Abstract
Crude methanol extract (ME) of broccoli (Brassica oleracea L.) sprout was fractioned by hexane, chloroform, ethyl acetate, butanol, and water. The contents of total polyphenols (19.89 mg GAE/g) and flavonoids (10.06 mg RE/g) were significantly higher in the butanol fraction (BF) than in the other fractions. The BF showed the highest DPPH (EC50 = 0.524 mg/mL) and ABTS (EC50 = 0.180 mg/mL) radical scavenging activities. High-performance liquid chromatography (HPLC) of crude ME showed that the most abundant phenolic compounds were rutin, quercetin, chlorogenic acid, catechin, and p-coumaric acid. The contents of quercetin, chlorogenic acid and p-coumaric acid were higher in the ethyl acetate fraction (EF) and BF than in the other fractions. Antioxidant activity and phenolic compound contents were correlated, suggesting that phenolics were responsible for the antioxidant activity. The hexane fraction (HF) and chloroform fraction (CF) decreased the viability of breast cancer stem cells (BCSCs), and the CF had the highest antiproliferative activity (IC50 = 69.47 mg/mL). The CF also suppressed the stemness characteristics of BCSCs and induced apoptotic cell death. The most abundant characteristic peak in CF was identified as oleic acid (area = 35.05%) by gas chromatography-mass spectrometry (GC–MS). Therefore, the broccoli sprout BF contained high levels of phenolic compounds that contributed to its antioxidant activity, and CF had a marked anti-proliferative effect on BCSCs.
Similar content being viewed by others
Introduction
To improve life expectancy and enhance health, the consumption of vegetables and fruits is important [1]. The constituents of vegetables, especially cruciferous and dark green leafy vegetables, can prevent diseases including cancer and cardiovascular conditions [2, 3]. Cruciferous vegetables are rich in beneficial bioactive compounds such as glucosinolates, anthocyanins, coumarins, carotenoids, and kaempferol, among other minor compounds [3]. Broccoli (Brassica oleracea L.) sprout is a cruciferous vegetable rich in antioxidant vitamins and phenolic compounds, and thus have high antioxidant and anti-inflammatory properties, and contains significantly more glucoraphanin and indolic glucosinolates than broccoli florets [4,5,6]. In addition, several studies have investigated the anticancer effect of broccoli sprout extract in skin, prostate, and colorectal cancers [7, 8]. However, few studies have investigated the effect of broccoli sprout extracts on breast cancer stem cells (BCSCs).
Breast cancer is the most common cancer among women worldwide, and is expected to increase in prevalence by more than 46% by 2040 [9]. Breast cancer is initiated by a subpopulation of cells with the characteristics of stem cells [10]. Because cancer stem cells have the ability to self-renew, invade, and migrate, they are resistant to chemotherapy and radiotherapy, and may be the main cause of breast cancer treatment failure [11]. Multidrug resistance associated protein 1 (ABCC1/MRP1) and P glycoprotein/multidrug resistance protein 1 (ABCB1/MDR1), which induce drug resistance by mediating intracellular drug release, are overexpressed in cancer stem cells, and ALDH-positive and CD44+/CD24− are phenotypes found in BCSCs and are closely associated with poor prognosis [12]. Therefore, eliminating the BCSC population by targeting these markers could improve the effectiveness of current treatment strategies.
The contents and activities of biological substances vary depending on the extraction conditions, including the solvent used [13]. In general, after obtaining a crude methanol extract (ME) capable of extracting both hydrophilic and hydrophobic components, the solvent selected in the order of polarity from n-hexane with the lowest polarity to water with the highest polarity is added [14]. In this study, we evaluated the antioxidant and antiproliferative activities of solvent fractions prepared in a stepwise manner from the crude ME of broccoli sprout, and analyzed the major phenolic compounds in the solvent fractions. In addition, for the first time we compared the antiproliferative and stemness-inhibitory effects of solvent fractions from the crude ME of broccoli sprout on BCSCs.
