For example, pathogenic species belonging to the phylum Chytridiomycota have been shown to be a major factor leading to the extinction of multiple amphibian species [1, 2]. In addition to the emergence of amphibian fungal pathogens, adaptation to increased temperatures due to climate change has led to the ability of multiple fungal species to overcome mammalian endothermic defenses, leading to their establishment as new human fungal pathogens [3, 4]. Climate change has also altered or expanded the geographic distribution of known pathogenic fungal species, resulting in the emergence of diseases in regions where they were not been previously reported [5]. Officials have already recognized and responded to the rapid increase in the emergence of new fungal pathogens. For instance, an updated Catalogue of Microbial Pathogens Transmitted to Humans released by the National Health Commission of China in 2022 included 107 new fungal pathogens, whereas only 12 new viruses and 4 new species of bacteria were added [6]. Intriguingly, certain fungal pathogens have recently been associated with coronavirus disease 2019 (COVID-19) [7,8,9,10] and cancer developments [11, 12].
The mortality rates of infections caused by known or new pathogenic fungi are often high. Underdiagnosis and the extreme lack of treatment options are important reasons for the high mortality rates [13]. Approved treatments for human fungal infections are limited to four classes of antifungal agents: polyenes, flucytosine, echinocandins, and azoles. Unfortunately, the wide-spread use of existing antifungals in both medicine and agriculture has greatly accelerated the acquisition and emergence of antifungal resistance, which is fundamentally an evolutionary response to selective pressures. The development of new antifungal agents is hampered by the similarity between fungal cells and the cells of their mammalian host, since molecules toxic to fungi tend to be toxic to humans. Because the prevalence of fungal infections has increased and has emerged as a pressing threat to public health, antifungal resistance has now been officially recognized by the listing of fungal pathogens on the Antibiotic Resistance Threats Report produced by the Centers for Disease Control and Prevention (USA) in 2019 [14]. The emergence of drug-resistant strains has further increased the threat of fungal infections; resistance to all four types of antifungal agents has been documented in clinical isolates of fungal pathogens. In response to the rising threats of fungal infections and antifungal resistance, the World Health Organization (WHO) released the Fungal Priority Pathogens List (FPPL) in October 2022 to focus and drive further research, surveillance, and policy interventions [15]. In the WHO FPPL, 19 fungal pathogens were ranked and categorized as critical, high, or medium priority pathogens (Table 1). In particular, Cryptococcus neoformans, Candida auris, Aspergillus fumigatus, and Candida albicans were ranked as “critical” fungal pathogens based on their antifungal resistance, mortality rates, lack of evidence-based diagnostic and treatment options, annual incidence, and complications and sequelae.
Cryptococcus neoformans, the top-ranked fungal pathogen in the WHO FPPL, is a globally distributed opportunistic fungal pathogen that is primarily of environmental origin and that can cause life-threatening cryptococcosis. C. neoformans contain two varieties: C. neoformans var. neoformans and C. neoformans var. grubii. A third variety, C. neoformans var. gattii, was later defined as a distinct species, Cryptococcus gattii. The most recent classification system divides these varieties into seven species [16, 17]. C. neoformans refers to C. neoformans var. grubii. A new species name, Cryptococcus deneoformans, is used for the former C. neoformans var. neoformans. C. gattii is divided into five species. Hence, for the ease of description and discussion, we use C. neoformans to refer to both C. deneoformans and C. neoformans. The mortality rate of cryptococcosis is alarmingly high, especially in patients with HIV infection, in whom the mortality rate ranges from 41% to 61%. Similar to most other fungal infections, access to diagnosis and treatment of cryptococcosis is limited in many countries with developing healthcare systems. Another important issue is that antifungal-resistant clinical isolates have been emerging rapidly, and the mechanisms of antifungal resistance are far from being fully understood. Taking C. neoformans as a paradigm, in this study we review the progress made over the past two decades on the global burden, clinical manifestation and management of cryptococcal infection and on antifungal resistance and tolerance. The purpose of this review is to highlight the emerging threats posed by fungal infections and to drive research efforts in fungal infections to improve future diagnoses, therapies, and interventions.
Methods
Literature search strategy
This scoping review was conducted in accordance with the guidelines described in the Preferred Reporting Items for Systematic Reviews and Meta-analyses extension for Scoping Reviews (PRISMA-ScR) [18]. To identify and select cases and studies for inclusion, a systematic literature search was performed in PubMed and ScienceDirect using the keyword Cryptococcus neoformans for studies published in English since 2000. We used Endnote 20 (Clarivate, Philadelphia, USA) to manage the articles.
Study eligibility criteria and data extraction
Conference abstracts and editorials were excluded, and case reports and research articles were included for review. To be included, the case reports must meet the following criteria: (1) be identified as case reports; (2) include sufficient details about patients; (3) refer to disease states in which C. neoformans was the causative pathogen; (4) provide sufficient details about clinical manifestations; (5) describe treatment details including the specific antifungal therapy administered, if applicable; and (6) describe patient outcomes. The full text of articles that met the inclusion criteria was then searched. Exclusion criteria included studies involving patients under 18 years of age, duplicate studies, and studies for which the full text was not accessible.
Screening of the results of the literature search against the eligibility criteria for case reports and research articles was performed by two independent authors, and any disagreement was mediated by a third author. Failure to reach a consensus was resolved by senior authors. Two groups of authors (group 1: FZ, ZL, and TC; group 2: SW, YS, and ZL) independently extracted information from eligible case reports. The extracted information included first author, year of publication, patient information (country, age, sex, medical or surgical history), clinical manifestation and duration of the infection, site of infection, course of treatment and patient outcome.
Results
Trends in the global burden of HIV-associated cryptococcal infection
Cryptococcus neoformans is a globally distributed opportunistic fungal pathogen of primarily environmental origin, commonly associated with bird feces (especially pigeon feces), soil, and decaying wood. Humans and animals typically acquire the infection from inhaling dust contaminated with bird feces, but direct transmission of cryptococcosis between humans or between animals has not been reported. After inhalation of fungal basidiospores or desiccated yeast cells from the environment, cryptococcal infection initially occurs in the lungs (cryptococcal pneumonia), followed by dissemination of fungal cells to the central nervous system (CNS) (cryptococcal meningitis, CM) and blood (cryptococcemia), which may be achieved through a Trojan horse mechanism. The majority of patients are immunocompromised, with the most significant risk factor being HIV infection.
A series of landmark studies were published in 2009 [19], 2017 [20], and 2022 [21]. Those systematic studies respectively estimated the global and regional burden of HIV-associated CM in 2007, 2014, and 2020, based on available incidence data in HIV-infected cohorts (mainly from the Joint United Nations Programme on HIV/AIDS) and population-based HIV impact assessment surveys. Notably, with the development of improved antiretroviral treatments and systemic antifungal treatments, the global incidence of HIV-associated CM declined from 960,000 cases in 2007 to 220,000 in 2014, and further to 150,000 in 2020 (Table 2). Despite the downward trend in the burden of cryptococcosis, the incidence of cryptococcal infections in immunocompromised individuals remains high (Table 2). For instance, in studies published in 2017 and 2022, the prevalence of cryptococcal antigenemia was estimated to be 6.0% among people with CD4 cell counts of less than 100 cells/µl and 4.4% among HIV-positive people with CD4 cell counts of less than 200 cells/µl, corresponding to 280,000 and 180,000 cases of cryptococcal antigenemia (Table 2), respectively.
