Abstract
Background
Prisons in Madagascar are at high risk of plague outbreak. Occurrence of plague epidemic in prisons can cause significant episode of urban plague through the movement of potentially infected humans, rodents and fleas. Rodent and flea controls are essential in plague prevention, by reducing human contact with plague reservoirs and vectors. Insecticide treatment is the key step available for the control of rat fleas which transmit the disease from infected rodents to human. The implementation of an adapted flea control strategy should rely on the insecticide susceptibility status of the targeted population. For the purpose of plague prevention campaign in prisons, we conducted insecticide resistance survey on Xenopsylla cheopis, the rat flea.
Methods
Fleas were collected on rats caught in six prisons of Madagascar. They were exposed to insecticide treated filter papers and mortality was recorded following World Health Organization protocol.
Results
The fleas collected in the prisons had different resistance patterns, while a high level of resistance to insecticides tested was described in the Antanimora prison, located in the heart of Antananarivo, the capital of Madagascar.
Conclusions
This finding is alarming in the context of public health, knowing that the effectiveness of flea control could be jeopardized by insecticide resistance. In order to establish more accurate rat fleas control in prisons, the main recommendations are based on continuous monitoring insecticide susceptibility of flea, insecticide rotation, and the development of a new method for flea control.
Similar content being viewed by others
Multilingual abstracts
Please see Additional file 1 for translations of the abstract into six official working languages of the United Nations.
Background
Plague is a highly transmissible disease caused by Yersinia pestis, a zoonotic bacterium that usually infects small mammals and their fleas [1]. Humans are extremely susceptible to plague and can get the disease by infected flea bites. This is the most common way of transmission between humans and infected rodents. When bubonic plague develops into the pneumonic form, inter-human airborne transmission may take place, and causing an epidemic of primary pneumonic plague among close contact [2]. The risk of transmission is important when epizootic plague kills susceptible rodent population. Hence infected fleas are in search of a new host such as humans. Thus, vector control must be prioritized to control the transmission. Rodent control may be the second step; since killing rodents without adequate flea control can increase transmission to humans, by spreading potentially infected fleas [2, 3].
Over the past four years, Madagascar has been the country most affected by plague [4]. The latter is endemic in Madagascar, above 800 m of altitude [5]. Mahajanga, a port city located in the western part of Madagascar has also been reported as a plague focus. In rural plague foci, the black rat, Rattus rattus is admitted as the main reservoir of plague, and associated flea species are known as the plague vectors [5]. Xenopsylla cheopis, the rat flea is accepted as the main vector, mainly found inside the human habitations, harbored by R. rattus. In urban areas, R. norvegicus replaces R. rattus. The former is more resistant to plague infection. This fact can explain very few reports of human cases and no epizootic is observable, despite a high seroprevalence in these rodents [6].
Plague is a disease of poverty in Madagascar, chiefly which threatens people from poor rural settlements. Yet in urban areas, severe overpopulation, lack of sanitation and hygiene in slum areas are chief factors related to plague outbreak [4]. Bubonic is the most common form of plague encountered in Madagascar, highlighting the promiscuity between humans, rodents and fleas.
These interactions are exacerbated in detention centers in Madagascar. According to a communication from the Malagasy Prison authorities, the maximum capacity of detention centers was about 10,360, but it actually houses 20,605 detainees [7]. In all prisons in Madagascar, hygiene conditions are very poor and sanitation facilities are insufficient [8]. According to Rubini et al. in 2016, the mortality risk factors associated with plague in the European medieval city can be found today in Malagasy detention centers [9]. In addition, rats are pests of everyday life in prisons [10]. Then in a case of plague outbreak, infected fleas can spread from the prison environment to the surroundings, with rats themselves, prisoners, prison guards, and visitors [9].