Materials and methods
Samples and chemicals
Hot air-dried broccoli sprout powder was purchased from Damaonherb Co. (Yeongcheon, South Korea). The 11 standard compounds for high-performance liquid chromatography (HPLC) such as gallic acid, caffeic acid, p-coumaric acid, chlorogenic acid, 4-hydroxybenzoic acid, sinapic acid, catechin hydrate, rutin hydrate, myricetin, quercetin, and epicatechin and the HPLC-grade solvents such as methanol and acetic acid were purchased from Sigma Chemical Co. (St. Louis, MO, USA).
Solvent fractionation
Broccoli sprout powder (100 g) was extracted with 2 L of 80% methanol, sonicated three times, and filtered using Whatman No. 2 filter paper. The extract was evaporated at 40 °C in a vacuum rotary evaporator and lyophilized to generate crude ME. Dried ME powder (25 g) was suspended in 500 mL of distilled water and fractionated by n-hexane, chloroform, ethyl acetate, n-butanol, and water (1:1) in a stepwise manner. Each fraction was evaporated at 40 °C and lyophilized to generate the hexane fraction (HF), chloroform fraction (CF), ethyl acetate fraction (EF), butanol fraction (BF), and residual water fraction (WF). Each extract was dissolved in dimethyl sulfoxide (DMSO) to 200 mg/mL for use in subsequent experiments.
Measurement of total polyphenol content
Distilled water (1.375 mL) and the fractions (125 μL) were added to 500 μL of Folin–Ciocalteu phenol reagent and incubated for 3 min. Then, 10% Na2CO3 (1 mL) was added, and the mixture was allowed to react in the dark for 30 min. The absorbance at 700 nm was measured using a microplate reader. Results are expressed as gallic acid equivalent (mg GAE/g).
Measurement of total flavonoid content
Distilled water (80 μL) was added to 40 μL of the fractions and 6 μL of 5% NaNO2. After incubation for 5 min, 10% AlCl3 (12 μL) was added and the mixture was incubated for 6 min. NaOH (40 μL; 1 N) and distilled water (42 μL) were then added, and the absorbance at 510 nm was determined using a microplate reader. Results are expressed as rutin equivalent (mg RE/g).
DPPH radical scavenging assay
DPPH radical scavenging activity was assayed as described previously, with some modifications [15, 16]. Each fraction in DMSO (40 µL) was added to 160 μL of 200 μM DPPH radical solution and incubated at 37 °C for 30 min. Scavenging activity was quantified using catechin as the positive control. EC50 values were calculated using Prism 7.0 software (GraphPad Software, Inc., La Jolla, CA, USA).
ABTS radical scavenging activity
ABTS radical scavenging activity was assayed as described previously, with some modifications [17]. ABTS solution (900 µL) was added to 100 µL of extract in DMSO and incubated for 2 min, and absorbance at 734 nm was measured using a UV1800 spectrophotometer (Shimadzu, Kyoto, Japan). Scavenging activity was calculated using α-tocopherol as the positive control.
Alkyl radical scavenging activity based on electron spin resonance
Alkyl radical scavenging activity was assayed as described previously [15] using a JES-FA200 electron spin resonance (ESR) spectrometer (JEOL, Tokyo, Japan). Signal intensities were compared using a magnetic ESR standard (Mn2+ marker), and results are expressed as relative height ratios. Catechin was used as the positive control.
High-performance liquid chromatography
Phenolic compounds were quantitatively analyzed using a slightly modified method [18], HPLC-ultraviolet detector (HPLC-UVD) (CBM-20A; Shimadzu, Tokyo, Japan) and Shim-pack ODS 5 μm column (Shimadzu). The mobile phase consisted of solvent A: water: methanol: acetic acid (95%, 2.5%, 2.5%; v/v/v), and solvent B: water: methanol; acetic acid (5%, 92.5%, 2.5%; v/v/v). The binary gradient was as follows: 0 min (100% A; 0% B), 10 min (90% A; 10% B), 48 min (20% A; 80% B), and 58 min (100% A; % B). The column and flow rate were maintained at 30 °C and 1.0 mL/min, respectively, and the injection volume and detection wavelength were 20 μL and 280 nm, respectively. The concentrations of phenolic compounds were calculated using a standard curve in triplicate.
