Abstract
Background
Our group developed a rehabilitation robot to assist with repetitive, active reaching movement of a paretic upper extremity. The robot is equipped with a servo motor-controlled arm-weight support and works in conjunction with neuromuscular electrical stimulation and vibratory stimulation to facilitate agonist-muscle contraction. In this before-and-after pilot study, we assessed the feasibility of applying the robot to improve motor control and function of the hemiparetic upper extremity in patients who suffered chronic stroke.
Methods
We enrolled 6 patients with chronic stroke and hemiparesis who, while sitting and without assistance, could reach 10 cm both sagitally and vertically (from a starting position located 10 cm forward from the patient’s navel level) with the affected upper extremity. The patients were assigned to receive reaching exercise intervention with the robot (Yaskawa Electric Co., Ltd. Fukuoka, Japan) for 2 weeks at 15 min/day in addition to regular occupational therapy for 40 min/day. Outcomes assessed before and after 2 weeks of intervention included the upper extremity component of the Fugl-Meyer Assessment (UE-FMA), the Action Research Arm Test (ARAT), and, during reaching movement, kinematic analysis.
Results
None of the patients experienced adverse events. The mean score of UE-FMA increased from 44.8 [SD 14.4] to 48.0 [SD 14.4] (p = 0.026, r = 0.91), and both the shoulder–elbow and wrist–hand scores increased after 2-week intervention. An increase was also observed in ARAT score, from mean 29.8 [SD 16.3] to 36.2 [SD 18.1] (p = 0.042, r = 0.83). Kinematic analysis during the reaching movement revealed a significant increase in active range of motion (AROM) at the elbow, and movement time tended to decrease. Furthermore, trajectory length for the wrist (“hand path”) and the acromion (“trunk compensatory movement”) showed a decreasing trend.
Conclusions
This robot-assisted modality is feasible and our preliminary findings suggest it improved motor control and motor function of the hemiparetic upper extremity in patients with chronic stroke. Training with this robot might induce greater AROM for the elbow and decrease compensatory trunk movement, thus contributing to movement efficacy and efficiency.
Trial registration UMIN Clinical Trial Registry, as UMIN000018132, on June 30, 2015. https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr_view.cgi?recptno=R000020398
Similar content being viewed by others
Background
Stroke is a leading cause of death and disability. In 2017, the number of patients treated for stroke in Japan was 1,115,000, with 109,844 deaths [1, 2]. Many survivors of stroke require nursing care to some extent; in fact, patients with stroke account for the largest percentage of claims under the Japanese Long-term Care Insurance System [3]. In a previous review, about 90% of patients with stroke had hemiparesis on admission, and less than 15% of them experienced complete motor recovery [4]. In stroke rehabilitation, some principles are well accepted: high-intensity, task-specific, goal-setting, and multidisciplinary-team care are needed to be effective [5]. Among these principles, “task-specific” might be controversial, because some theories of motor control suggest that, on the contrary, motor learning improves, and acquires greater generalizability, when a training program offers variability [6, 7]. The appropriate approach probably depends on the aim of rehabilitation (which can be subject-dependent): for example, a reaching movement with the arm is frequently needed in activities of daily living.
Robotic rehabilitation is a novel intervention method, and several reviews have noted that it leads to improved muscle strength and motor control of the affected upper extremity [8, 9]. A recent Cochrane review suggests that electromechanical and robot-assisted arm training might improve arm function, muscle strength of the upper extremity, and even activity of daily living after stroke [10]. Robotic devices can enable patients to perform task-specific, high-intensity rehabilitation due to increased repetition or amount of training.
At the same time, neuromuscular electrical stimulation (NMES) is widely employed as a rehabilitation technique. According to a previous study, NMES is effective at improving motor control and motor function of affected arms of patients with acute stroke [11], and the NMES system was more efficient when applied with a high-voltage pulsed current [12]. Although few studies have investigated untriggered NMES for the hemiparetic upper limb, continuous electrical stimulation with robotic training improved active range of motion and motor control [13], and we employed the NMES system without triggered electromyography (EMG) [14]. Continuous stimulation with NMES has been considered to be effective in facilitating contraction of paretic muscles [14]. Furthermore, the latest meta-analysis showed that electrical stimulation was effective for arm function and activity regardless of the stimulation type (NMES, EMG triggered, or sensory) [15].
Functional vibratory stimulation (FVS) is known to produce a favorable effect on spasticity, motor control, and gait after stroke [16]. Regarding hemiparetic upper extremities, previous studies have shown that focal vibration applied to paretic muscles is effective at decreasing spasticity with an amplitude of 91 Hz [17], and that it probably improves motor control with an amplitude of 120 Hz, especially in terms of smoothness of movement [18]. For the lower extremity, a previous study revealed that focal vibration improved gait by promoting contraction of the target muscle [19]. Moreover, not only did it promote contraction of the agonist muscle, low amplitude vibratory stimulation (80 Hz) also facilitated focused motorcortical activation [20, 21]. In addition, tendon or muscle vibration produces a tonic vibration reflex through both spinal and supraspinal pathways via repetitive activation of Ia afferent fibers [22, 23]. It is possible to artificially elicit the illusion of movement by vibrating the tendons or the muscles through the skin [24]; the illusion is probably mediated by the activation of muscle spindles [25]. This phenomenon indicates that vibration induces a strong proprioceptive feedback. On the other hand, it has been reported that the vastus lateralis muscle demonstrates a shift toward more appropriate muscle timing when vibration is applied during stance phase and transition to stance of the gait cycle in patients with spinal cord injury [26]. This indicates that strong sensory feedback from quadriceps vibration caused increased muscle excitation [26]. Thus, the combination of muscle vibration with NMES might help to recruit Ia afferent fibers and increase muscle force production. This phenomenon has already been demonstrated in healthy people in the plantar flexors [27]. To the best of our knowledge, however, the use of a robotic device equipped with electrical stimulation and vibration has not been reported.