Human ectoparasites such as louse, lice and bed bugs are recurrent problems in prisons [11]. Insecticide treatments have been conducted to reduce the inconvenience caused by parasites. In such environment, insecticide selection pressure could be very high among insects, due to the frequent use of insecticide. Then generalized development of resistance as a result of selection for certain genes could occur amongst insect population [12]. Insect resistance to insecticides could explain the failure of insect control in prisons. Nevertheless, insecticide treatment is the only weapon available against rat fleas. In Madagascar X. cheopis in many plague foci was found to be resistant to 12 different insecticides, with different levels of resistance depending on the study sites [13]. In a context of public health emergency and with regard to the high epidemic risk of plague encountered in detention centers in Madagascar, a campaign was launched to control rodents and fleas in many detention centers, mainly in Antanimora prison, located in the center of the capital [10]. Many prisons in the country have benefited from this preventive campaign. However, there is no data on the sensitivity of rat fleas in prisons. We will present here the results of insecticide bioassays conducted on fleas collected from six prisons of Madagascar, during the flea and rodents control conducted in 2012.
Methods
Study sites
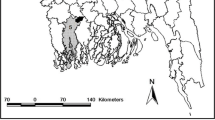
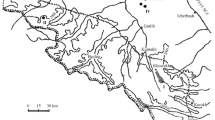
Six detention centers were investigated between May and November 2012 (Fig. 1). Two prisons are located in plague endemic area: Antanimora prison in the capital Antananarivo, and Mahajanga prison in the harbor city of the same name, located on the western part of Madagascar. The prisons of Farafangana, Toliary, Manajary and Morombe are located outside plague endemic areas. No case of plague has been declared from the detention centers of Madagascar.
Rodent trapping and flea collection
Rodents were trapped alive with wire mesh BTS traps (Besancon Technical Service, Besancon, France) settled in the evening and left overnight. Traps were baited with onions and dried fish during three consecutive nights. Fleas were combed from their rodents host and transported to the laboratory in a jar containing rice bran, covered with fine mesh tissue. Fleas were morphologically identified to species and reared in insectarium [14] until the required numbers of individuals for insecticide bioassays were obtained.
Insecticide bioassays
Insecticide bioassays were conducted according to World Health Organization (WHO) protocol [15]. Laboratory bioassays conditions were described in Miarinjara et al., 2016 [13]. Briefly, fleas were exposed to strips of insecticide impregnated paper during diagnostic time. For each insecticide test, impregnated paper provided by WHO were used (Vector Control Research Unit, Penang, Malaysia). Four replicates of ten fleas per tube were exposed to impregnated papers and negative controls consisted in two replicates exposed to papers treated with insecticide solvent only. We tested insecticide belonging to the families used in insect pest control, namely pyrethroids (0.05% deltamethrin, 0.05% lambdacyhalothrin, 0.15% cyfluthrin, 0.75% permethrin, 0.025% alphacypermethrin and 0.5% etofenprox), carbamates (0.1% bendiocarb and 0.1% propoxur), organochlorine (4% DDT and 4% dieldrin) and organophosphates (5% malathion and 1% fenitrothion). Numbers of dead and paralyzed fleas were counted during exposure time. Final mortality was recorded after 24 h and compared with ANOVA tests. In addition, for each insecticide tested mean mortality of fleas from prisons was compared (ANOVA test) with that recorded in some localities from previous studies (black points on Fig. 1) [13]. The analysis was performed with R software (R Version 3.1.1 2014) and RStudio environment (R Studio Version 0.98.976 2009–2013).
Results
Fleas from detention centers investigated were resistant to the large majority of tested insecticides (alphacypermethrin, lambdacyhalothrin, etofenprox, deltamethrin, DDT, propoxur and bendiocarb). Highest mortality rates were obtained with dieldrin (95.9% ± 10.1), permethrin (83.7% ± 27.8), fenitrothion (81.6% ± 25.4), cyfluthrin (81.25% ± 19.4) and malathion (75.4% ± 25.7) (Fig. 2). Different profiles of susceptibility were observed according to prison and tested insecticides (Fig. 3). Notwithstanding the fleas from Antanimora, the main prison, all tested flea populations were susceptible to dieldrin, which is still the most efficient, even banned from many countries worldwide. Permethrin was still efficient in 4/6 prisons, while some populations resistant to permethrin were still susceptible to fenitrothion. Lowest mortality rates were observed in Antanimora prison, where fleas were resistant to all tested insecticides. Fleas from Morombe prison were still susceptible to at least four insecticides.
