Abstract
An accurate, precise, sensitive, and simple spectroscopic method was developed and validated for simultaneous quantification analysis of tretinoin (TRT) and nicotinamide (NCT) with a ratio of 1:40 (TRT: NCT) in a synthetic mixture from dermal pharmaceutical preparations (solution and cream). Wavelengths were chosen in the first and second-order derivatives which are valid for the determination of NCT with the existence of TRT and excipients of the tested pharmaceutical preparations. Wavelength 253 nm was picked for the first-order derivative. Wavelengths 245 and 269 nm were picked for the second derivative. All previous wavelengths were zero-crossing points for TRT and its pharmaceutical preparations. Zero-order spectroscopy was used to determine TRT at the wavelength 348 nm, where no interference with NCT or any substance in the previous pharmaceutical preparation. The linearity range was studied and found to be 20–120 μg/mL and 0.5–5.0 μg/mL for NCT and TRT respectively. The correlation coefficient was 0.9995–0.9999 for NCT and 0.9998–0.9999 for TRT. The limit of detection (LOD) and the limit of quantification (LOQ) of NCT were 1.510 μg/mL and 4.590 μg/mL respectively at the wavelength 269 nm of the second-order derivative.
Similar content being viewed by others
Introduction
Nicotinamide (NCT), also called niacinamide is 3-pyridine carboxamide Fig. 1-A [1]. NCT is an amide form of nicotinic acid or niacin Fig. 1-B. NCT is white crystalline powder or colorless crystals. it has a molecular weight of 122.12 g/mol. It is also one of the hydrophilic B vitamins, called vitamin B3 [2].
Many multivitamins and supplementary pharmaceutical preparations contain NCT. It is also found in many dermal preparations, such as solutions, creams, and gels whether as a single active ingredient or with other active ingredients. NCT has concentrations of 3, 4, or 5% (w/w%) in the previous preparations, which are used for many skin conditions including acne [3].
Tretinoin (TRT) is an all-trans-retinoic acid, as shown in Fig. 1-C [4]. It is a first-generation carboxylic form of retinoid, which is a derivative of vitamin A. TRT has a molecular weight of 300.4 g/mol. It is a yellow powder. Furthermore, it is sensitive to light, heat, and oxygen in the air, especially in solutions. TRT is used for several skin conditions, such as acne (as a first-line treatment), psoriasis, and photoaging. It exists as a single active ingredient or with other active ingredients. Its concentration in solution, cream, and gel preparations is 0.025, 0.050, or 0.100% [5,6,7].
TRT can be found in combination with clindamycin or benzoyl peroxide [7]. However, there is no marketed international combination of NCT and TRT spread worldwide. Such a combination may only be found locally, like in the United States, for example, it is found as a compounded drug, which is marketed and distributed by Sincerus® Florida, LLC [8]. According to a study, a combination containing NCT and retinol (an alcoholic form of vitamin A) has good therapeutical potentials for acne [9]. Therefore, we developed an ultra-violet (UV) spectroscopy method to estimate NCT and TRT simultaneously in synthetic mixtures and pharmaceutical preparations.
There are many UV spectroscopic methods [10,11,12,13] in addition to high-performance liquid chromatography (HPLC) [14,15,16,17,18,19] and electrochemical methods [20,21,22], to determine NCT or TRT in combination with other active ingredients. However, no previous studies have developed a method to estimate NCT and TRT simultaneously in a binary mixture or in a pharmaceutical preparation.
The first and second-order derivative methods are simple and accurate for the direct determination of more than one ingredient in mixtures and pharmaceutical preparations, such as TRT with clindamycin [23].
NCT is freely soluble in water, ethanol, and methanol, while slightly soluble in diethyl ether. On the other hand, the solubility of TRT is limited as it is only soluble in dimethyl sulfoxide; slightly soluble in polyethylene glycol 400, octanol, and ethanol; and practically insoluble in mineral oil, glycerin, and water. It is also slightly soluble in methanol, which is used in many developed methods in previous studies since it overcomes ethanol in terms of TRT solubility [11, 24,25,26]. Thus, methanol is used as a solvent in this suggested method.