Consistent with global trends, the annual incidence of CM in sub-Saharan Africa has been declining (720,000 in 2007, 162,500 in 2014, and 82,000 in 2020), although this region has had, and continues to have, the greatest burden of cryptococcal infection (Table 2). The region with the second highest number of cases is Asia and the Pacific, where the incidence has seemingly reached a steady state (43,200 in 2014 and 44,000 in 2020) with a reduction in CM-associated deaths (from 39,700 to 2014 to 26,000 in 2020) (Table 2). Of note, the incidence of CM has increased by approximately twofold in Europe and North America (from 7400 to 2014 to 12,000 in 2020) and in Latin America (from 5300 to 2014 to 12,000 in 2020). These data highlight the need for further evaluation and refinement of the current control and prevention protocols in these two regions, even though the total burdens are currently at relatively low levels.
In general, there has been a significant geographic redistribution of the estimated global burden of HIV-associated CM since 2009, probably due to the improvement and expansion of highly active antiretroviral therapies (HAART). However, CM still accounts for 19% of AIDS-related deaths annually, according to estimates for both 2017 [20] and 2020 [21]. Although these series of studies have led to a systematic understanding of HIV-associated cryptococcal infections, it is still not possible to accurately assess the total annual incidence and mortality, given the wide variations in the diagnoses and treatments in different regions of the world.
Clinical manifestations of cryptococcal infection
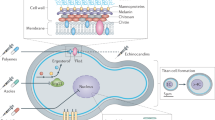
The most common clinical manifestations of cryptococcal infections affect the CNS. Such infections are associated with meningitis or meningoencephalitis, which have high mortality rates (Fig. 1). Another typical manifestation in brain is the formation of cryptococcomas, which are mass lesions caused by infection of the focal tissue. The formation of cryptococcomas depends on an inflammatory response; thus, this manifestation is more common in immunocompetent individuals (Fig. 1). Cryptococcal pneumonia is usually seen when the initial infection occurs through inhalation of infectious propagules, and the involvement of other organs can ensue following the development of cryptococcemia (Fig. 1). In addition to cryptococcal pneumonia and meningitis, C. neoformans can also cause cutaneous cryptococcosis resulting from a primary infection of open skin wounds or a secondary infection from cryptococcal dissemination (Fig. 1).
Clinical manifestation of cryptococcal infection. The most common clinical manifestation of cryptococcal infection are CNS infections, which cause cryptococcal meningitis (Left in the upper panel). Pulmonary infections are the result of initial infection through inhalation of infectious propagules (Right in the upper panel). Another manifestation is cryptococcomas (Lower panel), which is formed by an inflammatory response in brain, lungs, skin, and other organs, thus it is more common in immunocompetent hosts. It may subsequently appear in a complex granuloma, including various macrophages. CNS Central nervous system
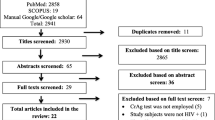
To obtain a systematic understanding of the clinical manifestations and features of cryptococcal infections, we reviewed case reports of cryptococcal infections during the past two decades. Of the 9432 records obtained from keyword searching during the study period, 1103 were identified as case reports. After removing excluded studies and studies that did not provide disease-specific treatment details, patient characteristics, or patient outcomes, 296 were screened for eligibility and full-text review, and 37 studies were eventually retained (Fig. 2). These 37 studies represented 38 individual patients (Table 3).
The age of the patients ranged from 21 to 81 years (median 51 years), and 71% (n = 27) were male. Of the 38 patients, 95% (n = 36) reported a medical or surgical history, 21% (n = 8) were infected with HIV, 18% (n = 7) had a history of organ transplantations (6 renal transplantations, 1 liver transplantation, and 1 orthotopic heart transplantation), 21% (n = 8) had diabetes mellitus, and 18% (n = 7) had liver hepatopathies (5 hepatitis B infection and 2 liver cirrhosis). It is worth mentioning that one patient had been breeding birds [22]. In the identified cases, patients with cryptococcal infections most commonly presented with fever (50%, n = 19), headache (29%, n = 11), and vomiting (21%, n = 8). Less common manifestations included altered mental status and/or confusion (13%, n = 5), cough (13%, n = 5), and drowsiness or fatigue (8%, n = 3). On physical examination, 11% (n = 4) were noted to have body weight loss, and 11% (n = 4) were observed to have weakness in the extremities. Time from symptom onset to hospital presentation ranged from 2 to 210 days. Even though all 38 patients received systemic antifungal treatment, the mortality was as high as 34% (n = 13); over half (7/13) of the deaths were associated with brain infections. Patients’ ages at death ranged from 30 to 75 years (median 45.5 years). Time from hospital presentation or symptom onset to death ranged from 2 to 420 days (median 25.5 days).
Our scoping review in this section included cryptococcal infections associated with the brain, lungs, skin, and other organs in both HIV-positive and HIV-negative cases. This diversity of infection conditions resulted in deviations in clinical manifestations and statistics as compared with HIV-associated CM, which tends to attract the attention of healthcare professionals. To provide a more concentrated analysis, we review the recent research progress focusing mainly on CM in the following sections; we apologize to those whose work could not be properly discussed and cited.
Diagnosis and management of CM
The key reasons for the high mortality of CM include: (1) delays in diagnosis, largely as a result of limited access to lumbar puncture (LP) and rapid diagnostic assays; (2) the limited availability and high cost of currently recommended antifungal agents and intensive care; and (3) the limited ability to monitor and manage treatment-limiting toxicity and the increased intracranial pressure that is frequently associated with CM. Therefore, improving diagnostic abilities and developing more effective treatments would reduce the mortality associated with CM. In this section, we summarize current updates in the diagnosis and antifungal treatment of CM.
Diagnosis
Several protocols are now available for the diagnosis of CM in HIV-infected patients, including India ink microscopy, cerebrospinal fluid (CSF) culture, and detection of cryptococcal antigen in serum or CSF (Fig. 3) [56, 57]. The use of India ink microscopy remains the primary diagnostic tool for identifying Cryptococcus in the CSF. Although India ink microscopy is readily available, it is associated with a low sensitivity of approximately 70–90% [58], particularly in patients with low fungal burdens. Thus, the use of India ink microscopy as the sole diagnostic tool could result in misdiagnosis, particularly soon after symptom onset or in patients undergoing antiretroviral therapies.
Diagnosis of cryptococcal infections. India ink microscopy, cerebrospinal fluid (CSF) culture, and detection of cryptococcal antigen in serum or CSF are three protocols for diagnosis of cryptococcal infections. India ink microscopy remains the primary diagnostic tool for identifying Cryptococcus in CSF. CSF fungal culture is the gold standard for diagnosis of cryptococcal meningitis. The detection of cryptococcal antigens, the capsular polysaccharide glucuronoxylomannan (GXM), is a very sensitive, specific, and effective test to detect cryptococcal infections. The antigen test was mainly performed through the Latex agglutination test (LAT), Enzyme Immunoassay (EIA) and Lateral flow assay (LFA).
CSF fungal culture is the gold standard for the diagnosis of CM, and a positive culture usually implies active cryptococcal disease. It also produces false negative results, similar to India ink microscopy, when the fungal burden is low. To partially overcome this drawback, the CSF volume applied for quantitative fungal culture has been modified from 10 µl to 100 µl in an updated culture protocol [59]. This change led to an improvement of the diagnostic sensitivity of CSF fungal culture from 82.4% to 94.2%. Additional intrinsic drawbacks of the quantitative CSF fungal culture are the general slow-growing nature of the fungus as well as a specific physiological status of Cryptococcus called the viable-but-nonculturable (VBNC) state [60]. Because of these factors, obtaining a quantitative result may require several weeks of culture, and a percentage of cells may not be successfully cultured. In addition, fungal culture requires proper laboratory settings and trained technicians. Nevertheless, quantitative CSF fungal culture remains central for the definitive diagnosis of CM.