Box-and-whisker plot of mortality rate after 24 h for each insecticide for all prison populations. Diamond-shaped points inside the boxes are mean values. Horizontal bars in boxes are the 50th percentiles (medians), and the bottom and the top of the box represent the 25th and the 75th percentiles, respectively. The two limits of vertical lines above and at the bottom of the box are the whiskers and represent the maximum and the minimum values of the data. Points outside the limit of vertical line are “outlier”, which are values outside 95% the confidence interval
By comparing flea mortalities recorded in prisons and those recorded in plague surveillance areas (non-prison) where plague occurred or have been suspected, no significant difference in mortalities exists between prison and non-prison flea population (Fig. 4). The average mortality profile was the same for all tested insecticides.
Discussions
This study is the first assessing the susceptibility of rat fleas in prisons. In this study, X. cheopis populations from Malagasy prisons were resistant to at least seven insecticides out of twelve. However resistance profile was very different from a prison to another. Few insecticides were still working (dieldrin, permetrin, cyfluthrin and fenitrothion) depending on prison. These insecticides may be recommended for the future intervention against fleas, vector of Y. pestis. Still, fleas from Antanimora prison, located in the capital showed high rate of resistance to insecticides, when compared with the other prisons. This population may have been subjected to higher insecticide pressure [12]. As this detention center is located in the capital, it can be obvious that insecticide products are more available. On the other hand, fleas from prisons located far away from the capital (Morombe and Toliary), showed less resistance to at least four insecticides (dieldrin, permethrin, cyfluthrin and fenitrothion).
Compared to previous results obtained with eight populations tested [13], the same profile of resistance was observed in fleas from prisons. However, it was assumed that insecticide selection pressure may be higher in prison due to frequent insecticide treatment against ectoparasites [16, 17]. Still, these results suggested that insecticide treatment in prisons is not as frequent as supposed and might not induce higher selection pressure on rat fleas. In large and overcrowded places of detention, external actors (International and Non-Gouvernemental Organizations) can as well support by organizing regular or punctual vector control campaigns.
As public health recommendations, policy of rat’s flea control and insecticide resistance management must be held in place. This policy may take in account, first, the results from insecticide bioassays. Each decision on the use of insecticide may be linked to data on the susceptibility of the targeted population. Hence it is important to make continuous surveillance of insecticide susceptibility of fleas from prisons, and gather many data as possible in other prisons.
Second, it is admitted that frequent use of the same insecticide leads to the establishment of resistant population [16, 17]. Experience from malaria vectors showed that the rotational use of insecticide of different modes of action could be done in order to keep their efficiency [12]. In this study, besides the case of Antanimora prison, alternating the use of pyrethroid (permetrin) and organophosphate (fenitrothion) can be recommended.
Third, WHO recommended the use of insecticide dust belonging to three chemical types (carbamate, organophosphate and pyrethroid) to fight against rat’ fleas [18]. For highly insecticide resistant population, such as fleas from Antanimora, other approaches on flea control must be taken into account. Systemic insecticide might be a promising alternative to target on host fleas [19,20,21]. Systemic insecticides are toxic for fleas when ingested by rodents, which make them more accurate on targeting rodents’ fleas than insecticide dusting. Fipronil, a phenylpyrazole insecticide, is considered as a good candidate in controlling rodent’s flea [21]. Besides, its chemical structure is different from insecticide recommended for flea control in plague foci, then can be efficient in localities where cross resistance is suspected among vectors. Besides, its systemic action is significant at low concentration [19]. Efficiency of systemic insecticides was tested on laboratory and feasibility on field was evaluated [19, 22,23,24,25,26]. This method relies on the palatability and the attractiveness of the bait toward the targeted rodent. So as to reduce rodents and fleas populations at the same time, systemic insecticide can be combined with slow-acting rodenticide [20].