The aqueous and alcoholic solutions of NCT, such as methanol, are transparent. NCT does not absorb visual light. Instead, it has a sharp absorbance peak in the UV domain at 262 nm [12, 27]. TRT absorbs visual light with wavelengths less than 440 nm. It has a wide peak around 348 nm (basically between 340 and 353 nm) [11, 23, 28]. According to the previous studies, the spectrum of TRT and its minor absorbance in the rest of the UV domain overlaps NCT. In contrast, NCT has no absorbance at wavelengths higher than 300 nm; thus, NCT does not interfere with the signal of TRT.
Materials and methods
Instruments
The ultra-violet spectrophotometric instrument is T80 + UV/V Spectrophotometer Instrument Ltd (UK). It is connected to a computer. The cells used are 1-cm width quartz cells. The weighing device is an analytical balance (Sartorius, model 2474, Germany). Other devices and instruments used to achieve the work are an ultrasonic bath (Power sonic, model 405, Korea), a centrifuge device (90-1 Centrifuge, Shanghai Surgical Instruments Factory, China), a porcelain mortar, volumetric dark flasks, and several scales of glass pipettes.
Solvents and chemicals
Standard active pharmaceutical ingredients are nicotinamide powder 99% (BDH Laboratory Supplies, England), tretinoin powder ≥ 98% gifted by Rama Pharma Co, Aleppo, and methanol of analytical grade (Merck, Germany).
Preparation of standard solutions
Standard solution of nicotinamide
First, 20 mg of NCT was weighed. Then it was transferred into a 20-mL flask and diluted with methanol to the mark to obtain a standard stock solution of NCT with a concentration of 1000 µg/mL. Then, six quantities of 0.2, 0.4, 0.6, 0.8, 1.0, and 1.2 mL were pipetted out to 10-mL flasks and diluted with methanol to prepare a series of standard solutions of NCT with concentrations of 20, 40, 60, 80, 100, and 120 µg/mL.
Standard solution of tretinoin
After weighing 20 mg of TRT, it was transferred into a 20-mL flask and diluted with methanol to obtain a standard stock solution of TRT with a concentration of 1000 µg/mL. Then 0.5 mL was pipetted out to a 20-mL flask to obtain a stock solution of TRT with a concentration of 25 µg/mL. Then, ten quantities from 0.2 to 2.0 mL were pipetted out to 10-mL flasks and diluted with methanol to prepare stock solutions of TRT with concentrations of 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, and 5.0 µg/mL.
Preparation of standard mixture of nicotinamide and tretinoin
The synthetic mixture of NCT and TRT was made in the ratio of 1:40 (TRT: NCT). The same procedure was followed in Sects. 2.3.1 and 2.3.2. Except that the same final 20-mL flask was used for both NCT and TRT with concentrations of 1000 µg/mL and 25 µg/mL respectively.
Preparation of synthetic mixture from pharmaceutical formulas
Solution formula
Locacid®, a dermal solution formula which is produced by Universal Pharma Co, Damascus. It is labeled to contains 1000 µg/mL of TRT (0.1% w/v%). The volume of the container is 30 mL. Two quantities of 0.5 mL of the dermal solution were pipetted. Each one was transferred to a 20-mL flask. A quantity of 20 mg of NCT was weighed and transferred to one of the previous flasks. Both flasks were diluted with methanol to obtain two work solutions. The first one (A) contains a synthetic mixture of; the dermal solution formula, which includes excipients and TRT, plus NCT with a ratio of 1:40 (TRT: NCT) (the same ratio as if the formula also contains 4% of NCT). The second one (B) only contains the formula of the dermal solution which includes excipients and TRT. This procedure was done to study the absorbance of all ingredients of the formula and compare it to the absorbance of the standard solution of TRT. Next, two quantities of 0.4 mL were pipetted from A and B. Each of them was transferred to 4 of 10-mL flasks to obtain 8 solutions. Four of them (A series) contain TRT, excipients, and NCT, while the others (B series) contain only TRT and excipients. Standard additions were added before diluting with methanol. Standard additions of 0%, 50%, 100%, and 150% of NCT and TRT were added to the A series to obtain final concentrations of 40, 60, 80, and 100 µg/mL and 1.0, 1.5, 2.0, and 2.5 µg/mL of NCT and TRT respectively. Standard additions of 0%, 50%, 100%, and 150% of TRT were added to the B series to obtain final concentrations of 1.0, 1.5, 2.0, and 2.5 µg/mL of TRT. The flask with a concentration of 1.0 µg/mL of TRT which has no standard addition was given according to the labeled 0.1% concentration of the formula.