LP followed by India ink microscopy or CSF fungal culture is often deferred until the disease is advanced. The detection of cryptococcal antigens, such as the capsular polysaccharide glucuronoxylomannan (GXM) in serum or CSF, has become an essential diagnostic approach and is used for presumptive diagnosis. It is a very sensitive, specific, and effective test that can detect the infection early, ahead of symptom onset and before the disease can develop into life-threatening CM.
Antigen tests are mainly performed in the form of latex agglutination tests (LAT) or enzyme immunoassays (EIA), which are sensitive, specific, and readily available from commercial sources. However, both tests require appropriate laboratory infrastructure and trained technicians, and immunoassay tests tend to be too expensive to allow routine use in resource-limited regions. A major advance, the cryptococcal antigen lateral flow assay (LFA), has revolutionized the diagnosis of CM, particularly in resource-limited settings [61]. LFA is stable at room temperature, requires no specimen preparation, provides results in minutes, and is 100-fold more sensitive to capsular polysaccharides than that of LAT [62, 63]. More significantly, it can be used to detect the cryptococcal antigen in versatile sample types, such as serum, CSF, plasma, and urine, enabling early diagnosis of CM even in facilities where LP or blood sampling is not feasible. Furthermore, semi-quantitative LFA titers have been developed for gross approximation of the fungal burden [64,65,66]. Further studies are needed to investigate the use of semi-quantitative LFA titers for screening potential infections and monitoring treatment responses.
Diagnosis of CM should be relatively easy in HIV-infected patients, given the high fungal burden. The WHO guidelines from 2018 recommended that HIV-infected adults and adolescents who have a CD4 cell count less than 100 cells/µl should be screened for cryptococcal antigen, and the CD4 cell count threshold was expanded to less than 200 cells/µl in the 2022 WHO guidelines. The preferred diagnostic approach recommended in the WHO guidelines of 2022 is prompt LP with measurement of CSF opening pressure and rapid cryptococcal antigen assay. LP with CSF India ink microscopy is the alternative approach, only if access to a cryptococcal antigen assay is not available or rapid results cannot be obtained. Under settings without immediate access to LP or when these diagnostic approaches are clinically contraindicated, rapid serum, plasma, or whole-blood cryptococcal antigen assays are the preferred diagnostic approaches.
CM recognition in immunocompetent and non-HIV-infected cases can be challenging due to the low early fungal burden, which makes fungal culture and LAT techniques less sensitive. In addition, the indolent presentation and subacute nature of symptoms often lead to late diagnoses and consequent disease severity. The improved sensitivity makes the LFA test the preferred diagnostic approach in this context. A low threshold for suspected CM is helpful in the management of the disease, and the use of accessible diagnostic assays to perform early diagnoses is required to achieve lower rates of morbidity and mortality.
Antifungal treatments
Antifungal treatments of IFDs in current clinical practice are limited to only four classes of systemic antifungal agents (azoles, polyenes, pyrimidines, and echinocandins). The limitation in antifungal treatment options for invasive cryptococcal infection is particularly significant, given that Cryptococcus species have intrinsic resistance to echinocandins, and some clinical isolates have been found to acquire resistance to azoles [67, 68]. Therefore, the polyene amphotericin B (AmB) has been prescribed as the primary antifungal drug for the management of cryptococcal infections, despite its toxicity and the very high cost of less toxic formulations [69].
The management of CM is divided into three phases: (1) induction, (2) consolidation, and (3) maintenance of antifungal treatment regimens. The WHO guidelines of 2018 [70] for the treatment of cryptococcal disease in patients infected with HIV recommended a 1-week induction regimen with AmB deoxycholate (1.0 mg/kg per day) and flucytosine (100 mg/kg per day, divided into four daily doses), followed by 1 week of fluconazole (1200 mg/day for adults, 12 mg/kg per day for children and adolescents up to a maximum dose of 800 mg/day); an 8-week consolidation regimen with fluconazole (800 mg/day for adults, 6–12 mg/kg per day for children and adolescents up to a maximum dose of 800 mg/day) following the induction phase; and a maintenance regimen with fluconazole (200 mg/day for adults, 6 mg/kg per day for adolescents and children) until immune reconstitution [70].
Various formulations of AmB are commercially available, including liposomal, deoxycholate, and lipid complex formulations. Of note, these formulations are not interchangeable [71], and only AmB deoxycholate and liposome bilayer-coated AmB (LAmB) have been recommended for managing CM. The WHO guidelines of 2018 raised the possibility that LAmB could be preferrable as a formulation over AmB deoxycholate, considering its equivalent efficacy and improved safety [72]. In March 2022, a phase 3 randomized, controlled, noninferiority trial conducted in five African countries concluded that single-dose LAmB combined with flucytosine and fluconazole was non-inferior to the treatment recommended in the 2018 WHO guidelines for HIV-associated CM and was associated with fewer adverse events [73]. Also considering the extrapolation of evidence supporting the use of a single high dose (10 mg/kg) of LAmB to children [74, 75], the WHO Guideline Development Group updated the recommendations in the 2022 WHO guidelines. These newer guidelines include a single high-dose LAmB-based regimen with 14 days of flucytosine and fluconazole as the preferred induction therapy for managing CM, while previously recommended alternative regimens remain valid [76]. Unfortunately, this updated regimen exacerbates the difficulty of accessing a referred treatment that is already posed by flucytosine, which is expensive and not always available in resource-limited countries. Future efforts should be directed towards improving the accessibility and affordability of flucytosine in low- and middle-income countries with high infection loads.
Antifungal resistance and tolerance in C. neoformans
Cryptococcus neoformans is susceptible to polyenes, flucytosine, and azoles, which are clinically used together in the three-phase therapy of CM. The polyene AmB was the first antifungal drug developed to treat systematic fungal infection, and it is fungicidal rather than fungistatic [77, 78]. Mechanistically, AmB binds to ergosterol-containing membranes, which are the major membranes found in fungal cells, yielding pores in membranes and exerting antifungal activity (Fig. 4) [79]. Acquisition of resistance to AmB by C. neoformans has rarely been reported, but the detection of AmB resistance can be technically challenging, and the true rate of AmB resistance is not known [78, 80, 81]. Nevertheless, cryptococcal isolates with altered AmB sensitivity have been reported [82,83,84,85]; however, the exact mechanisms leading to these changes remain unknown. Cryptococcal AmB resistance has been shown to be caused by alterations in ergosterol biosynthesis through mutations in sterol Δ8-7 isomerase [82,83,84, 86]. On the other hand, AmB resistant isolates without altered ergosterol biosynthesis have also been reported [87, 88], indicating the existence of alternative mechanisms that confer AmB resistance in C. neoformans.