Finally, as for improvement of resistance surveillance, knowing the insecticide resistance mechanism can accurately help finding out the most adapted insecticide for each scenario. However, hitherto very few details are known about insecticide resistance mechanisms involved for X. cheopis.
Conclusions
Resistance to insecticides could be a serious challenge in detention centers. Finding an effective insecticide product is crucial in an emergency context to deal with a potential epidemic occurring in prisons. Our results suggest that insecticide treatment of rat fleas differ from one prison to another. The study of the mechanisms involved in resistance of fleas to insecticide can be carried out and alternative vector control policies should be considered. In addition, prisoner intoxication is a concern and must be taken into account in the choice of product and method to be adopted. However, improving the living conditions of inmates is a key factor in reducing contact with rodents and their fleas.
References
Gage KL, Kosoy MY. Natural history of plague: perspectives from more than a century of research. Annu Rev Entomol. 2005;50:505–28. doi:10.1146/annurev.ento.50.071803.130337.
Barnes A, Kartman L. Control of plague vectors on diurnal rodents in the sierra Nevada of California by use of insecticide bait-boxes. J Hyg (Lond) 1960;58: 347–355. Available: http://journals.cambridge.org/abstract_S0022172400038468. Accessed 19 May 2017.
Gratz NG. Control of plague transmission. In: Plague manual. Epidemiology, distribution, surveillance and control. Geneva, Switzerland: World Health Organization; 1999. p. 97–134.
Bertherat EG. Plague in Madagascar: overview of the 2014–2015 epidemic season Weekly epidemiological record/Health Section of the Secretariat of the League of Nations 90.20 (2015): 250–252. Available: http://apps.who.int/iris/bitstream/10665/242357/1/WER9020_250-252.PDF. Accessed 19 May 2017.
Andrianaivoarimanana V, Kreppel K, Elissa N. Duplantier J-MM, Carniel E, Rajerison M, et al. understanding the persistence of plague foci in Madagascar. PLoS Negl Trop Dis. 2013;7:e2382. doi:10.1371/journal.pntd.0002382.
Duplantier J-M, Duchemin J-B, Chanteau S, Carniel E. From the recent lessons of the Malagasy foci towards a global understanding of the factors involved in plague reemergence. Vet Res. 2005;36:437–53. doi:10.1051/vetres:2005007.
International Committee of the Red Cross. Regards sur la détention à Madagascar: Biogaz, nutrition et santé: facteurs d’amélioration des conditions carcérales. Public Conference. Antananarivo, Madagascar; 2017. Available: http://www.newsmada.com/2017/02/14/conditions-carcerales-le-cicr-tire-la-sonnette-dalarme/. Accessed 19 May 2017.
U.S. Departement of Sate. Madagascar 2014 Human rights report [internet]. Madagascar; 2011. Available: https://www.state.gov/documents/organization/236588.pdf. Accessed 19 May 2017.
Rubini M, Gualdi-Russo E, Manzon VSV, Rinaldo N, Bianucci R. Mortality risk factors show similar trends in modern and historic populations exposed to plague. J Infect Dev Ctries. 2016;10:488–93. doi:10.3855/jidc.7974.
International Committee of the Red Cross. Madagascar: Preventing plague in prisons [Internet]. 2013. Available: http://www.icrcvideonewsroom.org/open.asp?id=141. Accessed 19 May 2017.
Clouzeau D. Bilan Sanitaire Globalde la Prison de Mahajanga en Vue d’Améliorer la Qualité de Vie en Détention. [Internet]. Madagascar; 1999. Available: http://onlinepdfcatalog.com/t/takelaka.dts.mg1.html. Accessed 19 May 2017.