Cell culture
Human breast cancer MCF-7 cells were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). The BCSC line MCF-7/SCs were sorted from MCF-7 cells based on CD44+ and CD24−, and characterized as described previously [19]. MCF-7 cells and MCF-7/SCs were cultured in Dulbecco’s modified Eagle’s medium (DMEM) and Roswell Park Memorial Institute (RPMI) medium containing 10% heat-activated fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were maintained at 37 °C in an atmosphere containing 5% CO2.
Cell viability assay
MCF-7 cells and MCF-7/SCs were seeded (5000/well) in 96-well plates and incubated for 24 h. Cells were treated with the fractions for 24 h and exposed to 100 μL of 1 mg/mL 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) at 37 °C for 2 h. Next, 150 μL of DMSO was added to each well to solubilize formazan, and the plates were shaken for 30 min in the dark. Absorbance at 570 nm was evaluated using a microplate reader. IC50 values were calculated using GraphPad Prism 7.0 software.
Colony formation assay
MCF-7/SCs (400/mL) were seeded for 24 h and treated with CF for 10 days. Colonies were washed twice with PBS, fixed with 4% paraformaldehyde, and stained with 2% crystal violet for 30 min.
Flow cytometry
Flow cytometry was performed using a FACSCalibur flow cytometer (BD Biosciences, Franklin, NJ, USA), as described previously [20]. Identical numbers of cells (1 × 105/dish) were seeded for 24 h and treated with the CF for 24 h. To assay ALDH activity, the ALDEFLUOR Assay Kit (Stem Cell Technologies, Vancouver, BC, Canada) was used according to the manufacturer’s instructions. Diethylaminobenzaldehyde (DEAB), an inhibitor of ALDH, was used as the negative control. PE-conjugated anti-human CD24 and FITC-conjugated anti-human CD44 antibodies (BD Pharmingen, San Diego, CA, USA) were added to 100 μL of immunofluorescence staining buffer and incubated for 10 min at 4 °C. The cells were washed with PBS and the CD44+/CD24− cell population was analyzed. To identify apoptotic cells, the Annexin V-FITC Apoptosis Detection Kit (BD Pharmingen, San Diego, CA, USA) was used following the supplier’s instructions. Briefly, cells were suspended in annexin V-FITC (1:20 dilution in 1 × binding buffer) and propidium (PI; 1:50) and analyzed within 30 min.
Western blot assay
MCF-7/SCs were prepared at 4 × 105 in 100 mm dishes. After incubation for 24 h and treatment with the solvent fractions, cells were lysed with RIPA lysis buffer and protein concentrations in the lysates were quantified using a BCA Protein Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA). The lysates were resolved by sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Except for β-actin (1:10,000), the primary antibodies were used at 1:1000 dilutions, and secondary antibodies (anti-rabbit and -mouse IgG) were used at 1:5000 dilutions. Protein bands were developed using the ECL Plus Kit (Biosesang, Seongnam, South Korea).
Wound healing assay
MCF-7/SCs (2 × 105/mL) were seeded in six-well plates until 95% confluence, and a scratch wound was made in each well using a sterile pipette tip. Next, the cells were treated with the CF and incubated. Wounds were visualized using an inverted phase-contrast microscope at 4 × magnification.
Invasion assay
The upper chamber of 24-well Transwell plates (Corning, Corning, NY, USA) was filled with Matrigel and normal culture medium. After the gel had solidified, each well was loaded with 2.5 × 105 cells in serum-free medium, with or without CF, and the lower chamber was loaded with culture medium. After 24 h, the cells were fixed with 4% paraformaldehyde and stained with 2% crystal violet. The cells were observed under a phase-contrast microscope.
Hoechst 33342 staining
MCF-7/SCs were seeded into wells (1 × 104/well) and incubated for 24 h. Next, cells were treated with the CF for 24 h, stained with Hoechst 33,342 solution (10 μg/mL) for 10 min in the dark, and visualized using a fluorescence microscope (× 100) (IX73; Olympus Corporation, Tokyo, Japan).