Antifungals, targets, and antifungal resistance mechanism. Three main classes of drugs are used in the treatment of cryptococcal infections: polyenes (AmB), azoles (fluconazole), flucytosine. C. neoformans is intrinsic resistant to echinocandins (shown in dotted lines). AmB binds to ergosterol in the cell membrane, which forming pores and exerting fungicidal activity. Fluconazole targets the ergosterol biosynthetic enzyme Erg11. Flucytosine blocks DNA synthesis. C. neoformans cells develop resistance to different drugs through different mechanisms. The exact mechanism leading to AmB resistance is not yet clear. Fluconazole resistance has been reported to be associated with aneuploidy, heteroresistance and mutations in the ERG11 gene. Flucytosine resistance may be associated with mutations in genes related to flucytosine conversion, hypermutation, and transposon mutagenesis. The echinocandins resistance is attributed to the interaction of Cdc50 and Crm1, and preventing drug uptake could also arise echinocandins resistance
Despite the rarity of AmB-resistant strains, outcomes of AmB therapy appear to be unsatisfactory, and relapses of cryptococcal infections are common; therefore, further investigation into the mechanisms of interaction between fungi and antifungal agents is warranted. In bacteria, both bactericidal resistance and tolerance can affect the outcome of antibiotic therapy. Unlike bacteria with genetically heritable resistance that can replicate in the presence of a drug at concentrations above the minimum inhibitory concentration (MIC), bacteria that are considered “tolerant” are genetically susceptible and can withstand the killing effect of high doses of bactericidal antibiotics. Time-kill curve-based assays are used to evaluate bacterial tolerance to microbicidal antibiotics. In the context of Cryptococcus, Rodero et al. found a correlation between the time-kill curve of AmB and the clinical outcomes of 16 patients with cryptococcal meningitis [89]. A correlation between the time-kill curve and the clinical outcome was also reported by Córdoba et al., whose study encompassed a larger number of patients and isolates (74 clinical strains isolated from 60 patients) [90]. These findings suggest that cryptococcal tolerance to fungicidal AmB may have an impact on the therapeutic outcome of cryptococcal meningitis.
Flucytosine was first chemically synthesized in 1957 as a potent antibacterial and antitumoral compound [91]. As a prodrug, it enters cells via cytosine permease Fcy2, and its function depends on its conversion to 5-fluorouracil by cytosine deaminase Fcy1 and further processing by the uracil phosphoribosyltransferase Fur1. The converted metabolites then inhibit thymidylate synthase activity and consequently inhibits DNA and RNA synthesis [92, 93]. The emergence of resistance to flucytosine prevents its use as a monotherapy drug for fungal infection treatment [92, 94,95,96,97], and it is used in combination with AmB as a first-line induction treatment for cryptococcal infections [76, 98, 99].
Although flucytosine resistance has been well-studied in Candida spp. [100,101,102], little work has been done to investigate resistance in C. neoformans. It has been reported that nonsense mutations within the DNA mismatch repair protein coding gene MSH2 confer flucytosine resistance in clinical isolates of C. neoformans [103]. This study clearly showed that hypermutator phenotypes are associated with the acquisition of resistance to antifungals, including flucytosine, in C. neoformans. Billmyre et al. demonstrated that DNA mismatch repair defects enable rapid acquisition of resistance to flucytosine in C. deuterogattii, the sister species of C. neoformans, and they further identified important mutations in known resistance genes (FUR1 and FCY2) and a capsule biosynthesis-related gene UXS1 [104]. This study provides direct evidence that support the recent appreciation that hypermutation may be a common mechanism that accelerates the acquisition of antifungal resistance in pathogenic fungi. In addition, transposon mutagenesis has been shown to be a contributor to the acquisition of flucytosine resistance during the environment-to-host transition in C. neoformans [105]. While these studies identified potential resistance-related mechanisms that influence flucytosine conversion (Fig. 4), further study is needed to clarify mechanisms leading to flucytosine resistance.
Fluconazole is the most commonly used antifungal agent for the treatment of cryptococcal infections (Fig. 4). It inhibits fungal ergosterol biosynthesis via binding to the cytochrome P450 enzyme sterol 14-demethylase (Erg11 or Cyp51), leading to disrupted cell membrane integrity [106]. Given that an 8-week consolidation regimen of fluconazole followed by a maintenance regimen of low-dose fluconazole is recommended by the 2022 WHO guidelines [76] for CM treatment, the prolonged use and changes in recommended dosages exerted selection pressure and contributed to the prevalence of fluconazole resistance in cryptococcal clinical isolates [107]. A series of thresholds of fluconazole susceptibility has been established to classify clinical isolates: an isolate that exhibits an MIC of most 8 µg/ml is considered susceptible, an isolate with an MIC of 16–32 µg/ml is considered dose-dependent susceptible, and an isolate with an MIC of at least 64 µg/ml is considered resistant [104, 107].
Heteroresistance to fluconazole and other azole antifungal agents is clinically ubiquitous (Fig. 4), and it contributes to the relapse of cryptococcosis during fluconazole maintenance therapy [103, 108]. C. neoformans is innately heteroresistant to fluconazole [109], which primarily occurs by transient duplications of chromosomes [110, 111]. Chromosome 1, which harbors the genes ERG11 and AFR1 (encoding an ABC transporter), is the first chromosome to be duplicated at fluconazole levels higher than the MIC [110], and further increases in drug levels result in the disomy of chromosome 4, which contains SEY1 (encoding a GTPase), GLO3 and GCS2 (encoding the ADP-ribosylation factor GTPase activating proteins) [112]. The duplicated chromosomes or aneuploidy can be readily lost during maintenance in drug-free conditions [110]. Fluconazole resistance in C. neoformans has also been associated with mutations in the ERG11 gene [113, 114]. It is worth to mention that new-generation triazole antifungals with higher activities against resistant and emerging fungal pathogens have been developed either from fluconazole or itraconazole, such as voriconazole, posaconazole, and isavuconazole [115]. The new-generation triazoles have been tested for the treatment of invasive aspergillosis and candidiasis [116], indicating clinical implications for CM treatment.
C. neoformans is intrinsically resistant to echinocandins (Fig. 4), which is paradoxical, as the inhibitory target of echinocandins (β-1,3-glucan synthase) is essential in Cryptococcus [117,118,119]. Huang et al. discovered that a mutation in CDC50, which encodes the β-subunit of membrane lipid flippase, can mediate echinocandin resistance via preventing drug uptake in C. neoformans [117]. Forward genetic screening for cdc50Δ suppressor mutations led to the identification of a homolog of the mechanosensitive channel protein Crm1 that is involved in Cdc50-mediated caspofungin resistance [120]. Cdc50 interacts with Crm1 to regulate calcium homeostasis and caspofungin resistance via calcium/calcineurin signaling [121].
Given the rapid emergence of antifungal resistance and the lack of treatment options, advances in technology to detect antifungal resistance and research focused on understanding antifungal resistance mechanisms will contribute to develop novel antifungal drugs and therapeutic strategies.
Conclusion
The rising threat of C. neoformans is compounded by the accumulating evidence for its capability to infect immunocompetent individuals and the emergence of antifungal-resistant variants. More global surveillance data on antifungal susceptibility combined with molecular typing of C. neoformans would facilitate the correlation of antifungal resistance or tolerance with different genotypes, enabling the use of genotyping strategies to permit data-driven evaluation of risk and promoting the development of corresponding treatment strategies. The construction and integration of robust fungal disease surveillance systems would permit the development of a detailed understanding of global and local epidemiology of C. neoformans and other fungal pathogens, which is especially important for hyper-virulent or drug-tolerant variants. In addition, developing systematic approaches to comprehensively explore the mechanisms of fungal pathogenicity and antifungal resistance and tolerance promises to lead to the identification of new targets for antifungal drugs and the development and optimization of new and existing diagnostic and therapeutic approaches, thus providing important safeguards to reduce the morbidity and mortality of fungal infections.
Data availability
All data generated or analyzed during this study are included in this published article.