Considering these facts, our group undertook to develop a rehabilitation robot to assist with repetitive, active reaching movement of the paretic upper extremity; patent acquisitions [28,29,30] and product development were accomplished with a medical–engineering collaboration within Kagoshima University and collaboration between industry (Yaskawa Electric Co., Ltd., Fukuoka, Japan) and academia (Kagoshima university). The robot is equipped with a servo motor-controlled arm-weight support via a wire—the system is programmed to assist the patient’s paretic arm to move between two switches (sensors) located at various three-dimensional positions, which provide a variety of reaching tasks—and works in conjunction with NMES and vibratory stimulation to facilitate agonist-muscle contraction, because the combination might strengthen proprioceptive feedback and tonic vibration reflex. Indeed, this device was applicable and beneficial for a patient with incomplete spinal cord injury [31]. In the before-and-after pilot study reported here, we assessed the feasibility of our novel approach of applying the robot equipped with electrical stimulation and vibration to improve motor control and function of the hemiparetic upper extremity in patients who suffered chronic stroke.
Results
All six patients fully accomplished the procedure, including assessments, before and after the intervention. No adverse events were observed during the study.
Clinical measures
The changes in the UE-FMA and ARAT scores before and after 2 weeks intervention are shown in Table 1. UE-FMA and ARAT scores increased significantly. After the intervention, the mean UE-FMA score was significantly higher (p = 0.026, r = 0.91). Both the shoulder–elbow score (p = 0.041, r = 0.83) and the wrist–hand score (p = 0.039, r = 0.84) of UE-FMA were significantly higher. A significant increase was also observed in ARAT scores (p = 0.042, r = 0.83).
Changes in MAS scores were not statistically significant. The mean MAS score assessment did not change in terms of the wrist flexor muscles (from 0.5 [SE 0.2] to 0.5 [SE 0.3]; p > 0.999). It decreased slightly in the elbow flexor muscles (from 1.3 [SE 0.5] to 1.0 [SE 0.4]; p = 0.157) and in the flexor digitorum muscles (from 0.3 [SE 0.2] to 0.2 [SE 0.2]; p = 0.317).
Kinematic analysis
All of the participants achieved task movements with the target button placed 10 cm or 20 cm from the start button in the sagittal and vertical directions. Only four of the six patients reached the target button when attempting to reach 30 cm from the start button. Three patients could reach the ipsilateral target and two patients could reach the contralateral target. Therefore, the movement time and the trajectory length were compared only among participants who achieved the task movement. However, AROM of the elbow and shapes of trajectories were measured and compared in all conditions, even if a patient could not successfully perform the tasks.
The AROM of the elbow for all targets tended to increase after the completion of the intervention, and it increased significantly for the target 30 cm from the start button (p = 0.028, r = 0.9), as well as in the contralateral workspace (p = 0.028, r = 0.9). On the other hand, AROM showed no significant change when the target button was in the ipsilateral workspace (p = 0.116, r = 0.64). Changes in AROM of elbow for the three above-mentioned conditions are reported in Table 2.
Movement time while reaching showed no significant changes. However, mean values of movement time generally became shorter in all conditions, especially among participants who completed the task.
Table 3 shows changes in trajectory lengths for reaching to the target 20 cm from the start button before and after intervention. All participants completed this task successfully. Trajectory length tended to decrease with both wrist and shoulder in all five conditions, although none of the changes was statistically significant.
Trajectory shape for the wrist tended to be close to a straight line from the start button to the target button, or raised smoothly after 2 weeks intervention, which was consistent with the tendency towards decreasing trajectory length.
Figure 1 shows trajectories of patient 2 as an example. The trajectory (standardized thick line) of the wrist after intervention raised smoothly to the target, although it was slightly above the target (Fig. 1b). Meanwhile, the trajectory of the shoulder shortened in the direction of the horizontal axis (Fig. 1d). The SD of trajectories (areas around the standardized lines, displayed in Fig. 1b, d) in wrist and shoulder decreased after intervention.
Trajectories of patient 2. Condition for the task movement: the target button 30 cm away from the start button in the sagittal and vertical directions. The vertical axis expresses height and the horizontal axis expresses forward length; each box is a square with sides 40 cm long. The start button is located at the bottom left, and the target button is located towards the upper right. The trajectory is shown as a standardized thick line. Compared with the task before intervention (a), the trajectory of the wrist after intervention raised smoothly (b), although it was slightly above the target. At the same time, the trajectory of the shoulder after intervention (d) was shortened in the direction of the horizontal axis compared with the shape before intervention (c)
Additionally, trajectories of reaching movements with non-paretic arms appeared to be considerably tight for both shoulder and wrist of all patients. The result for the non-paretic arm of patient 2 is presented in Fig. 2.
Trajectories for the non-paretic arm of patient 2. Condition for the task movement: the target button 30 cm away from the start button in the sagittal and vertical directions. The wrist trajectory (a) showed a nearly straight line from the start button to the target button, and the shoulder (b) did not shift forward
Furthermore, we considered seven aspects of kinematic analysis, including some that are subjective, to further understand individual patient trajectories during the reaching condition in which the patient could reach the maximum distance both before and after the intervention; four out of the six patients (patients 1 to 4) reached the target button 30 cm away from the start button in the sagittal and vertical directions, and two out of the six patients (patients 5 and 6) reached the target button 20 cm away in the same directions (Table 4).
Changes in movement time of the wrist, trajectory length for the wrist and shoulder, and forward displacement of the shoulder are objective assessments (Fig. 3, Table 4). As shown in Fig. 3, the movement time tended to decrease (p = 0.058, r = 0.77). Although there was no significant change in trajectory length for the wrist (p = 0.345, r = 0.39), the length trended to decrease for the shoulder (p = 0.116, r = 0.64). In addition, forward displacement for the shoulder tended to decrease (p = 0.08, r = 0.72) (Table 4).
Changes in objective assessment for kinematic analysis before and after 2-week intervention for all six patients. Conditions for the task movement were that individual patient (Pt.) could reach the maximum distance both before and after the 2 weeks intervention; Pt. 1 to 4 reached the target 30 cm away from the start in the sagittal and vertical directions, and patients 5 and 6 reached the target 20 cm away in the same directions. a Movement time for the wrist, b trajectory length for the wrist, c trajectory length for the shoulder, and d forward displacement for the shoulder. P1 to P6 correspond to patients 1 to 6 in the tables, respectively
We also made three subjective assessments: deviations of trajectory for the wrist and shoulder, and changes in movement pattern revealed by the trajectory (Table 4). Movement pattern, which is presumed to indicate the movement strategy, apparently changed in three patients, although it is a subjective assessment. Patient 3 became able to raise her arm smoothly; patient 4 did likewise, and he might have gotten used to the reaching movement because the smooth shape after intervention implies that the time from pressing the target button to pulling his arm down had shortened.