Gas chromatography-mass spectrometry
Gas chromatography-mass spectrometry (GC–MS) of CF was carried out using the Shimadzu GCMS-QP-2010 Plus instrument in the Bio-Health Materials Core-Facility of Jeju National University, with a DB-5MS GC column (30 m length, 0.25 mm internal diameter, 0.25 µm film thickness). The injection volume of 1 µL (100 µg/mL dissolved in methanol) was delivered in splitless mode. Helium was used as the carrier gas at a constant flow rate of 1 mL min−1. The temperature ranged from 80 to 300 °C (80 °C hold for 5 min; 80–280 °C at 5 °C/min for 10 min; and 280–300 °C at 10 °C/min for 10 min). The total run time was 67 min, and mass spectra were detected using W9N08 Wiley library ver. 9.0 at a similarity cut-off of 90%.
Statistical analysis
Groups were compared using GraphPad Prism 7.0 software, Student’s t-test, and one-way analysis of variance (ANOVA) with Tukey’s multiple comparison test. Data are expressed as means ± standard deviation (SD) of three replicates and statistical significance was set at P < 0.05. Pearson’s rank correlation was performed using the ‘correlation’ tool in Prism 7.0 software.
Results and discussion
Total polyphenol and flavonoid contents
Phenolic compounds, including flavonoids, have various biological and pharmacological activities and are present in many vegetables and fruits [21]. As shown in Table 1, the butanol fraction (BF) had a higher polyphenol content (19.89 mg GAE/g) than the other fractions, followed by the ethyl acetate fraction (EF) (7.94 mg GAE/g). The hexane fraction (HF) had the lowest polyphenol content (2.32 mg GAE/g). The BF had the highest total flavonoid content (10.06 mg RE/g), followed by the chloroform fraction (CF) (4.71 mg RE/g), HF (4.22 mg RE/g), EF (1.59 mg RE/g), and residual water fraction (WF) (0.66 mg RE/g). Overall, these results indicate that the BF is rich in antioxidants such as polyphenols and flavonoids.
Antioxidant activity
Oxidative stress caused by free radicals, such as superoxide (O2∙−, OOH∙) and hydroxyl (OH∙) radicals, triggers the progression of several senescence-related diseases. Antioxidants interact with and scavenge free radicals, thereby reducing cellular damage [22]. As shown in (Table 2), the BF had the lowest EC50 values in DPPH∙ (0.524 mg/mL) and ABTS∙ (0.180 mg/mL) radical scavenging assays. For ESR-alkyl∙ radical scavenging activity, the WF had the lowest EC50 value (0.0.31 mg/mL). The HF showed the highest EC50 value in DPPH∙ (18.366 mg/mL), ABTS∙ (11.528 mg/mL) and ESR-alkyl∙ (3.477 mg/mL) radical scavenging assays. Overall, the HF and CF had high EC50 values and low radical scavenging activities, whereas BF had the lowest EC50 value and exhibited marked antioxidant activity.
HPLC analysis of phenolic compounds
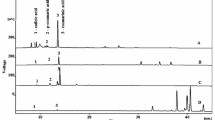
Phenolic compounds reportedly have anticancer and anti-inflammatory effects [23]. Therefore, we used High-performance liquid chromatography (HPLC) to analyze the contents of 11 representative phenolic compounds (gallic acid, catechin, 4-hydroxybenzoic acid, chlorogenic acid, caffeic acid, epicatechin, p-coumaric acid, sinapic acid, rutin, myricetin, and quercetin) in the crude methanol extract (ME). As shown in Fig. 1, the most abundant phenolic compounds in the crude ME were rutin, quercetin, chlorogenic acid, catechin, and p-coumaric acid, followed by 4-hydroxybenzoic acid, sinapic acid, epicatechin, and caffeic acid. The contents of rutin, quercetin, chlorogenic acid, catechin and p-coumaric acid, which were present at high levels in the crude ME, were markedly higher in the EF and BF than in the other fractions. The BF, which had greater antioxidant activity than the other fractions, also had the highest contents of gallic acid, catechin, 4-hydroxybenzoic acid, chlorogenic acid, caffeic acid, epicatechin, rutin, myricetin, and quercetin (Table 3). The chlorogenic acid content of BF was 6.35-, 10.54-, 14.76-, 93.66-, 126.38- fold that of EF, CF, HF, and WF, respectively. The rutin content was 7.48-fold that of the EF and was not detected in the HF, CF, or WF. The quercetin content was 2.56- and 5.08-fold that of the EF and WF, respectively, and was not detected in the HF or CF. The p-coumaric acid content was highest in the EF, followed by the BF and CF, and was not detected in the HF or WF. The sinapic acid content in the EF was 1.87-, 70.09-, and 79.78-fold that of the BF, CF, and WF, respectively. Therefore, the phenolic contents of the fractions differed significantly depending on the type of solvent, and that of the BF was highest.