Cream formula
Retinoram®, a dermal cream formula which is produced by Rama Pharma Co, Aleppo. It is labeled to contain 1000 µg/g of TRT (0.1% w/w%). The net weight of the cream is 30 g. Two quantities of 500 mg of the cream were weighed. Each one was transferred to a mortar with 10 mL of methanol for trituration then transferred to a 20-mL flask. Next, each of them was sonicated for 10 min to ensure the maximum disintegration of the cream and full dissolution of TRT. After sonication, a quantity of 20 mg of NCT was weighed and added to one of the previous flasks. Both were diluted with methanol. Then, each of them was centrifuged to obtain two work solutions. The first one (A) contains a synthetic mixture of; the cream formula, which includes excipients and TRT, plus NCT with a ratio of 1:40 (TRT: NCT) (the same ratio as if the formula also contains 4% of NCT). The second one (B) only contains the formula of the cream which includes excipients and TRT. Next, two quantities of 0.4 mL were pipetted from A and B. Each of them was transferred to 4 of 10-mL flasks to obtain 8 solutions. Four of them (A series) contain TRT, excipients, and NCT, while the others (B series) contain only TRT and excipients. Standard additions were added before diluting with methanol. Standard additions of 0%, 50%, 100%, and 150% of NCT and TRT were added to the A series to obtain final concentrations of 40, 60, 80, and 100 µg/mL and 1.0, 1.5, 2.0, and 2.5 µg/mL of NCT and TRT respectively. Standard additions of 0%, 50%, 100%, and 150% of TRT were added to the B series to obtain final concentrations of 1.0, 1.5, 2.0, and 2.5 µg/mL of TRT. The flask with a concentration of 1.0 µg/mL of TRT which has no standard addition was given according to the labeled 0.1% concentration of the formula.
Results and discussion
First and second-order derivative spectrophotometric optimal wavelengths
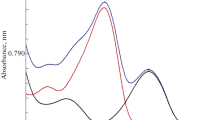
UV spectrogram scan of pure NCT solution, pure TRT solution, and the binary mixture of both; using methanol as solvent appear in Fig. 2. We may notice two problems. The first one is that although the TRT absorption area has a wide region with no absorption of NCT or any excipients of TRT pharmaceutical formulas, the NCT absorption area is overlapped by TRT as mentioned in the last paragraph of the introduction and shown in Fig. 3 where a spectrogram of TRT linearity series are also shown in this figure. NCT is also overlapped by the absorption of the pharmaceutical formulas of TRT especially the cream as shown in Fig. 4. The second one is that NCT with a concentration of 80 µg/mL has absorption around the limit of linearity range at the zero-order (D0) 262 nm peak because of the high absorptivity of NCT at this wavelength; thus, first and second orders derivatives (D1 and D2) are needed and produced. Zero-crossing points of TRT and pharmaceutical preparations have been found in D1 and D2 for the determination of NCT at those points. Thus, we can solve the overlapping problem and determine NCT in the mixtures with concentrations that may exceed 120 µg/mL, yet they stay under the upper limit of the linearity range. While TRT concentration can be determined in zero-order at 348 nm as had been analyzed in many other studies [10, 11, 28,29,30].
For the first-order derivative (amplified 10 times [coefficient × 10]), wavelength 253 nm is a zero-crossing point for TRT and its pharmaceutical preparations spectra. While NCT has a sharp peak at this wavelength as shown in Fig. 5. A low concentration of 30 µg/mL of NCT was used for the demonstration. For TRT, concentrations in the middle of its range 1.0–3.0 µg/mL were used for the demonstration.
For the second-order (amplified 10 times [coefficient × 10]), more than two wavelengths were found to be zero-crossing points for TRT and its preparations spectra. Only two of them were selected (245 nm and 269 nm), where NCT has a sharp peak as shown in Fig. 6. However, 245 nm was not a zero-crossing point for the cream preparation due to its excipients; thus, 269 nm is the only wavelength for the cream formula.
Method validation
Linearity
The spectra of NCT and TRT were taken and NCT linearity was studied at three wavelengths, which are D1 253 nm, D2 245 nm, and D2 269 nm, while TRT linearity was studied at D0 348 nm. D1 and D2 are amplified by 10 times for better resolution in the whole study. Additionally, correlation coefficient (r2), the limit of detection (LOD), and the limit of quantification (LOQ) were found. Results are shown in Table 1. We may notice from the results; accepted linearity ranges and correlation coefficients, and low LOD—LOQ for both NCT in the three wavelengths and TRT. Data were collected and processed according to ICH procedures, except for the stability and robustness studies [31].