Discussion
This study revealed the feasibility of using a robotic device equipped with electrical stimulation and vibration for rehabilitation of patients with hemiparetic upper limb due to stroke. All six of the patients in this study completed a 2-week intervention without adverse events and showed improved motor control and motor function.
We found that the mean UE-FMA scores increased by 3.2 points. This is the same as the minimal detectable change [32]. Hence, a 2-week intervention might contribute to improvement of motor control of the affected upper-limb. Although changes in scores did not exceed minimal detectable change, we should take account of the current intervention period (2 weeks) when comparing to previous studies. In a previous systematic review and meta-analysis by Veerbeek et al., which investigated the effects of robot-assisted therapy for the paretic upper limb, the mean UE-FMA in the robot-assisted therapy groups was 2.23 points better than in the control groups [8]. On the other hand, the most recent multicenter randomized controlled trial (RCT) reported a mean improvement of 2.54 (0.07 to 5.06) points over usual care at 6 months [33]. In terms of the patient with chronic stroke, such RCTs showed that the robotic rehabilitation group had mean increases in the UE-FMA: 3.25 points after robotic rehabilitation at least 45 min three times a week for 8 weeks (total 24 sessions) [34]; approximately 4 points after 60 min three times a week for 6 weeks (total 18 sessions) [35]; and 3.87 points after high-intensity, repetitive, task-oriented movements (1024 per session on average) for 12 weeks (total 36 sessions) [36]. The current shoulder–elbow scores, including coordination, increased by 1.5 points and the wrist–hand scores increased by 1.7 points. Therefore, for both proximal and distal upper extremities, the intervention in the current study appears to have been effective.
Mean ARAT score changed by 6.4 points, which exceeds the minimal clinically important difference (5.7 points) for patients with chronic stroke [37]. Few studies of robotic rehabilitation have assessed ARAT. The latest RCT compared motor functions of paretic upper extremities among robotic training, intensive training, and usual care [33], but unlike FMA, no significant improvement compared with the controls in ARAT was revealed. However, if “instrumented ARAT” had been used in the previous RCT, a difference might have been detected in subtle arm alterations due to the more sensitive quantification of arm function [38]. Given that fact, our current results seem to be meaningful. In the present robot-assisted modality, the electrical stimulation, and the vibration are applied to the proximal region of the upper limb. However, improvement of shoulder–elbow (proximal) function might also affect stabilizing proximal function during manipulation of objects with regular occupational therapy or in activities of daily living. Although participants were only asked to repeat a reaching movement forward and upward in this study, it is considered that such repetitive task training was also effective to improve motor function in the paretic upper limb.
The reaching exercise with this robotic device—in addition to regular occupational therapy—appears to be effective for patients with chronic stroke. The strongest aspect of the device seems to be supporting the affected arm against gravity. In previous studies, gravity-supported exercise for patients with chronic stroke with hemiparesis provided functional recovery of upper extremities measured by the FMA [39, 40]. In addition, in the current study an adjusting system controlled by a servo motor, which could adjust the amount of arm weight relieved depending on the ability of patient, might allow the patient to more comfortably perform a greater number of repetitive reaching exercises. However, the amount of arm-weight support was not predefined in the present machine, nor was it obtained after calculating motor torque. This could be improved by determining the actual percentage of weight reduced in relation to the patient’s arm. This differs from patient to patient, so a relationship based on an individual’s weight could be used to better estimate the force acting to leverage the arm’s weight. The estimated amount of support could then be regulated according to the patient’s requirements. For instance, the approximate weight of the total arm is about 4.9% of total body weight [41], so a Japanese arm will weigh about 2.5 kg. In the current study, therefore, about half of arm weight was reduced because the force reduced by the robot was set between 1200 and 1500 g. However, we do not know whether this weight relief is optimal for rehabilitation of the paretic arm. Accordingly, the appropriate amount of support should be examined in the future for patients with hemiparesis, in the same way as it was examined by Coscia et al. with healthy subjects [42].
“Tonic vibration reflex increases with the initial muscle contraction and increases with vibration frequency up to 100–150 Hz but decreases beyond” [43]. Thus, in the current study, FVS was used with an amplitude of approximately 100 Hz and was applied to muscles of the affected side only while the patient was reaching for the target. FVS might therefore be able to decrease excessive spasticity and promote agonist-muscle contraction.
High-intensity and task-specific training are considered important for stroke rehabilitation [5, 44, 45]. Robot-assisted therapy contributes to high-intensity practice due either to an increase in number of repetitions [46] or to the robot being added to regular practice [9], or to both. The frequency of repetitions of reaching movements varied from patient to patient, ranging from 200 to 700 per 15-min session. Although the frequency was smaller than in previous studies [36, 47, 48], the increases in outcome measures that we observed suggest that our lower frequency of repetitions was not inferior to that in other studies. Thus, our reaching robot might enable a participant to experience an efficient and effective rehabilitation exercise in 2 weeks of intervention. In the same way, the duration of intervention per day could be kept short. However, the most appropriate intensity, as to the repetitive numbers and the training time, should be reconsidered in a future study.
As we describe and show in Table 4 and Fig. 3, certain aspects of kinematic analysis were defined in a previous review concerning robotic rehabilitation of the paretic upper limb and assessment of movement quality [49]. According to this review, “temporal efficiency”, the time required to perform the movement, is expected to decrease with recovery. The current results revealed a tendency towards reductions in movement time. Furthermore, the review noted that “efficient movement” implies the shortest possible trajectory. In the current study, trajectory length after intervention tended to be shorter than that before intervention.
The exact mechanism of motor improvement induced by robotic rehabilitation has not yet been generally understood. Kinematic analysis is the preferred method to quantify improvement in motor performance and to elucidate the mechanism of motor learning in robot-assisted rehabilitation [8]. While many indicators have been proposed for kinematic analysis for the hemiparetic upper limb after stroke, active range of motion (AROM), movement time, and trajectory length are frequently described, although specifics of their use differ slightly [50,51,52,53]. Previous studies in which paretic upper limb kinematics were assessed during a movement task have reported a tendency for AROM in the elbow joint to increase and movement time to decrease [50,51,52,53]. Furthermore, trunk compensation with paretic upper-limb movement was assessed in previous studies [52, 54], although a consistent conclusion has not been established. We obtained results that are similar in several ways to results of previous studies, as follows.