Correlation of phenolics with antioxidant activities
Pearson product-moment correlation analysis showed a strong positive relationship between TPC and the DPPH∙ (R2 = 0.9244), ABTS∙ (R2 = 0.9399), and ESR-alkyl∙ radical scavenging (R2 = 0.9398) activities (Table 4). In contrast, there was a weak positive correlation between TFC and the DPPH∙ (R2 = 0.6244), ABTS∙ (R2 = 0.4159), and ESR-alkyl∙ radical scavenging (R2 = 0.445) activities. In general, antioxidant activity is closely related to phenolic compounds, and significant correlations between total phenolics and antioxidant activity have been reported [16]. Our results suggest that the antioxidant activity of the fractions is mediated by TPC rather than TFC.
As shown in Table 4, there were significant correlations between the individual phenolic compounds and antioxidant activities. Specifically, strong correlations were observed between DPPH∙ radical scavenging activity and chlorogenic acid, epicatechin, rutin, and myricetin (R2 = 0.8978, 0.9376, 0.9078, and 0.9082 respectively; P < 0.01). Strong correlations were found between ABTS∙ radical scavenging activity and caffeic acid, epicatechin, myricetin, quercetin (R2 = 0.9666, 0.9692, 0.9183, and 0.9329, respectively; P < 0.01). The correlations between ESR-alkyl∙ radical scavenging activity and caffeic acid, epicatechin, myricetin, quercetin had R2 values of 0.979, 0.9639, 0.9325, and 0.9626, respectively (P < 0.001). These results suggest that the antioxidant activity of BF is mediated by TPC, rather than TFC; correspondingly, the DPPH∙ and ABTS∙ radical scavenging activities of the BF are significantly higher than the other fractions.
Effects of solvent fractions on the viability of breast cancer stem cells
Cancer stem cells have self-renewal ability and their invasion, migration, and resistance to chemotherapy and radiotherapy leads to a poor prognosis [24]. Because the main cause of breast cancer treatment failure is breast cancer stem cell (BCSC)s [11], we evaluated the cytotoxicity of the fractions against BCSCs by MTT assay. Viability was decreased more significantly in MCF-7/SCs than MCF-7 cells by the HF and CF (Fig. 2). The CF had the lowest IC50 value (69.47 µg/mL) and exerted a dose-dependent cytotoxic effect on MCF7/SCs (Table 5). These results indicated that the CF with negligible antioxidant activity had a considerable antiproliferative effect on MCF7/SCs. Therefore, we proceeded with the experiment to determine whether CF attenuates the cancer stem characteristics of MCF7/SCs.
Effect of the chloroform fraction on the stemness characteristics of MCF7/SCs
The inhibitory effect of the CF on cell growth was confirmed by colony formation assay (Fig. 3A). CD44+/CD24− BCSCs express genes related to cell motility and angiogenesis [25]. FACS analysis showed a marked decrease in the CD44+/CD24− population of MCF-7/SCs treated with CF at 100 μg/mL, at both 24 and 48 h (Fig. 3B). In addition, ALDH activity, a metabolic cancer stem cell marker [26], was markedly reduced by non-lethal concentrations of CF (Fig. 3C). At non-lethal concentrations, the migration and invasion of MCF-7/SCs were suppressed by CF (Fig. 3D, E). Western blotting showed that the levels of β-catenin, CD44, and MRP1 were significantly decreased in MCF-7/SCs treated with CF in a dose-dependent manner (Fig. 3F). Taken together, these results show that the CF inhibits the stemness of MCF-7/SCs.