Abbreviations
- WHO:
-
World Health Organization
- FPPL:
-
Fungal Priority Pathogens List
- PRISMA-ScR:
-
Preferred reporting items for systematic reviews and meta-analyses extension for scoping reviews
- IFD:
-
Invasive fungal disease
- HIV:
-
Human immunodeficiency virus
- HAART:
-
Highly active antiretroviral therapy
- AIDS:
-
Acquired immunodeficiency syndrome
- CM:
-
Cryptococcal meningitis
- CNS:
-
Central nervous system
- CSF:
-
Cerebrospinal fluid
- CrAg:
-
Cryptococcal antigen
- LP:
-
Lumbar puncture
- GXM:
-
Glucuronoxylomannan
- LAT:
-
Latex agglutination test
- EIA:
-
Enzyme immunoassay
- LFA:
-
Lateral flow assay
- AmB:
-
Amphotericin B
- LAmB:
-
Liposome bilayer-coated amphotericin B
- ABLC:
-
Amphotericin B lipid complex
- MIC:
-
Minimum inhibitory concentration
- MBL:
-
Monoclonal B-cell lymphocytosis
- CD:
-
Cluster of differentiation
- HCV:
-
Hepatitis C virus
- TB:
-
Tuberculosis
- ICD:
-
Implantable cardioverter-defibrillator
- MBL:
-
Monoclonal B-cell lymphocytosis
- CA19-9:
-
Carbohydrate antigen 19-9
References
Daszak P, Cunningham AA, Hyatt AD. Emerging infectious diseases of wildlife—threats to biodiversity and human health. Science. 2000;287:443–9.
Smith KF, Sax DF, Lafferty KD. Evidence for the role of infectious disease in species extinction and endangerment. Conserv Biol. 2006;20:1349–57.
Nnadi NE, Carter DA. Climate change and the emergence of fungal pathogens. PLoS Pathog. 2021;17:e1009503.
Wu X, Lu Y, Zhou S, Chen L, Xu B. Impact of climate change on human infectious diseases: empirical evidence and human adaptation. Environ Int. 2016;86:14–23.
Casadevall A. Climate change brings the specter of new infectious diseases. J Clin Investig. 2020;130:553–5.
Nation Health Commission of China. An updated catalogue of microbial pathogens transmitted to humans 2022. http://www.nhc.gov.cn/wjw/yjzj/202112/94fcc4480ea2403e9c51c641645d6c20.shtml. Accessed 20 Feb 2023.
Hoenigl M, Seidel D, Sprute R, Cunha C, Oliverio M, Goldman GH, et al. COVID-19-associated fungal infections. Nat Microbiol. 2022;7:1127–40.
Verweij PE, Gangneux J-P, Bassetti M, Brüggemann RJ, Cornely OA, Koehler P, et al. Diagnosing COVID-19-associated pulmonary aspergillosis. Lancet Microbe. 2020;1:e53–5.
Raut A, Huy NT. Rising incidence of mucormycosis in patients with COVID-19: another challenge for India amidst the second wave? Lancet Respir Med. 2021;9:e77.
Hughes S, Troise O, Donaldson H, Mughal N, Moore LSP. Bacterial and fungal coinfection among hospitalized patients with COVID-19: a retrospective cohort study in a UK secondary-care setting. Clin Microbiol Infect. 2020;26:1395–9.
Dohlman AB, Klug J, Mesko M, Gao IH, Lipkin SM, Shen X, et al. A pan-cancer mycobiome analysis reveals fungal involvement in gastrointestinal and lung tumors. Cell. 2022;185:3807–22.
Narunsky-Haziza L, Sepich-Poore GD, Livyatan I, Asraf O, Martino C, Nejman D, et al. Pan-cancer analyses reveal cancer-type-specific fungal ecologies and bacteriome interactions. Cell. 2022;185:3789–806.
Fisher MC, Hawkins NJ, Sanglard D, Gurr SJ. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science. 2018;360:739–42.
Centers of Disease Control and Prevention. Antibiotic resistance threats in the United States 2019. https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf. Accessed 20 Feb 2023.
World Health Organization. WHO fungal priority pathogens list to guide research, development and public health action 2022. https://www.who.int/publications/i/item/9789240060241. Accessed 20 Feb 2023.
Hagen F, Khayhan K, Theelen B, Kolecka A, Polacheck I, Sionov E, et al. Recognition of seven species in the Cryptococcus gattii/Cryptococcus neoformans species complex. Fungal Genet Biol. 2015;78:16–48.
Zhao Y, Lin J, Fan Y, Lin X. Life cycle of Cryptococcus neoformans. Annu Rev Microbiol. 2019;73:17–42.
Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. Prisma extension for scoping reviews (PRISMA-SCR): checklist and explanation. Ann Intern Med. 2018;169:467–73.
Park BJ, Wannemuehler KA, Marston BJ, Govender N, Pappas PG, Chiller TM. Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS. AIDS. 2009;23:525–30.
Rajasingham R, Smith RM, Park BJ, Jarvis JN, Govender NP, Chiller TM, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis. 2017;17:873–81.
Rajasingham R, Govender NP, Jordan A, Loyse A, Shroufi A, Denning DW, et al. The global burden of HIV-associated cryptococcal infection in adults in 2020: a modelling analysis. Lancet Infect Dis. 2022;22:1748–55.
Huong NT, Altibi AM, Hoa NM, Tuan LA, Salman S, Morsy S, et al. Progressive cutaneous cryptococcosis complicated with meningitis in a myasthenia gravis patient on long-term immunosuppressive therapy—a case report. BMC Infect Dis. 2017;17:311.
Abbas A, Sikkel MB, Collins JPW, Davies CWH, Chen F. A cryptic diagnosis: disseminated cryptococcal disease presenting as an asymptomatic pulmonary nodule and a skin lesion: a case report. Cases J. 2008;1:430.
Cacacho A, Ashraf U, Rehmani A, Niazi M, Khaja M. Atypical radiographic presentation of Cryptococcus pneumonia in a newly diagnosed HIV patient. Case Rep Infect Dis. 2019;2019:9032958.
Chakradeo K, Paul Chia YY, Liu C, Mudge DW, De Silva J. Disseminated cryptococcosis presenting initially as lower limb cellulitis in a renal transplant recipient—a case report. BMC Nephrol. 2018;19:18.
Chen M, Wang X, Yu X, Dai C, Chen D, Yu C, et al. Pleural effusion as the initial clinical presentation in disseminated cryptococcosis and fungaemia: an unusual manifestation and a literature review. BMC Infect Dis. 2015;15:385.
Darazam IA, Rabiei MM, Moradi O, Gharehbagh FJ, Roozbeh M, Nourinia R, et al. A case of fingolimod-associated cryptococcal meningitis. Curr HIV Res. 2022;20:337–42.
Deus G, Gomez-Zorrilla S, Echeverria-Esnal D, Siverio A, Guerri-Fernandez R, Ares J, et al. Osteoarticular cryptococcosis successfully treated with high-dose liposomal amphotericin B followed by oral fluconazole. Infect Drug Resist. 2021;14:719–22.
Essouma M, Tangie LN, Temgoua MN, Kenfack UG, Ndam AN, Danwang C. Severe clinical immunodeficiency in a patient with human immunodeficiency virus infection and relatively high CD4 counts: a case report. J Med Case Rep. 2019;13:86.
Gough S, Borgetti S, Fernandes CR. Orthostatic hypotension as the initial presentation of disseminated cryptococcosis in a kidney transplant recipient. IDCases. 2022;29:e01567.