An increase in AROM of the elbow was considered to indicate improved movement efficacy, and a decrease in movement time suggests improved efficiency in the paretic upper limb [49].
The trajectory length traced by the wrist decreased in five out of six patients (Table 3). In previous studies, an indicator of trajectory was often used to express movement efficiency or accuracy [55]. In the current study, we considered a decreased wrist trajectory length to signify improvement of movement efficiency facilitated by a reduction of wasted effort. Only patient 6 showed an increase in trajectory length (see in Table 3), but we do not consider it a worsening of performance. We speculate that it is because the patient had more severe paresis (UE-FMA scored 21) than the other five patients, so the task performance was insufficient before the intervention. As this patient’s AROM increased after intervention (as seen in Table 2), it might result in a longer path length than before intervention.
A reduction in the trajectory length traced by the shoulder and the forward displacement of the shoulder (Fig. 3, Table 4) were considered as a decrease in trunk compensation during the reaching movement. Although statistically significant changes were not seen in the current results, patients 2, 3, and 4 showed a reduction in trunk compensation. Such reduction might indicate improvement in movement efficacy.
Limitations of this study are the small sample size and lack of a control group. The small number of subjects prevents us from generalizing our results to all patients with stroke and hemiplegia. An improvement in the patients has been reported here, but whether it was due to exercise with the robot, to the regular occupational therapies, or to the combination of both is not clear because there was no control group. Given the overall function of the paretic upper-extremity clinically—the improvement of proximal and distal function—the machine was relatively focused on improving proximal function. When planning the current feasibility study, therefore, we had thought that regular occupational therapy, which primarily focuses on enhancing distal function (i.e., hand dexterity), would need to be added to the robotic training. In addition, the time between stroke onset and enrollment in the study was quite long. Patient 2, with the longest elapsed time (about 12 years) did not show improvement in ARAT score (see in Table 1). However, a feasibility study could benefit by being conducted with a range of patient conditions. Furthermore, the effect of arm-weight support, NMES, and FVS on recovery of upper-limb function was not proven in this study, because we utilized a before-and-after design during only 2 weeks of intervention due to limited length of stay in the hospital, and the intervention included regular occupational therapy for 40 min per day. A future study, accordingly, needs to address the effect of the three components individually or in combination with a larger RCT design and with longer intervention and follow-up periods. In addition, it is not yet clear which indicator of kinematic analysis best suggests improvement in upper limb movement. However, the parameters we used have been employed widely in previous studies. Parameters related to speed and acceleration, such as the normalized jerk and number of velocity peaks that are often used as the standard approach for evaluating smoothness [56, 57], should be incorporated in kinematic analysis to allow further understanding of the mechanism of motor improvement induced by robot-assisted rehabilitation. A future study should recruit larger numbers of participants and define distinct indicators of kinematic analysis.
Conclusion
Our robot-assisted modality is feasible and our results suggest that it might improve motor control and motor function of the hemiparetic upper extremity in patients who suffered chronic stroke. Training with this robot might enhance AROM of the elbow and decrease compensatory trunk movement, thus contributing to movement efficacy and efficiency.
Methods
Subjects
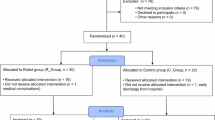
Subjects were recruited from patients admitted to the Kagoshima University Hospital Kirishima Rehabilitation Center, Japan, from June 30, 2015 to May 12, 2017. Inclusion criteria were as follows: (1) age between 20 and 80 years; (2) hemiparesis of the upper extremity with a diagnosis of first-time stroke (hemorrhage or infarction); (3) at least 24 weeks after onset of unilateral cerebral hemisphere stroke; (4) Brunnstrom recovery stage (BRS) of the upper extremity [58] ≥ 4; (5) ability to sit in a chair without assistance for 15 min; (6) ability to reach 10 cm both sagittally and vertically from the starting position with the affected upper extremity. Specifically, the starting position was set to be front of the patient, along with the sagittal direction between the patient’s navel and two buttons of the robot. In addition, the start button was set 10 cm away from the patient’s navel with the height at the navel, and the target button was set 10 cm farther from the start button in a sagittal line with the height at 10 cm above the start button. Exclusion criteria were as follows: (1) any medical condition for which electrical stimulation and vibration are contraindicated; (2) severe contractures, pain, or sensory disturbance of the affected upper extremity; (3) cognitive disorders that would interfere with understanding the procedure; (4) any other neurological or orthopedic diseases; (5) addition or dosage change—during the intervention—of any of the following prescription medicines that could influence upper limb function and performance: anti-spasticity drugs, dopaminergic drugs, antidopaminergic drugs, benzodiazepines and donepezil hydrochloride; (6) concurrently receiving botulinum injections (within 4 months), transcranial magnetic stimulations, or transcranial direct current stimulations.
Six patients with stroke (mean age, 58.2 years [SD 12.3]; range 38–66; 4 men and 2 women) met the inclusion criteria and were assigned to this study. All of the patients in this study had been diagnosed with cerebral infarction (2 subcortical, 2 basal ganglia, 1 capsula interna, 1 corona radiata). The mean time after stroke onset was 176 weeks (SD 208.5; range 38–593). Characteristics of participants are presented in Table 5.
Study design
This study utilized a before-and-after pilot design to examine feasibility and effectiveness of the reaching robot. Patients who met the inclusion criteria and did not fail the exclusion criteria received reaching exercise with the robot for 2 weeks at 15 min per day in addition to regular occupational therapy for 40 min per day.
Intervention
A reaching robot (Yaskawa Electric Co., Ltd. Fukuoka, Japan) with servo motor-controlled arm-weight support and concomitant electrical stimulation and vibration was employed in the intervention. This system (Fig. 4) has two video cameras on its upper frame for recording the patient’s movement. Features of this robot include (1) adjustment of the height, distance, and direction of the target button within reaching area, (2) adjustment of the amount of weight support for the arm (range 500–2500 g), and (3) facilitation with NMES and FVS to agonist muscles. The robot used in the current study is a prototype of a commercial version “Arm Rehabilitation Robot” (CoCoroe AR2®, Yaskawa Electric Co., Ltd., Fukuoka, Japan). However, the commercial version is not equipped with video cameras.