CF attenuates stem like-cell characteristics in MCF-7/SCs as a BCSC. A Colonies were formed via CF treatment for 10 days (B) CD44+/CD24−/low population measured by FACS after 24 or 48 h of treatment with CF. C ALDH+ population after CF treatment for 24 h determined using an Aldefluor assay kit; negative control, diethylaminobenzaldehyde (DEAB). D Migrated cells after 24 or 48 h of treatment with CF by wound healing assay. Phase-contrast micrographs (100 × magnification). E Cell invasion determined using Transwells after 24 h of treatment with CF (0–50 µg/mL). Phase-contrast micrographs (100 × magnification). (F) Stemness markers after 24 h of treatment with CF detected by Western blotting. β-actin was used as the loading control. Data are means ± standard deviation (N = 3). *P < 0.05 for each group compared with the control
Induction of apoptosis by the chloroform fraction
Apoptosis is characterized by morphological changes such as condensed and fragmented chromatin with apoptotic bodies [27, 28]. Hoechst 33342 staining revealed condensed nuclei in CF-treated cells, but not in control cells (Fig. 4A). Also, the CF induced early and late apoptotic cell death in a time- and dose-dependent manner. The percentage of apoptotic cells was increased by 24.73 ± 7.03% and 54.53 ± 16.92% by treatment with 100 µg/mL CF for 24 and 48 h, respectively (Fig. 4B). Furthermore, the levels of apoptosis-related proteins detected by western blotting showed that the Bax to Bcl-2 ratio decreased in a dose-dependent manner. Furthermore, the CF reduced the levels of pre-caspases-9, -3, and -7, whereas the proteolytically cleaved PARP level increased significantly in a concentration-dependent manner (Fig. 4C). Therefore, the CF induced apoptosis of MCF-7/SCs.
CF induces apoptosis of MCF-7/SCs. A Apoptotic nuclei observed by Hoechst 33,342 staining after 24 h of treatment with CF. B Cells were treated with CF for 24 h and stained with annexin V-FITC/PI. C Apoptosis markers after 24 h of treatment with CF by Western blotting. β-actin was used as the loading control. Data are means ± standard deviation (N = 3). *P < 0.05 for each group compared with the control. The English in this document has been checked by at least two professional editors, both native speakers of English. For a certificate, please see: http://www.textcheck.com/certificate/e1cbmq
GC–MS analysis of the chloroform fraction
The CF was analyzed by Gas chromatography-mass spectrometry (GC–MS) to identify its lipophilic constituents. The dominant compounds in CF were fatty acids, such as oleic acid, pentadecanoic acid, and methyl palmitate (Table 6). Interestingly, CF had the highest content of oleic acid (35.053%), which likely explains its effect on BCSCs. Oleic acid reportedly promotes growth and migration in MDA-MB-231, MCF-7 breast cancer cells and colorectal cancer cells [29, 30], on the contrary, induces autophagy in hepatocellular carcinoma and suppresses the growth of BT-474, SK-Br3 breast cancer cells [31,32,33]. Therefore, although BF has relatively high polyphenol and flavonoid content, CF, a non-polar solvent, showed the best antiproliferative effect than BF, which is presumed to be due to the very high content of oleic acid in CF. Further in-depth studies of the roles of oleic acid on BCSCs are needed. Overall, the broccoli sprout CF inhibited the growth and stem cell properties of BCSCs, suggesting that broccoli sprouts have potential as functional foods against breast cancer.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- ABTS:
-
2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic-acid)
- BCSCs:
-
Breast cancer stem cells
- BF:
-
Butanol fraction
- CF:
-
Chloroform fraction
- DPPH:
-
2,2-Diphenyl-1-picryhydrazyl