Nauen R. Insecticide resistance in disease vectors of public health importance. Pest Manag Sci. John Wiley & Sons, Ltd. 2007;633:628–633. doi:10.1002/ps.1406.
Miarinjara A, Boyer S. Current perspectives on plague vector control in Madagascar: susceptibility status of Xenopsylla cheopis to 12 insecticides. PLoS Negl Trop Dis. 2016;10:e0004414. doi:10.1371/journal.pntd.0004414.
Boyer S, Miarinjara A, Elissa N. Xenopsylla cheopis (Siphonaptera: Pulicidae) susceptibility to deltamethrin in Madagascar. PLoS One. 2014;9:e111998. doi:10.1371/journal.pone.0111998.
OMS. Resistance aux insecticides et lutte antivectorielle, Dix-septième rapport du Comité OMS d’experts des Insecticides. In: Série de raports techniques N°443; 1970.
Corbel V, N’Guessan R, Brengues C, Chandre F, Djogbenou L, Martin T, et al. Multiple insecticide resistance mechanisms in Anopheles gambiae and Culex quinquefasciatus from Benin, West Africa. Acta Trop. 2007;101:207–16. doi:10.1016/j.actatropica.2007.01.005.
Corbel V, Hougard J-M, N’Guessan R, Chandre F. Evidence for selection of insecticide resistance due to insensitive acetylcholinesterase by carbamate-treated nets in Anopheles gambiae s.S. (Diptera: Culicidae) from Côte d’Ivoire. J Med Entomol. 2003;40:985–8. doi:10.1016/j.actatropica.2007.01.005.
World Health Organization. Pesticides and their application for the control of vectors and pets of public health importance [Internet]. Sixth edit. C.F. Curtis (London School of Hygiène and Tropical Medecine) Geneva, Switzerland: Department of Control of Neglected Tropical Diseases WHO Pesticide evaluation scheme (WHOPES); 2006. Available: http://apps.who.int/iris/handle/10665/69223
Leirs H, Larsen KS, Lodal J. Palatability and toxicity of fipronil as a systemic insecticide in a bromadiolone rodenticide bait for rat and flea control. Med Vet Entomol. 2001;15:299–303. doi:10.1046/j.0269-283x.2001.00302.x.
Rood MP. Poché RM. effect of a feed-through insecticide (imidacloprid) on the flea index of a Norway rat (Rattus norvegicus) focus in Los Angeles, California. In: Proc 24th Vertebr Pest Conf; 2010.
Rajohnson D, Miarinjara A, Rahelinirina S, Boyer S. Effectiveness of fipronil as a systemic control agent against Xenopsylla cheopis (Siphonaptera: Pulicidae) in Madagascar. J Med Entomol. 2017:1–7. doi:10.1093/jme/tjw200.
Mbise TJ, Bennington E, Brooks JE, Jackson WB, Bushland RC, Drummond RO, et al. Control of rodent fleas using systemic insecticides. Int J Trop Insect Sci1994;15: 235–239. doi:10.1017/S1742758400015514.
Jachowski DS, Brown NL, Wehtje M, Tripp DW, Millspaugh JJ, Gompper ME. Mitigating plague risk in utah prairie dogs: evaluation of a systemic flea-control product. Wildl Soc Bull. 2012;36:167–75. doi:10.1002/wsb.107
Miller BE, Bennett WC. Graves GN. Gregory JP. A study of sylvatic plague and evaluation of systemic insecticides for the control of wild rodents and their fleas. U.S. Army Medical Research and Development Command: Wheeler JR; 1972.
Clark PH, Cole MM. Systemic insecticides for control of oriental rat fleas: bait tests with hooded white rats. J Econ Entomol. 1968;61:505–8. Available: http://www.ncbi.nlm.nih.gov/pubmed/5642109\nhttp://www.ncbi.nlm.nih.gov/pubmed/5642117
Clark P, Cole M. Systemic insecticides for control of oriental rat fleas: preliminary tests in Guinea pigs, 1967–69. J Econ Entomol. 1971;64(6):1477–9.