Participants performed reaching exercises in the horizontal and vertical planes while seated in a chair with a backrest, with their hip joints and knee joints flexed at 90 degrees. The seat height was adjusted to the participant’s lower leg length. The table height was adjusted to the level of the participant’s navel, which is the same height as the start button. The target button was in the sagittal direction; the height and the distance of the target button were determined by the occupational therapist. The participant’s arm was supported by the forearm cuff and the wire connected to the servo motor-controlled arm-weight support system. The primary functional elements of this robot are illustrated in Fig. 5.
Setting for training with the robot. A wire (a) connecting the device to the forearm cuff adjusts the amount of arm-weight support. The patient repeats a reaching movement from the start button (b) to the target button (c), accompanied with the arm-weight support, electrical stimulation (d), and vibratory stimulation (e). Two video cameras (f) on the upper frame of the device record the reaching movement for kinematic analysis
The amount of support (grams, g) was set for each participant by the occupational therapist in charge of the patient to allow a “comfortable” repetitive reaching exercise during 15 min. The amount of arm-weight support (i.e., extent of causing the arm to move upward and forward) could be adjusted “assist-as-needed” during reaching to the target button (forward path) and returning to the start button (backward path). As a result, the amount of weight support was approximately 1200 to 1500 g during reaching to the target button (forward path), and 700 to 900 g during returning to the start button (backward path). NMES was applied to the anterior part of the deltoid muscle and the triceps brachii muscle via a pair of surface electrodes, and FVS was applied with a vibrating motor (Fig. 5).
NMES (ITO ESPURGE, Ito Co., Ltd. Tokyo) was set to continuous mode, and the stimulation pulse was a symmetrical biphasic waveform with pulse width 250 μs and frequency 50 Hz. Intensity of the electrical current was adjusted to achieve slight contraction of the agonist muscle without visible movements of the limb or joint [14]. FVS (Vibrating motor: FM34F, Tokyo Parts Industrial Co., Ltd. Isesaki, Japan) was set to work only during the reaching movement (forward path; in other words, while the patient was extending the arm from start to target button); the vibrating motor would be switched on when the start button was pressed and switched off when the target button was pressed. Frequency of vibration was fixed to approximately 100 Hz. Participants were instructed to perform the reaching exercise at their preferred speed under the supervision of the occupational therapist. Patients received robotic training for 15 min and regular occupational therapy for 40 min per day for 2 weeks by the same occupational therapist in charge of each patient. Regular occupational therapy is primarily focused on hand dexterity, not on shoulder or elbow active movement. This is to confirm the hypothesis that the current reaching-robotic intervention, primarily focused on shoulder and elbow function, improves the ability to flex and extend the shoulder and elbow joints.
Outcome measures
Clinical measures
Outcome measures were assessed before and after 2 weeks of intervention. A trained and experienced therapist, who had no connection with the trial, evaluated all clinical measures. The upper extremity component of the Fugl-Meyer Assessment (UE-FMA) indicates the extent of motor control development [59], with a score consisting of two parts: a wrist–hand score (range 0–24) and a shoulder–elbow score including coordination (range 0–42). The maximum UE-FMA score (66) signifies optimal recovery.
The Action Research Arm Test (ARAT) is an assessment to evaluate arm motor function [60]. It includes 19 items divided into 4 subscales: grasp, grip, pinch, and gross movement. Each item is scored from 0 to 3, with zero indicating no movement and 3 indicating normal movement. The maximum score (57) indicates optimal performance.
The modified Ashworth Scale (MAS) is used to assess the muscle tone [61] in the biceps brachii, wrist flexor, and flexor digitorum muscles. For data analysis, the MAS scores (0, 1, 1+, 2, 3, and 4) were assigned numerical values designated as “computed MAS scores” (0, 1, 2, 3, 4, and 5, respectively) [62].
Kinematic analysis
Kinematic analysis was carried out to evaluate the quality of the reaching movement. Two video cameras were set on the upper frame of the reaching robot system, on the upper right and upper left in front of the patients. Reaching movements with the paretic upper limbs were recorded with a three-dimensional motion analysis system with KinemaTracer (KISSEI COMTEC Co., Ltd. Matsumoto, Japan) before and after the 2-week intervention. For assessing individual and intra-joint movements, reflective markers were placed on the acromion process, the lateral epicondyle, and the radial styloid process representing the shoulder, elbow, and wrist movements, respectively. This protocol is similar to those of previous studies (only 3 markers on the limb) [63,64,65], even though the definition of the joint angular is different.
The reaching movement was defined as a continuous movement from the start button to the target button (forward path), and from the target button back to the start button (backward path). The elbow extension during the reaching movement was defined as the angle between the vector of the acromion process and the lateral epicondyle, and the vector of the lateral epicondyle and the radial styloid process. The movements of shoulder and wrist were postulated as the movement of the acromion process and the radial styloid process, respectively.
For the assessment of kinematic analysis before and after intervention, each patient was required to perform the task movement—reaching movement between the start button and the target button—about five times without using the beneficial components of this robot (support of arm weight, NMES, and FVS). The target button was placed in five different positions: 10 cm, 20 cm, and 30 cm away (3 positions) from the start button in the sagittal and vertical directions, and 30 cm away in the sagittal, vertical, and horizontal directions (2 positions)—namely, in the ipsilateral and contralateral workspace, respectively (Fig. 6). The measurement was terminated if the patient could not touch the target button even once. As a reference, the same task was conducted with the non-paretic upper extremity once during the intervention period.