Stability
The stability was studied at room temperature (25 ℃), at low relative humidity, and in a dark flask for both NCT and TRT using methanol as solvent. However, NCT is already known for its good stability in both aqueous and alcoholic solutions. NCT 1 mg/mL in methanol can be purchased and transported worldwide by many suppliers. Our study shows that NCT is stable in methanol in storage at room temperature for more than 1 week [32].
The results of the TRT stability study are shown in Table 2. They show that TRT is stable in methanol in storage at room temperature for less than 3 days. This study was done for 3 concentrations of 0.75, 1.50, and 2.25 µg/mL each day.
Accuracy
Accuracy study was achieved for TRT at D0 348 nm and NCT at D1 253 nm, D2 245 nm, and D2 269 nm. Three concentrations and three repetitions for each were analyzed at each of the previous wavelengths. Table 3 shows the mean, the relative standard deviation (RSD%), and the range of percentage values of the total 9 samples of NCT and TRT.
Precision
The precision study was achieved for TRT at D0 348 nm and NCT at D1 253 nm, D2 245 nm, and D2 269 nm. Three concentrations with three repetitions for each were analyzed at each of the previous wavelengths for the intraday repeatability precision. The same number of samples were analyzed after 24 h and again after 48 h for the intermediate inter-day precision with a total of 27 samples. RSD% values for all cases are shown in Table 4.
Specificity
Specifity studies were achieved by using the mixture of NCT and TRT with several ratios for the same wavelengths as previous Sections “Accuracy” and “Precision” results are shown in Table 5. The method has good recovery values for a wide range of the ratio of the binary mixture contents compared to the most used ratio of 1:40 (TRT: NCT).
Robustness
A robustness study was achieved for TRT and NCT in only D2 245 nm for three different instrumental parameters changes and three repetitions for a minor change in each parameter independently. First, the scan speed was changed from medium to fast. Second, the scan range was changed from 200–400 nm to 190–350 nm. Third, the bandwidth of the light beam was changed from 0.2 nm wide to 0.1 nm wide. The developed method appeared to be robust for TRT with the three tests but only robust with two of them for NCT. Recovery varied when the bandwidth of the spectrometer was changed. That was probably due to dependence on its strict method of zero-crossing point. NCT had recovery ranges of: 98.13–101.91% for scan speed parameter, 100.26–101.91% for scan range parameter, and 102.56–116.10% for bandwidth parameter. TRT had recovery ranges of: 100.16–101.42% for scan speed parameter, 99.67–100.84% for scan range parameter, and 98.33–99.35% for bandwidth parameter.
Application of the proposed method for pharmaceutical preparations
The pharmaceutical preparations were analyzed for their content of TRT and the content of the added NCT from standard solutions. In this study standard additions method has been used for both NCT and TRT with three repetitions. All amounts and concentrations that were prepared for this test are mentioned in Section “Preparation of synthetic mixture from pharmaceutical formulas”.
Dermal solution preparation
The recovery% and RSD% of both TRT and NCT for each selected wavelength are shown in Table 6. As shown, the recovery values are within the accepted range in ICH guidelines.
TRT in pharmaceutical preparations has an accepted assay range of 90–120% in several pharmacopeias like the British Pharmacopeia (BP) and the United States Pharmacopeia (USP) because the concentration of TRT in the preparations is 0.1% or less [33, 34].
Dermal cream preparation
The recovery% and RSD% of both TRT and NCT for each selected wavelengths are shown in Table 6. The studied cream has no zero-crossing point around 245 nm D2 as we mentioned before in Section “First and second-order derivative spectrophotometric optimal wavelength” thus NCT couldn't be studied at that point.
Conclusion
It could be concluded from the results obtained in the present paper that the developed method for simultaneous determination of NCT and TRT in binary mixtures is simple, accurate, robust, precise, and rapid. This method can be used for routine quality control tests to directly determine NCT and TRT individually or simultaneously in a binary combination of either solution or cream formula with their excipients in the mixture without any prior separation. Moreover, it's recommended to analyze TRT within 48 h after being dissolved in methanol because TRT is stable within this period according to the stability test.
Availability of data and materials
Data and materials are included in this manuscript.