Kassi FK, Bellet V, Doumbia A, Krasteva D, Drakulovski P, Kouakou GA, et al. First case of mixed infection with Cryptococcus deuterogattii and Cryptococcus neoformans VNI in an ivorian HIV-positive patient. JMM Case Rep. 2016;3:e005037.
Kuwahara M, Yurugi S, Ando J, Takeuchi M, Miyata R, Harada M, et al. A case of cryptococcal necrotizing fasciitis and immune reconstitution inflammatory syndrome in a renal transplantation recipient. Int J Surg Case Rep. 2021;85:106199.
Liyanage DS, Pathberiya LPS, Gooneratne IK, Caldera MHPC, Perera PWS, Gamage R. Cryptococcal meningitis presenting with bilateral complete ophthalmoplegia: a case report. BMC Res Notes. 2014;7:328.
Mansoor S, Juhardeen H, Alnajjar A, Abaalkhail F, Al-Kattan W, Alsebayel M, et al. Hyponatremia as the initial presentation of cryptococcal meningitis after liver transplantation. Hepat Mon. 2015;15:e29902.
Nakajima T, Oba Y, Takashima J, Ueno K, Kikuchi A, Yamada T, et al. Cryptococcus endocarditis: a case report and review of the literature. J Infect Chemother. 2019;25:901–5.
Ni W, Huang Q, Cui J. Disseminated cryptococcosis initially presenting as cellulitis in a patient suffering from nephrotic syndrome. BMC Nephrol. 2013;14:20.
Probst C, Pongratz G, Capellino S, Szeimies RM, Schölmerich J, Fleck M, et al. Cryptococcosis mimicking cutaneous cellulitis in a patient suffering from rheumatoid arthritis: a case report. BMC Infect Dis. 2010;10:239.
Richardson TE, Lee NE, Cykowski MD, Chang SA, Powell SZ. Necrotizing fasciitis as the initial presentation of disseminated infection with fluconazole-resistant Cryptococcus neoformans. JMM Case Rep. 2014;1:e003608.
Ruan Q, Zhu Y, Chen S, Zhu L, Zhang S, Zhang W. Disseminated cryptococcosis with recurrent multiple abscesses in an immunocompetent patient: a case report and literature review. BMC Infect Dis. 2017;17:369.
Sayabovorn N, Chongtrakool P, Chayakulkeeree M. Cryptococcal fungemia and Mycobacterium haemophilum cellulitis in a patient receiving ruxolitinib: a case report and literature review. BMC Infect Dis. 2021;21:27.
Shimoda Y, Ohtomo S, Arai H, Ohtoh T, Tominaga T. Subarachnoid small vein occlusion due to inflammatory fibrosis—a possible mechanism for cerebellar infarction in cryptococcal meningoencephalitis: a case report. BMC Neurol. 2017;17:157.
Su X-H, Li W-P, Lin Q, Zheng X-J, Fang T, Jiang Y, et al. Case report: a special case of cryptococcal infection-related inflammatory syndrome in a non-HIV infected and non-transplant patient. BMC Neurol. 2022;22:247.
Thornton CS, Larios O, Grossman J, Griener TP, Vaughan S. Pulmonary Cryptococcus infections as a manifestation of idiopathic CD4 lymphocytopenia: case report and literature review. BMC Infect Dis. 2019;19:862.
Trimarchi H, Rengel T, Andrews J, Paulero M, Iotti A, Forastiero A, et al. Belatacept and mediastinal histoplasmosis in a kidney transplant patient. J Nephropathol. 2016;5:84–7.
van der Stoep JH, Sigstad E, Bredberg A. Sarcoidosis with cryptococcal infection apparently engaging only immune-privileged body compartments: a case report. BMC Infect Dis. 2020;20:440.
Vechi HT, Theodoro RC, de Oliveira AL, Gomes RM, Soares RD, Freire MG, et al. Invasive fungal infection by Cryptococcus neoformans var. grubii with bone marrow and meningeal involvement in a HIV-infected patient: a case report. BMC Infect Dis. 2019;19:220.
Wagemakers A, Ang CW, Hagen F, Bot JC, Bomers MK, Visser MC, et al. Case report: chronic relapsing cryptococcal meningitis in a patient with low mannose-binding lectin and a low naïve CD4 cell count. BMC Infect Dis. 2019;19:846.
Wappler-Guzzetta EA, Gray AL, Dagostino J, Kerstetter JC. Diffuse adrenal gland and pancreas necrosis in a patient with disseminated cryptococcosis—case report. Life (Basel). 2022;12:1667.
Wei J, Li X-Y, Zhang Y. Central nervous system cryptococcoma mimicking demyelinating disease: a case report. BMC Neurol. 2020;20:297.
Wienemann T, Müller A-K, MacKenzie C, Bielor C, Weyers V, Aktas O, et al. Cryptococcal meningoencephalitis in an IGG2-deficient patient with multiple sclerosis on FINGOLIMOD therapy for more than five years—case report. BMC Neurol. 2020;20:158.
Yao K, Qiu X, Hu H, Han Y, Zhang W, Xia R, et al. Pulmonary cryptococcosis coexisting with central type lung cancer in an immuocompetent patient: a case report and literature review. BMC Pulm Med. 2020;20:161.
Yehia BR, Eberlein M, Sisson SD, Hager DN. Disseminated cryptococcosis with meningitis, peritonitis, and cryptococcemia in a HIV-negative patient with cirrhosis: a case report. Cases J. 2009;2:170.
Zhang Y, Cooper B, Gui X, Sherer R, Cao Q. Clinical diversity of invasive cryptococcosis in AIDS patients from Central China: report of two cases with review of literature. BMC Infect Dis. 2019;19:1003.
Zhang Y, Zhang SX, Trivedi J, Toll AD, Brahmer J, Hales R, et al. Pleural fluid secondary to pulmonary cryptococcal infection: a case report and review of the literature. BMC Infect Dis. 2019;19:710.
Zhu M, Wang Q, Wang M, Wang S, Gong Q, Zhang X, et al. Rare hepatic cryptococcosis mimicked metastatic liver cancer and confirmed by metagenomic next-generation sequencing in an immunocompetent patient: a case report. Infect Drug Resist. 2022;15:5753–8.
Bicanic T, Meintjes G, Wood R, Hayes M, Rebe K, Bekker L-G, et al. Fungal burden, early fungicidal activity, and outcome in cryptococcal meningitis in antiretroviral-naive or antiretroviral-experienced patients treated with amphotericin B or fluconazole. Clin Infect Dis. 2007;45:76–80.
Brouwer AE, Rajanuwong A, Chierakul W, Griffin GE, Larsen RA, White NJ, et al. Combination antifungal therapies for HIV-associated cryptococcal meningitis: a randomised trial. Lancet. 2004;363:1764–7.
Boulware DR, Rolfes MA, Rajasingham R, von Hohenberg M, Qin Z, Taseera K, et al. Multisite validation of cryptococcal antigen lateral flow assay and quantification by laser thermal contrast. Emerg Infect Dis. 2014;20:45–53.
Dyal J, Akampurira A, Rhein J, Morawski BM, Kiggundu R, Nabeta HW, et al. Reproducibility of CSF quantitative culture methods for estimating rate of clearance in cryptococcal meningitis. Med Mycol. 2016;54:361–9.
Ye X, Gui X, Freed DC, Ku Z, Li L, Chen Y, et al. Identification of adipocyte plasma membrane-associated protein as a novel modulator of human cytomegalovirus infection. PLoS Pathog. 2019;15:e1007914.