Five different positions for the start button (blue) and target button (yellow) (a–e) during each of 5 tasks for the kinematic analysis before and after 2 weeks intervention. The target button is set 10 cm (a), 20 cm (b), or 30 cm (c) in the sagittal and vertical directions, seen from the left side. In the ipsilateral and contralateral conditions (d) and (e), the target button is set 30 cm away in the sagittal, vertical, and horizontal directions. Ipsilateral here means the same side as the affected limb in the patient
Kinematic outcome variables during reaching movement included time of reaching movement, active range of motion (AROM) in terms of elbow angle (degrees), and trajectory lengths traced by wrist and shoulder. The AROM of the elbow was calculated as the difference between the maximum and minimum angular degrees. Shapes of trajectories for the shoulder (acromion process) and the wrist (radial styloid process) were also described. The trajectory is described as a figure seen from the lateral side, in coordinates with the vertical axis as height and the horizontal axis as forward length. The shape of the trajectory is presented as a standardized thick line and area including one standard deviation (SD), which was calculated from 4 to 6 consecutive reaching movements in all of the five conditions (the number of consecutive reaching movements differed by patient). The trajectory length was defined as the average traveling length of the wrist or the shoulder during one round trip between the start button and the target button. The trajectory length was applied to indicate smoothness of movement; the shapes of the trajectories were used to compare change of movement patterns. Forward displacement of the shoulder was measured as the distance along the horizontal axis in the Lissajous figure. Trajectory length for the shoulder and forward displacement of the shoulder can be considered as compensation via the trunk to supplement inadequate movement of the upper limb; alternatively, it might reflect a change of movement pattern as a strategy to achieve the reaching movement.
Statistical analysis
All variables are summarized as mean and SD or SE. The UE-FMA, ARAT, and MAS scores were assessed before and immediately after 2 weeks intervention. In the same way, AROM of elbow flexion angle (degrees) and trajectory lengths with wrist and shoulder joints were assessed using kinematic analysis. All of the clinical and kinematic data were analyzed with the Wilcoxon signed-rank test to compare pre-treatment data with post-treatment data. Statistical analysis was performed with the Statistical Package for the Social Sciences (SPSS; SPSS Inc. Chicago, IL, version 18.0 for Windows), and a p value of < 0.05 was considered to indicate statistical significance. Effect size (ES) was calculated as r = Z/√ N, using the Z score.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- UE-FMA:
-
Fugl-Meyer Assessment
- ARAT:
-
Action Research Arm Test
- MAS:
-
Modified Ashworth Scale
- UMIN-CTR:
-
University hospital Medical Information Network Clinical Trial Registry
- NMES:
-
Neuromuscular electrical stimulation
- FVS:
-
Functional vibratory stimulation
- BRS:
-
Brunnstrom recovery stage
- AROM:
-
Active range of motion
- CI:
-
Cerebral infarction
- EMG:
-
Electromyography
References
https://www.mhlw.go.jp/toukei/saikin/hw/kanja/17/index.html. 2019. Accessed June 30 2019.
https://www.mhlw.go.jp/toukei/saikin/hw/jinkou/geppo/nengai17/index.html. 2018. Accessed June 30 2019.
https://www.mhlw.go.jp/toukei/saikin/hw/k-tyosa/k-tyosa16/index.html. 2017. Accessed June 30 2019.
Hendricks HT, van Limbeek J, Geurts AC, Zwarts MJ. Motor recovery after stroke: a systematic review of the literature. Arch Phys Med Rehabil. 2002;83:1629–37.
Langhorne P, Bernhardt J, Kwakkel G. Stroke rehabilitation. Lancet. 2011;377:1693–702.
Schmidt RA. A schema theory of discrete motor skill learning. Psychol Rev. 1975;82:225–60.
Morasso P, Casadio M, Giannoni P, Masia L, Sanguineti V, Squeri V, et al. Desirable features of a “humanoid” robot-therapist. Conf Proc IEEE Eng Med Biol Soc. 2009;2009:2418–21.
Veerbeek JM, Langbroek-Amersfoort AC, van Wegen EE, Meskers CG, Kwakkel G. Effects of robot-assisted therapy for the upper limb after stroke: a systematic review and meta-analysis. Neurorehabil Neural Repair. 2017;31:107–21.
Norouzi-Gheidari N, Archambault PS, Fung J. Effects of robot-assisted therapy on stroke rehabilitation in upper limbs: systematic review and meta-analysis of the literature. J Rehabil Res Dev. 2012;49:479–96.
Mehrholz J, Pohl M, Platz T, Kugler J, Elsner B. Electromechanical and robot-assisted arm training for improving activities of daily living, arm function, and arm muscle strength after stroke. Cochrane Database Syst Rev. 2018. https://doi.org/10.1002/14651858.
Hsu SS, Hu MH, Wang YH, Yip PK, Chiu JW, Hsieh CL. Dose-response relation between neuromuscular electrical stimulation and upper-extremity function in patients with stroke. Stroke. 2010;41:821–4.
Noma T, Matsumoto S, Shimodozono M, Iwase Y, Kawahira K. Novel neuromuscular electrical stimulation system for the upper limbs in chronic stroke patients: a feasibility study. Am J Phys Med Rehabil. 2014;93:503–10.
Miyasaka H, Orand A, Ohnishi H, Tanino G, Takeda K, Sonoda S. Ability of electrical stimulation therapy to improve the effectiveness of robotic training for paretic upper limbs in patients with stroke. Med Eng Phys. 2016;38:1172–5.
Shimodozono M, Noma T, Matsumoto S, Miyata R, Etoh S, Kawahira K. Repetitive facilitative exercise under continuous electrical stimulation for severe arm impairment after sub-acute stroke: a randomized controlled pilot study. Brain Inj. 2014;28:203–10.
Yang JD, Liao CD, Huang SW, Tam KW, Liou TH, Lee YH, et al. Effectiveness of electrical stimulation therapy in improving arm function after stroke: a systematic review and a meta-analysis of randomised controlled trials. Clin Rehabil. 2019;33:1286–97.
Murillo N, Valls-Sole J, Vidal J, Opisso E, Medina J, Kumru H. Focal vibration in neurorehabilitation. Eur J Phys Rehabil Med. 2014;50:231–42.
Noma T, Matsumoto S, Shimodozono M, Etoh S, Kawahira K. Anti-spastic effects of the direct application of vibratory stimuli to the spastic muscles of hemiplegic limbs in post-stroke patients: a proof-of-principle study. J Rehabil Med. 2012;44:325–30.
Tavernese E, Paoloni M, Mangone M, Mandic V, Sale P, Franceschini M, et al. Segmental muscle vibration improves reaching movement in patients with chronic stroke. A randomized controlled trial. NeuroRehabilitation. 2013;32:591–9.
Kawahira K, Higashihara K, Matsumoto S, Shimodozono M, Etoh S, Tanaka N, Sueyoshi Y. New functional vibratory stimulation device for extremities in patients with stroke. Int J Rehabil Res. 2004;27:335–7.