Abbreviations
- BP:
-
British pharmacopeia
- D0 :
-
Zero-order spectrum
- D1 :
-
First-order derivative
- D2 :
-
Second order derivative
- HPLC:
-
High-performance liquid chromatography
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantification
- NCT:
-
Nicotinamide
- PP:
-
Pellagra preventing substance
- r2 :
-
Correlation coefficient
- RARs:
-
Retinoic acid receptors
- RSD%:
-
Relative standard deviation
- TRT:
-
Tretinoin
- USP:
-
United States pharmacopeia
- UV:
-
Ultra-violet
- λmax :
-
Wavelength with maximum absorbance
References
National Center for Biotechnology Information. PubChem Compound Summary for CID 936, Nicotinamide (cited 2021 Oct 26). https://pubchem.ncbi.nlm.nih.gov/compound/nicotinamide.
O’Rourke A. Martindale: The Complete Drug Reference, vol 67, 36th ed. Sweetman S, editor. American Journal of Health-System Pharmacy. Pharmaceutical Press; 2010. 320, 1577, p. 1957–1959.
Walocko FM, Eber AE, Keri JE, AL-Harbi MA, Nouri K. The role of nicotinamide in acne treatment. Dermatol Ther. 2017;30(5):1–7.
National Center for Biotechnology Information. PubChem Compound Summary for CID 444795, Tretinoin (cited 2022 Feb 3). https://pubchem.ncbi.nlm.nih.gov/compound/444795.
European Pharmacopoeia Commission. European Pharmacopoeia, vol 2. 7.0. Strasbourg: Council of Europe. 2011; p. 3125–3126.
Whalen K, Anderson S, Finkel R, Birnbaum AK, Gauthier TP, Carris N, et al. Lippinchots Illustrated Reviews, 6th ed. In: Whalen K, Finkel R, Panavelil TA, editors. Wolters Kluwer. 2014; p. 431–433.
Zaenglein AL, Pathy AL, Schlosser BJ, Alikhan A, Baldwin HE, Berson DS, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74(5):945-973.e33.
NDC 72934–1204 Topical Gel 011020 NIACINAMIDE 4%/TRETINOIN 0.05% Drug Codes, Packaging, Active Ingredients (cited 2022 Jan 19). https://fda.report/NDC/72934-1204.
Emanuele E, Bertona M, Altabas K, Altabas V, Alessandrini G. Anti-inflammatory effects of a topical preparation containing nicotinamide, retinol, and 7-dehydrocholesterol in patients with acne: a gene expression study. Clin Cosmet Investig Dermatol. 2012;5:33–7.
Bordbar M, Yeganeh-Faal A, Ghasemi J, Ahari-Mostafavi MM, Sarlak N, Baharifard MT. Simultaneous spectrophotometric determination of minoxidil and tretinoin by the H-point standard addition method and partial least squares. Chem Pap. 2009;63(3):336–44.
Gupta A, Gulati M, Pandey NK. A validated UV spectrophotometric method for simultaneous estimation of tretinoin and benzoyl peroxide in bulk and semi solid dosage form. Rasayan J Chem. 2009;2(3):649–54.
Darweesh SA, Khalaf HS, Al-khalisy RS, Yaseen HM, Mahmood RM. Advancement and validation of new derivatives spectrophotometric method for individual and simultaneous estimation of diclofenac sodium and nicotinamide. Orient J Chem. 2018;34(3):1625–32.
López-de-Alba PL, López-Martínez L, Cerdá V, Amador-Hernández J. Simultaneous determination and classification of riboflavin, thiamine, nicotinamide and pyridoxine in pharmaceutical formulations, by UV-visible spectrophotometry and multivariate analysis. J Braz Chem Soc. 2006;17(4):715–22.
Ye YR, Bektic E, Buchta R, Houlden R, Hunt B. Simultaneous determination of tretinoin and clindamycin phosphate and their degradation products in topical formulations by reverse phase HPLC. J Sep Sci. 2004;27(1–2):71–7.
Kril MB, Burke KA, DiNunzio JE, Rao GR. Determination of tretinoin in creams by high-performance liquid chromatography. J Chromatogr A. 1990;522:227–34.
Sheliya K, Shah K, Kapupara P. Development and validation of analytical method for simultaneous estimation of mometasone furoate, hydroquinone and tretinoin in topical formulation by RP-HPLC. J Chem Pharm Res. 2014;6(4):934–40.