- EC50 :
-
The effective concentration of drug that causes 50% of the maximum response
- EF:
-
Ethyl acetate fraction
- GC–MS:
-
Gas chromatography mass spectrometry
- HF:
-
Hexane fraction
- HPLC:
-
High performance liquid chromatography
- IC50 :
-
Inhibitory concentration that causes 50% of the maximum inhibition
- ME:
-
Methanol extract
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- WF:
-
Water fraction
References
Guine RPF, Florenca SG, Barroca MJ, Anjos O (2020) The link between the consumer and the innovations in food product development. Foods 9(9):1317
Pollock RL (2016) The effect of green leafy and cruciferous vegetable intake on the incidence of cardiovascular disease: a meta-analysis. JRSM Cardiovasc Dis 5:1–9
Manchali S, Murthy KNC, Patil BS (2012) Crucial facts about health benefits of popular cruciferous vegetables. J Funct Foods 4(1):94–106
Gao J, Yu X, Ma F, Li J (2014) RNA-seq analysis of transcriptome and glucosinolate metabolism in seeds and sprouts of broccoli (Brassica oleracea var. italic). PLoS ONE 9(2):e88804
Kim E-J, Kim M-H (2015) Antioxidant activity of solvent fraction from broccoli sprouts cultivated at the plant factory system. Korean J Food Nutr 28(1):1–8
Jang HW, Moon J-K, Shibamoto T (2015) Analysis and antioxidant activity of extracts from broccoli (Brassica oleracea L.) sprouts. J Agr Food Chem 63(4):1169–1174
Abdulah R, Faried A, Kobayashi K, Yamazaki C, Suradji EW, Ito K, Suzuki K, Murakami M, Kuwano H, Koyama H (2009) Selenium enrichment of broccoli sprout extract increases chemosensitivity and apoptosis of LNCaP prostate cancer cells. BMC Cancer 9(1):1–12
Dinkova-Kostova AT, Jenkins SN, Fahey JW, Ye L, Wehage SL, Liby KT, Stephenson KK, Wade KL, Talalay P (2006) Protection against UV-light-induced skin carcinogenesis in SKH-1 high-risk mice by sulforaphane-containing broccoli sprout extracts. Cancer Lett 240(2):243–252
Heer E, Harper A, Escandor N, Sung H, McCormack V, Fidler-Benaoudia MM (2020) Global burden and trends in premenopausal and postmenopausal breast cancer: a population-based study. Lancet Glob Health 8(8):e1027–e1037
Charafe-Jauffret E, Ginestier C, Iovino F, Wicinski J, Cervera N, Finetti P, Hur MH, Diebel ME, Monville F, Dutcher J et al (2009) Breast cancer cell lines contain functional cancer stem cells with metastatic capacity and a distinct molecular signature. Cancer Res 69(4):1302–1313
Prieto-Vila M, Takahashi R-u, Usuba W, Kohama I, Ochiya T (2017) Drug resistance driven by cancer stem cells and their niche. Int J Mol Sci 18(12):2574
Ji XW, Lu Y, Tian HF, Meng XR, Wei MJ, Cho WC (2019) Chemoresistance mechanisms of breast cancer and their countermeasures. Biomed Pharmacother 114
Chavan U, Shahidi F, Naczk M (2001) Extraction of condensed tannins from beach pea (Lathyrus maritimus L.) as affected by different solvents. Food Chem 75(4):509–512
Abubakar AR, Haque M (2020) Preparation of medicinal plants: basic extraction and fractionation procedures for experimental purposes. J Pharm Bioallied Sci 12(1):1
Kang HR, Koh SY, Ryu J-y, Osman A, Lee CK, Lim JH, Kim HA, Im GH, Cho SK (2016) Antioxidant activities and physicochemical properties of chocolate fermented by Lactobacillus plantarum CK10. Korean J Food Preserv 23(4):576–584
Ryu J-y, Choi Y, Hong K-H, Chung YS, Cho SK (2020) Effect of roasting and brewing on the antioxidant and antiproliferative activities of tartary buckwheat. Foods 9(9):1331
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Bio Med 26(9–10):1231–1237
Cuong D, Ha TW, Park CH, Kim NS, Yeo HJ, Chun SW, Kim C, Park SU (2019) Effects of LED lights on expression of genes involved in phenylpropanoid biosynthesis and accumulation of phenylpropanoids in wheat sprout. Agronomy-Basel 9(6):307