Rosenkranz K, Pesenti A, Paulus W, Tergau F. Focal reduction of intracortical inhibition in the motor cortex by selective proprioceptive stimulation. Exp Brain Res. 2003;149:9–16.
Rosenkranz K, Rothwell JC. Differential effect of muscle vibration on intracortical inhibitory circuits in humans. J Physiol. 2003;551:649–60.
De Gail P, Lance JW, Neilson PD. Differential effects on tonic and phasic reflex mechanisms produced by vibration of muscles in man. J Neurol Neurosurg Psychiatry. 1966;29:1–11.
Ribot-Ciscar E, Butler JE, Thomas CK. Facilitation of triceps brachii muscle contraction by tendon vibration after chronic cervical spinal cord injury. J Appl Physiol. 2003;94:2358–67.
Roll JP, Vedel JP. Kinaesthetic role of muscle afferents in man, studied by tendon vibration and microneurography. Exp Brain Res. 1982;47:177–90.
Roll JP, Vedel JP, Ribot E. Alteration of proprioceptive messages induced by tendon vibration in man: a microneurographic study. Exp Brain Res. 1989;76:213–22.
Cotey D, Hornby TG, Gordon KE, Schmit BD. Increases in muscle activity produced by vibration of the thigh muscles during locomotion in chronic human spinal cord injury. Exp Brain Res. 2009;196:361–74.
Magalhães FH, Kohn AF. Vibration-induced extra torque during electrically-evoked contractions of the human calf muscles. J Neuroeng Rehabil. 2010;7:26.
Kawahira K, Tsujio S, Sueyoshi Y. VIBRATION STIMULATION THERAPY APPARATUS, ITS USE METHOD, AND COMPUTER PROGRAM. Patent. JP 4852741 (WO2006134999). 2011. https://jstore.jst.go.jp/nationalPatentDetail.html?pat_id=17909 https://patentscope2.wipo.int/search/ja/detail.jsf?docId=WO2006134999&tab=PCTBIBLIO&_cid=JP1-K5N59I-75957-1. Accessed Jan 23 2020.
Kawabe M, Kitano T, Misu K, Sadakane K, Tomisaki H, Kawahira K, Shimodozono M, Hayashi R. TRAINING DEVICE. Patent. JP 6210363 (WO2014189046). 2017. https://ipforce.jp/patent-jp-B9-6210363 (https://patentscope2.wipo.int/search/en/detail.jsf?docId=WO2014189046). Accessed Jan 23 2020.
Misu K, Akae H, Kitano T, Kawabe M, Tomisaki H, Kawahira K, Shimodozono M, Yu Y, Hayashi R. EXERCISE APPARATUS. Patent. JP 6210364 (WO2014189032). 2017. https://ipforce.jp/patent-jp-B9-6210364 (https://patentscope2.wipo.int/search/en/detail.jsf?docId=WO2014189032). Accessed Jan 23 2020.
Hoei T, Kawahira K, Fukuda H, Sihgenobu K, Shimodozono M, Ogura T. Use of an arm weight-bearing combined with upper-limb reaching apparatus to facilitate motor paralysis recovery in an incomplete spinal cord injury patient: a single case report. J Phys Ther Sci. 2017;29:176–80.
See J, Dodakian L, Chou C, Chan V, McKenzie A, Reinkensmeyer DJ, et al. A standardized approach to the Fugl-Meyer assessment and its implications for clinical trials. Neurorehabil Neural Repair. 2013;27:732–41.
Rodgers H, Bosomworth H, Krebs HI, van Wijck F, Howel D, Wilson N, et al. Robot assisted training for the upper limb after stroke (RATULS): a multicentre randomised controlled trial. Lancet. 2019;394:51–62.
Klamroth-Marganska V, Blanco J, Campen K, Curt A, Dietz V, Ettlin T, et al. Three-dimensional, task-specific robot therapy of the arm after stroke: a multicentre, parallel-group randomised trial. Lancet Neurol. 2014;13:159–66.
Volpe BT, Lynch D, Rykman-Berland A, Ferraro M, Galgano M, Hogan N, et al. Intensive sensorimotor arm training mediated by therapist or robot improves hemiparesis in patients with chronic stroke. Neurorehabil Neural Repair. 2008;22:305–10.
Lo AC, Guarino PD, Richards LG, Haselkorn JK, Wittenberg GF, Federman DG, et al. Robot-assisted therapy for long-term upper-limb impairment after stroke. N Engl J Med. 2010;362:1772–83.
Van der Lee JH, Wagenaar RC, Lankhorst GJ, Vogelaar TW, Devillé WL, Bouter LM. Forced use of the upper extremity in chronic stroke patients: results from a single-blind randomized clinical trial. Stroke. 1999;30:2369–75.
Carpinella I, Cattaneo D, Ferrarin M. Quantitative assessment of upper limb motor function in Multiple Sclerosis using an instrumented Action Research Arm Test. J Neuroeng Rehabil. 2014;11:67.
Jordan K, Sampson M, King M. Gravity-supported exercise with computer gaming improves arm function in chronic stroke. Arch Phys Med Rehabil. 2014;95:1484–9.
Krabben T, Prange GB, Molier BI, Stienen AH, Jannink MJ, Buurke JH, et al. Influence of gravity compensation training on synergistic movement patterns of the upper extremity after stroke, a pilot study. J Neuroeng Rehabil. 2012. https://doi.org/10.1186/1743-0003-9-44.
Clauser CE, McConville JT, Young JW. Weight, volume, and center of mass of segments of the human body. AMRL-TR-69-70, Aerospace Medical Research Laboratory, Wright-Patterson Air Force Base, Ohio, 1969.
Coscia M, Cheung VC, Tropea P, Koenig A, Monaco V, Bennis C, et al. The effect of arm weight support on upper limb muscle synergies during reaching movements. J Neuroeng Rehabil. 2014;11:22.
Park HS, Martin BJ. Contribution of the tonic vibration reflex to muscle stress and muscle fatigue. Scand J Work Environ Health. 1993;19:35–42.
Kawahira K, Shimodozono M, Etoh S, Kamada K, Noma T, Tanaka N. Effects of intensive repetition of a new facilitation technique on motor functional recovery of the hemiplegic upper limb and hand. Brain Inj. 2010;24:1202–13.