Jarvis JN, Percival A, Bauman S, Pelfrey J, Meintjes G, Williams GN, et al. Evaluation of a novel point-of-care cryptococcal antigen test on serum, plasma, and urine from patients with HIV-associated cryptococcal meningitis. Clin Infect Dis. 2011;53:1019–23.
Percival A, Thorkildson P, Kozel TR. Monoclonal antibodies specific for immunorecessive epitopes of glucuronoxylomannan, the major capsular polysaccharide of Cryptococcus neoformans, reduce serotype bias in an immunoassay for cryptococcal antigen. Clin Vaccine Immunol. 2011;18:1292–6.
Kabanda T, Siedner MJ, Klausner JD, Muzoora C, Boulware DR. Point-of-care diagnosis and prognostication of cryptococcal meningitis with the cryptococcal antigen lateral flow assay on cerebrospinal fluid. Clin Infect Dis. 2013;58:113–6.
Wake RM, Britz E, Sriruttan C, Rukasha I, Omar T, Spencer DC, et al. High cryptococcal antigen titers in blood are predictive of subclinical cryptococcal meningitis among human immunodeficiency virus-infected patients. Clin Infect Dis. 2017;66:686–92.
Jarvis JN, Tenforde MW, Lechiile K, Milton T, Boose A, Leeme TB, et al. Evaluation of a novel semiquantitative cryptococcal antigen lateral flow assay in patients with advanced HIV disease. J Clin Microbiol. 2020;58:e00441–20.
Skipper C, Tadeo K, Martyn E, Nalintya E, Rajasingham R, Meya DB, et al. Evaluation of serum cryptococcal antigen testing using two novel semiquantitative lateral flow assays in persons with cryptococcal antigenemia. J Clin Microbiol. 2020;58:e02046–19.
Denning DW. Echinocandin antifungal drugs. Lancet. 2003;362:1142–51.
Lee Y, Puumala E, Robbins N, Cowen LE. Antifungal drug resistance: molecular mechanisms in Candida albicans and beyond. Chem Rev. 2020;121:3390–411.
Perfect JR, Dismukes WE, Dromer F, Goldman DL, Graybill JR, Hamill RJ, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the infectious Diseases Society of America. Clin Infect Dis. 2010;50:291–322.
World Health Organization. Guidelines for the diagnosis, prevention and management of cryptococcal disease in HIV-infected adults, adolescents and children 2018. https://www.who.int/publications/i/item/9789241550277. Accessed 20 Feb 2023.
Fleury M, Fonzo-Christe C, Normand C, Bonnabry P. Confusion between two amphotericin B formulations leading to a paediatric rehospitalisation. Drug Saf Case Rep. 2016;3:4.
Stone NR, Bicanic T, Salim R, Hope W. Liposomal amphotericin B (AmBisome®): a review of the pharmacokinetics, pharmacodynamics, clinical experience and future directions. Drugs. 2016;76:485–500.
Jarvis JN, Lawrence DS, Meya DB, Kagimu E, Kasibante J, Mpoza E, et al. Single-dose liposomal amphotericin B treatment for cryptococcal meningitis. N Engl J Med. 2022;386:1109–20.
Mehta P, Vinks A, Filipovich A, Vaughn G, Fearing D, Sper C, et al. High-dose weekly ambisome antifungal prophylaxis in pediatric patients undergoing hematopoietic stem cell transplantation: a pharmacokinetic study. Biol Blood Marrow Transplant. 2006;12:235–40.
Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, et al. Grade guidelines: 8. Rating the quality of evidence—indirectness. J Clin Epidemiol. 2011;64:1303–10.
World Health Organization. Guidelines for diagnosing, preventing and managing cryptococcal disease among adults, adolescents and children living with HIV 2022. https://www.who.int/publications/i/item/9789240052178. Accessed 20 Feb 2023.
Oura M, Sternberg TH, Wright ET. A new antifungal antibiotic, amphotericin B. Antibiot Annu. 1955;3:566–73.
Ellis D. Amphotericin B: spectrum and resistance. J Antimicrob Chemother. 2002;49(Suppl 1):7–10.
Gray KC, Palacios DS, Dailey I, Endo MM, Uno BE, Wilcock BC, et al. Amphotericin primarily kills yeast by simply binding ergosterol. Proc Natl Acad Sci USA. 2012;109:2234–9.
Moore CB, Sayers N, Mosquera J, Slaven J, Denning DW. Antifungal drug resistance in Aspergillus. J Infect. 2000;41:203–20.
Rex JH, Pfaller MA, Walsh TJ, Chaturvedi V, Espinel-Ingroff A, Ghannoum MA, et al. Antifungal susceptibility testing: practical aspects and current challenges. Clin Microbiol Rev. 2001;14:643–58.
Khyriem AB, Sujatha S, Parija SC. Antifungal susceptibility of Cryptococcus neoformans to amphotericin B and fluconazole. Indian J Pathol Microbiol. 2006;49:307–8.
Arechavala AI, Ochiuzzi ME, Borgnia MD, Santiso GM. Fluconazole and amphotericin B susceptibility testing of Cryptococcus neoformans: results of minimal inhibitory concentrations against 265 isolates from HIV-positive patients before and after two or more months of antifungal therapy. Rev Iberoam Micol. 2009;26:194–7.
Lozano-Chiu M, Paetznick VL, Ghannoum MA, Rex JH. Detection of resistance to amphotericin B among Cryptococcus neoformans clinical isolates: performances of three different media assessed by using E-test and National Committee for Clinical Laboratory Standards M27-A methodologies. J Clin Microbiol. 1998;36:2817–22.
Perfect JR, Cox GM. Drug resistance in Cryptococcus neoformans. Drug Resist Updates. 1999;2:259–69.
Kelly SL, Lamb DC, Taylor M, Corran AJ, Baldwin BC, Powderly WG. Resistance to amphotericin B associated with defective sterol ∆8→7 isomerase in a Cryptococcus neoformans strain from an AIDS patient. FEMS Microbiol Lett. 1994;122:39–42.
Joseph-Horne T, Loeffler RS, Hollomon DW, Kelly SL. Amphotericin B resistant isolates of Cryptococcus neoformans without alteration in sterol biosynthesis. J Med Vet Mycol. 1996;34:223–5.
Sangalli-Leite F, Scorzoni L, Mesa-Arango AC, Casas C, Herrero E, Soares Mendes Gianinni MJ, et al. Amphotericin B mediates killing in Cryptococcus neoformans through the induction of a strong oxidative burst. Microbes Infect. 2011;13:457–67.
Rodero L, Cordoba S, Cahn P, Soria M, Lucarini M, Davel G, et al. Timed-kill curves for Cryptococcus neoformans isolated from patients with AIDS. Med Mycol. 2000;38:201–7.
Córdoba S, Vivot W, Szusz W, Isla G, Davel G. Comparison of different in vitro tests to detect Cryptococcus neoformans not susceptible to amphotericin B. Mycopathologia. 2015;179:359–71.
Duschinsky R, Pleven E, Heidelberger C. The synthesis of 5-fluoropyrimidines. J Am Chem Soc. 1957;79:4559–60.
Tassel D. Treatment of candida sepsis and Cryptococcus meningitis with 5-fluorocytosine. JAMA. 1968;206:830.
Polak A, Scholer HJ. Mode of action of 5-fluorocytosine and mechanisms of resistance. Chemotherapy. 1975;21:113–30.
Vermes A. Flucytosine: a review of its pharmacology, clinical indications, pharmacokinetics, toxicity and drug interactions. J Antimicrob Chemother. 2000;46:171–9.
Pfaller MA. Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment. Am J Med. 2012;125(Suppl 1):3–13.