Ibrahim F, El-Deen AE, El Abass SA, Shimizu K. An ecofriendly green liquid chromatographic method for simultaneous determination of nicotinamide and clindamycin phosphate in pharmaceutical gel for acne treatment. J Food Drug Anal. 2017;25(3):741–7.
Banik K, Rath S, Gupta BK. Chromatographic estimation of niacinamide in anti aging dermatological product. Asian J Pharm Anal. 2019;9(2):62–6.
Khan AR, Khan KM, Perveen S, Butt N. Determination of nicotinamide and 4-aminobenzoic acid in pharmaceutical preparation by LC. J Pharm Biomed Anal. 2002;29(4):723–7.
Barthus RC, Mazo LH, Poppi RJ. Simultaneous determination of vitamins C, B6 and PP in pharmaceutics using differential pulse voltammetry with a glassy carbon electrode and multivariate calibration tools. J Pharm Biomed Anal. 2005;38(1):94–9.
Wang LH. Simultaneous determination of retinal, retinol and retinoic acid (all-trans and 13-cis) in cosmetics and pharmaceuticals at electrodeposited metal electrodes. Anal Chim Acta. 2000;415(1–2):193–200.
Alghamdi AF. Development and validation of anodic stripping voltammetry method for the determination of tretinoin in human urine and plasma using glassy carbon electrode. J King Saud Univ Sci. 2020;32(5):2635–40.
Tehrani MB, Namadchian M, Vatan SF, Souri E. Derivative spectrophotometric method for simultaneous determination of clindamycin phosphate and tretinoin in pharmaceutical dosage forms. DARU J Pharm Sci. 2013;21–9.
British Pharmacopoeia Commission. Tretinoin. In: British Pharmacopoeia 2009. London: Crown; 2009.
Pharmacopoeia Society of Japanese. The Japanese Pharmacopoeia, 17th ed. Tokyo: Yakuji Nippo Ltd. 2016; p. 1306.
Pankti D, Kusum M, Mehul P. Development and validation of UV-Visible Spectrophotometric method for simultaneous estimation of momentasone furoate, hydroquinone and tretinoin from their pharmaceutical dosage form. Int J Pharm Sci Rev Res. 2013;21(1):296–300.
Kumar SVV, Kavitha J, Lakshmi KS. Uv spectrophotometric quantification of niacinamide in pharmaceutical dosage form by multivariate calibration technique. Res J Pharm Technol. 2021;14(4):2013–20.
Jiang XG, Xi NZ. A reversed-phase HPLC method for determining tretinoin. Zhongguo Yao Li Xue Bao. 1994;15(5):458–61.
Ridolfi DM, Marcato PD, Justo GZ, Cordi L, Machado D, Durán N. Chitosan-solid lipid nanoparticles as carriers for topical delivery of tretinoin. Colloids Surf B Biointerfaces. 2012;93:36–40.
Shin SC, Kim HJ, Oh IJ, Cho CW, Yang KH. Development of tretinoin gels for enhanced transdermal delivery. Eur J Pharm Biopharm. 2005;60(1):67–71.
ICH Harmonised Tripartite Guidline. Validation of analytical procedures: text and methodology Q2(R1). Geneva; 2005. https://database.ich.org/sites/default/files/Q2%28R1%29Guideline.pdf.
Nicotinamide 1.0 mg/ml in Methanol | LGC Standards (cited 2021 Nov 1). https://www.lgcstandards.com/FR/en/Nicotinamide-1-0-mg-ml-in-Methanol/p/LGCAMP0552.00-01.
British Pharmacopoeia Commission. Tretinoin Solution. In: British Pharmacopoeia 2009. London: Crown; 2009.
United States Pharmacopeial Convention. The United States Pharmacopeia 30; The National Formulary 25. 30th ed. Rockville, Maryland: United States Pharmacopeial Convention. 2007; p. 3389–3390.
Acknowledgements
The authors express their gratitude to Rama Pharma Co. for providing the tretinoin powder.
Funding
This work wasn't funded by any third party.
Author information
Authors and Affiliations
Contributions
NS planned and supervised the study and participated in writing and reviewing the manuscript, AS carried out sample preparation, analysis, and writing the manuscript. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Sarkis, N., Sawan, A. Development and validation of derivative UV spectroscopic method for simultaneous estimation of nicotinamide and tretinoin in their binary mixtures and pharmaceutical preparations. BMC Chemistry 16, 15 (2022). https://doi.org/10.1186/s13065-022-00809-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13065-022-00809-x