Shimodozono M, Noma T, Nomoto Y, Hisamatsu N, Kamada K, Miyata R, et al. Benefits of a repetitive facilitative exercise program for the upper paretic extremity after subacute stroke: a randomized controlled trial. Neurorehabil Neural Repair. 2013;27:296–305.
Boninger ML, Wechsler LR, Stein J. Robotics, stem cells, and brain-computer interfaces (BCI) in rehabilitation and recovery from stroke: updates and advances. Am J Phys Med Rehabil. 2014;93(Suppl 3):145–54.
Sale P, Franceschini M, Mazzoleni S, Palma E, Agosti M, Posteraro F. Effects of upper limb robot-assisted therapy on motor recovery in subacute stroke patients. J Neuroeng Rehabil. 2014. https://doi.org/10.1186/1743-0003-11-104.
Conroy SS, Whitall J, Dipietro L, Jones-Lush LM, Zhan M, Finley MA, et al. Effect of gravity on robot-assisted motor training after chronic stroke: a randomized trial. Arch Phys Med Rehabil. 2011;92:1754–61.
Nordin N, Xie SQ, Wünsche B. Assessment of movement quality in robot- assisted upper limb rehabilitation after stroke: a review. J Neuroeng Rehabil. 2014. https://doi.org/10.1186/1743-0003-11-137.
Kamper DG, McKenna-Cole AN, Kahn LE, Reinkensmeyer DJ. Alterations in reaching after stroke and their relation to movement direction and impairment severity. Arch Phys Med Rehabil. 2002;83:702–7.
Aprile I, Rabuffetti M, Padua L, Di Sipio E, Simbolotti C, Ferrarin M. Kinematic analysis of the upper limb motor strategies in stroke patients as a tool towards advanced neurorehabilitation strategies: a preliminary study. Biomed Res Int. 2014. https://doi.org/10.1155/2014/636123.
Hsieh YW, Liing RJ, Lin KC, Wu CY, Liou TH, Lin JC, et al. Sequencing bilateral robot-assisted arm therapy and constraint-induced therapy improves reach to press and trunk kinematics in patients with stroke. J Neuroeng Rehabil. 2016. https://doi.org/10.1186/s12984-016-0138-5.
Bartolo M, De Nunzio AM, Sebastiano F, Spicciato F, Tortola P, Nilsson J, et al. Arm weight support training improves functional motor outcome and movement smoothness after stroke. Funct Neurol. 2014;29:15–21.
Thielman GT, Dean CM, Gentile AM. Rehabilitation of reaching after stroke: task-related training versus progressive resistive exercise. Arch Phys Med Rehabil. 2004;85:1613–8.
Colombo R, Pisano F, Micera S, Mazzone A, Delconte C, Carrozza MC, et al. Assessing mechanisms of recovery during robot-aided neurorehabilitation of the upper limb. Neurorehabil Neural Repair. 2008;22:50–63.
Teulings HL, Contreras-Vidal JL, Stelmach GE, Adler CH. Parkinsonism reduces coordination of fingers, wrist, and arm in fine motor control. Exp Neurol. 1997;146:159–70.
Buma FE, van Kordelaar J, Raemaekers M, van Wegen EEH, Ramsey NF, Kwakkel G. Brain activation is related to smoothness of upper limb movements after stroke. Exp Brain Res. 2016;234:2077–89.
Brunnstrom S. Motor testing procedures in hemiplegia: based on sequential recovery stages. Phys Ther. 1966;46:357–75.
Fugl-Meyer AR, Jääskö L, Leyman I, Olsson S, Steglind S. The post-stroke hemiplegic patient. 1. A method for evaluation of physical performance. Scand J Rehabil Med. 1975;7:13–31.
Yozbatiran N, Der-Yeghiaian L, Cramer SC. A standardized approach to performing the Action Research Arm Test. Neurorehabil Neural Repair. 2008;22:78–90.
Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther. 1987;67:206–7.
Chua KS, Kong KH, Lui YC. Botulinum toxin A in the treatment of hemiplegic spastic foot drop-clinical and functional outcomes. Singapore Med J. 2000;41:209–13.
Scano A, Chiavenna A, Malosio M, Molinari TL, Molteni F. Robotic assistance for upper limbs may induce slight changes in motor modules compared with free movements in stroke survivors: a cluster-based muscle synergy analysis. Front Hum Neurosci. 2018;12:290.
Antonella B, Alessandro S, Andrea C, Alfonso M, Simona MS, Simone P, et al. A multiparameter approach to evaluate post-stroke patients: an application on robotic rehabilitation. Appl Sci. 2018;8:2248. https://doi.org/10.3390/app8112248.
Caimmi M, Chiavenna A, Scano A, Gasperini G, Giovanzana C, Molinari TL, et al. Using robot fully assisted functional movements in upper-limb rehabilitation of chronic stroke patients: preliminary results. Eur J Phys Rehabil Med. 2017;53:390–9.
Acknowledgements
None.
Funding
This work was supported in part by Yaskawa Electric Co., Ltd. Fukuoka, Japan.
Author information
Authors and Affiliations
Contributions
YA, TN, and MS designed the study and interpreted the data; YA and TN collected and analyzed the data. YA wrote the manuscript; MS, SE, RM, and KK revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The procedures used in this study complied with the Declaration of Helsinki 1975, as revised in 2013. After the study protocol was explained to prospective participants, written informed consent was obtained according to the ethical guidelines of the hospital. The study was carried out with the approval of the ethics committee of Kagoshima University (26-209) and was registered with UMIN-CTR (UMIN000018132).
Consent for publication
Not applicable.
Competing interests
This work was supported in part by Yaskawa Electric Co., Ltd. Fukuoka, Japan. Dr. Shimodozono has received lecture fees and consulting fees from Yaskawa Electric Co. The other authors report no conflicts.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Amano, Y., Noma, T., Etoh, S. et al. Reaching exercise for chronic paretic upper extremity after stroke using a novel rehabilitation robot with arm-weight support and concomitant electrical stimulation and vibration: before-and-after feasibility trial. BioMed Eng OnLine 19, 28 (2020). https://doi.org/10.1186/s12938-020-00774-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12938-020-00774-3