Chandenier J, Adou-Bryn KD, Douchet C, Sar B, Kombila M, Swinne D, et al. In vitro activity of amphotericin B, fluconazole and voriconazole against 162 Cryptococcus neoformans isolates from Africa and Cambodia. Eur J Clin Microbiol Infect Dis. 2004;23:506–8.
Schwarz P, Dromer Françoise, Lortholary O, Dannaoui E. In vitro interaction of flucytosine with conventional and new antifungals against Cryptococcus neoformans clinical isolates. Antimicrob Agents Chemother. 2003;47:3361–4.
Schwarz P, Dromer Françoise, Lortholary O, Dannaoui E. Efficacy of amphotericin B in combination with flucytosine against flucytosine-susceptible or flucytosine-resistant isolates of Cryptococcus neoformans during disseminated murine cryptococcosis. Antimicrob Agents Chemother. 2006;50:113–20.
Schwarz P, Janbon G, Dromer Françoise, Lortholary O, Dannaoui E. Combination of amphotericin B with flucytosine is active in vitro against flucytosine-resistant isolates of Cryptococcus neoformans. Antimicrob Agents Chemother. 2007;51:383–5.
Hope WW, Tabernero L, Denning DW, Anderson MJ. Molecular mechanisms of primary resistance to flucytosine in Candida albicans. Antimicrob Agents Chemother. 2004;48:4377–86.
Papon N, Noël T, Florent M, Gibot-Leclerc S, Jean D, Chastin C, et al. Molecular mechanism of flucytosine resistance in Candida lusitaniae: contribution of the FCY2, FCY1, and FUR1 genes to 5-fluorouracil and fluconazole cross-resistance. Antimicrob Agents Chemother. 2007;51:369–71.
McManus BA, Moran GP, Higgins JA, Sullivan DJ, Coleman DC. A Ser29Leu substitution in the cytosine deaminase Fca1p is responsible for clade-specific flucytosine resistance in Candida dubliniensis. Antimicrob Agents Chemother. 2009;53:4678–85.
Rhodes J, Beale MA, Vanhove M, Jarvis JN, Kannambath S, Simpson JA, et al. A population genomics approach to assessing the genetic basis of within-host microevolution underlying recurrent cryptococcal meningitis infection. G3 (Bethesda). 2017;7:1165–76.
Billmyre RB, Applen Clancey S, Li LX, Doering TL, Heitman J. 5-Fluorocytosine resistance is associated with hypermutation and alterations in capsule biosynthesis in Cryptococcus. Nat Commun. 2020;11:127.
Gusa A, Williams JD, Cho J-E, Averette AF, Sun S, Shouse EM, et al. Transposon mobilization in the human fungal pathogen Cryptococcus is mutagenic during infection and promotes drug resistance in vitro. Proc Natl Acad Sci USA. 2020;117:9973–80.
Pasko MT, Piscitelli SC, Van Slooten AD. Fluconazole: a new triazole antifungal agent. DICP. 1990;24:860–7.
Bongomin F, Oladele RO, Gago S, Moore CB, Richardson MD. A systematic review of fluconazole resistance in clinical isolates of Cryptococcus species. Mycoses. 2018;61:290–7.
Stone NRH, Rhodes J, Fisher MC, Mfinanga S, Kivuyo S, Rugemalila J, et al. Dynamic ploidy changes drive fluconazole resistance in human cryptococcal meningitis. J Clin Investig. 2019;129:999–1014.
Sionov E, Chang YC, Garraffo HM, Kwon-Chung KJ. Heteroresistance to fluconazole in Cryptococcus neoformans is intrinsic and associated with virulence. Antimicrob Agents Chemother. 2009;53:2804–15.
Sionov E, Lee H, Chang YC, Kwon-Chung KJ. Cryptococcus neoformans overcomes stress of azole drugs by formation of disomy in specific multiple chromosomes. PLoS Pathog. 2010;6:e1000848.
Sionov E, Chang YC, Kwon-Chung KJ. Azole heteroresistance in Cryptococcus neoformans: emergence of resistant clones with chromosomal disomy in the mouse brain during fluconazole treatment. Antimicrob Agents Chemother. 2013;57:5127–30.
Ngamskulrungroj P, Chang Y, Hansen B, Bugge C, Fischer E, Kwon-Chung KJ. Characterization of the chromosome 4 genes that affect fluconazole-induced disomy formation in Cryptococcus neoformans. PLoS ONE. 2012;7:e33022.
Gago S, Serrano C, Alastruey-Izquierdo A, Cuesta I, Martín-Mazuelos E, Aller AI, et al. Molecular identification, antifungal resistance and virulence of Cryptococcus neoformans and Cryptococcus deneoformans isolated in Seville, Spain. Mycoses. 2016;60:40–50.
Rodero L, Mellado E, Rodriguez AC, Salve A, Guelfand L, Cahn P, et al. G484S amino acid substitution in lanosterol 14-alpha demethylase (ERG11) is related to fluconazole resistance in a recurrent Cryptococcus neoformans clinical isolate. Antimicrob Agents Chemother. 2003;47:3653–6.
Peyton LR, Gallagher S, Hashemzadeh M. Triazole antifungals: a review. Drugs Today (Barc). 2015;51:705–18.
Jović Z, Janković SM, Ružić Zečević D, Milovanović D, Stefanović S, Folić M, et al. Clinical pharmacokinetics of second-generation triazoles for the treatment of invasive aspergillosis and candidiasis. Eur J Drug Metab Pharmacokinet. 2019;44:139–57.
Huang W, Liao G, Baker GM, Wang Y, Lau R, Paderu P, et al. Lipid flippase subunit Cdc50 mediates drug resistance and virulence in Cryptococcus neoformans. mBio. 2016;7:e00478-16.
Thompson JR, Douglas CM, Li W, Jue CK, Pramanik B, Yuan X, et al. A glucan synthase FKS1 homolog in Cryptococcus neoformans is single copy and encodes an essential function. J Bacteriol. 1999;181:444–53.
Maligie MA, Selitrennikoff CP. Cryptococcus neoformans resistance to echinocandins: (1,3)beta-glucan synthase activity is sensitive to echinocandins. Antimicrob Agents Chemother. 2005;49:2851–6.
Cao C, Wang Y, Husain S, Soteropoulos P, Xue C. A mechanosensitive channel governs lipid flippase-mediated echinocandin resistance in Cryptococcus neoformans. mBio. 2019;10:e01952–19.
Pianalto KM, Billmyre RB, Telzrow CL, Alspaugh JA. Roles for stress response and cell wall biosynthesis pathways in caspofungin tolerance in Cryptococcus neoformans. Genetics. 2019;213:213–27.
Acknowledgements
Not applicable.
Funding
This work was financially supported by the National Key Research and Development Program of China [2022YFC2303000 (L.W.), 2021YFC2300400 (L.W.)], the CAS Interdisciplinary Innovation Team (L.W.), and Henan Agricultural University [30900880 (Y.Z.), 30500946 (Y.Z.)]. The funders had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
YZ and LW conceived the review and wrote the first draft. YZ, FZ, ZL, TC, SW, YS, and ZL reviewed the case reports. YZ, LY and LZ generated the tables and figures. YZ, MC, GL, CD, YX, WL and LW reviewed and revised the updated version of the manuscript and improved the corresponding descriptions. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zhao, Y., Ye, L., Zhao, F. et al. Cryptococcus neoformans, a global threat to human health. Infect Dis Poverty 12, 20 (2023). https://doi.org/10.1186/s40249-023-01073-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40249-023-01073-4