Acknowledgments
The rodent control campaign undertook in detention centers mentioned in this study was made possible thanks to the collaboration of the International Committee of Red Cross (ICRC), the Prison Service (Direction Générale de l’Administration Pénitentiaire), the “Brigade anti-Rat” of the Public Health Ministry of Health, the Swiss Embassy and the staff of the Unité Peste of the Institut Pasteur de Madagascar (IPM). We thank the technicians of Unité Peste and Unité d’Entomologie Medicale (IPM) for collecting and rearing fleas used in this study. We are grateful to the staffs of ICRC, particularly M. Jean-Marcel Kavaruganda, one of the initiator of this project, and the Prisons Staffs, particularly the Direction Générale de l’Administration Pénitentiaire, for their great collaboration.
Funding
The fleas tested here were collected during rodent control campaign funded by the Swiss Embassy-Antananarivo (Ref # 771.22.00–1-MAE/RML). Laboratory works were funded by Institut Pasteur de Madagascar.
Availability of data and materials
Data supporting the results in this publication are available in the following published and full access articles:
Boyer, S., A. Miarinjara, and N. Elissa. 2014. Xenopsylla cheopis (Siphonaptera: Pulicidae) Susceptibility to Deltamethrin in Madagascar. PLoS One. 9: e111998.
Miarinjara, A., and S. Boyer. 2016. Current Perspectives on Plague Vector Control in Madagascar: Susceptibility Status of Xenopsylla cheopis to 12 Insecticides. PLoS Negl. Trop. Dis. 10: e0004414.
Author information
Authors and Affiliations
Contributions
AM is major contributor in writing the manuscript, performed laboratory bioassays and data analysis. JV contributed in writing the manuscript and collaborated in data acquisition. JMK initiated the project and all the collaborations between entities implicated, and contributed to manuscript writing. MR is the principal investigator of the project and contributed in writing the manuscript. SB contributed to data analysis and manuscript writing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This research in Madagascar was systematically made possible thanks to extant conventions between the Institut Pasteur de Madagascar, the International Committee of the Red Cross and the local government represented by the Direction Générale de l’Administration Pénitentiaire. The rats were caught either during sanitation campaign as request of the Ministry of Health and the ICRC on the basis of a National Health Priority, or during an investigative plague mission. In this last case, a letter was sent to the local authority, local general inspector, and to the national authority of the Ministry of Health, to explain the main objective of the field mission. The mission orders were authorized by the local sanitary authority, the CSB (Centre de Santé de Base), SSD (Service de Santé de District) and Fokotany (smallest public authority in Madagascar) authorities. Additional authorizations were not required because Rattus species on which fleas were caught are considered to be pest species and have no protected status (see IUCN and CITES lists). Rats were caught alive in wire-mesh and Sherman traps. All animals were killed by cervical dislocation. All members of the IPM involved in the fieldwork have been trained to handle and kill rodents. The study was conducted in accordance with the guidelines (http://campus.pasteur.mg/a-savoir/charte-dethique-de-linstitut-pasteur/) adapted to wild rodents. Animals were treated in a humane manner, and in accordance with guidelines of the American Society of Mammalogists.
Young mice used to feed flea population were from the Institut Pasteur de Madagascar animal breeding facility. They were not purchased or donated, but were bred for this purpose.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional file
Additional file 1:
Multilingual abstracts in the six official working languages of the United Nations. (PDF 622 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Miarinjara, A., Vergain, J., Kavaruganda, J.M. et al. Plague risk in vulnerable community: assessment of Xenopsylla cheopis susceptibility to insecticides in Malagasy prisons. Infect Dis Poverty 6, 141 (2017). https://doi.org/10.1186/s40249-017-0356-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40249-017-0356-5