To NB, Nguyen YTK, Moon JY, Ediriweera MK, Cho SK (2020) Pentadecanoic acid, an odd-chain fatty acid, suppresses the stemness of MCF-7/SC human breast cancer stem-like cells through JAK2/STAT3 signaling. Nutrients 12(6):1663
Koh SY, Moon JY, Unno T, Cho SK (2019) Baicalein suppresses stem cell-like characteristics in radio- and chemoresistant MDA-MB-231 human breast cancer cells through up-regulation of IFIT2. Nutrients 11(3):624
Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. Sci World J 2013:1–16
Halliwell B (1995) How to characterize an antioxidant: an update. Biochem Soc Symp 73–101
Cosme P, Rodriguez AB, Espino J, Garrido M (2020) Plant phenolics: bioavailability as a key determinant of their potential health-promoting applications. Antioxidants-Basel 9(12):1263
Yang L, Shi P, Zhao G, Xu J, Peng W, Zhang J, Zhang G, Wang X, Dong Z, Chen F (2020) Targeting cancer stem cell pathways for cancer therapy. Signal Transduct Target Ther 5(1):1–35
Fillmore C, Kuperwasser C (2007) Human breast cancer stem cell markers CD44 and CD24: enriching for cells with functional properties in mice or in man? Breast Cancer Res 9(3):1–3
Vassalli G (2019) Aldehyde dehydrogenases: not just markers, but functional regulators of stem cells. Stem Cells Int 2019:1–15
Saraste A, Pulkki K (2000) Morphologic and biochemical hallmarks of apoptosis. Cardiovasc Res 45(3):528–537
Sanders E (1997) Invited review methods for detecting apoptotic cells in tissues. Histol Histopathol 12:1169–1177
Marcial-Medina C, Ordoñez-Moreno A, Gonzalez-Reyes C, Cortes-Reynosa P, Salazar EP (2019) Oleic acid induces migration through a FFAR1/4, EGFR and AKT-dependent pathway in breast cancer cells. Endocr Connect 8(3):252–265
Shen C-J, Chang K-Y, Lin B-W, Lin W-T, Su C-M, Tsai J-P, Liao Y-H, Hung L-Y, Chang W-C, Chen B-K (2020) Oleic acid-induced NOX4 is dependent on ANGPTL4 expression to promote human colorectal cancer metastasis. Theranostics 10(16):7083
Giulitti F, Petrungaro S, Mandatori S, Tomaipitinca L, de Franchis V, D'Amore A, Filippini A, Gaudio E, Ziparo E, Giampietri C (2021) Anti-tumor effect of oleic acid in hepatocellular carcinoma cell lines via autophagy reduction. Front Cell Dev Biol 9
Menendez JA, Vellon L, Colomer R, Lupu R (2005) Oleic acid, the main monounsaturated fatty acid of olive oil, suppresses Her-2/neu (erb B-2) expression and synergistically enhances the growth inhibitory effects of trastuzumab (Herceptin (TM)) in breast cancer cells with Her-2/neu oncogene amplification. Ann Oncol 16(3):359–371
Kim TH, Kwon SC, Kim JN, Yoon JH, Cho SG (2020) Ginseng seed oil inhibits the growth of estrogen receptor-positive breast cancer cells. Anticancer Res 40(8):4529–4535
Acknowledgements
This work was supported by the 2021 education, research and student guidance grant funded by Jeju National University.
Author information
Authors and Affiliations
Contributions
JSK: formal analysis; investigation; methodology; software; writing—original draft; Writing—review and editing. DMC: formal analysis; investigation; methodology; software; writing—original draft. YBB: investigation; SKC: conceptualization; data curation; funding acquisition; project administration; resources; supervision; validation; visualization; writing—review and editing. All author read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
There are no conflicts of interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, J.S., Cuong, D.M., Bae, Y.B. et al. Antioxidant and antiproliferative activities of solvent fractions of broccoli (Brassica oleracea L.) sprout. Appl Biol Chem 65, 34 (2022). https://doi.org/10.1186/s13765-022-00700-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13765-022-00700-2