Abstract
Background
Coccidiosis is a poultry disease that occurs worldwide and is caused by Eimeria species. The infection is associated with reduced feed efficiency, body weight gain, and egg production. This study aimed to investigate the current status of coccidiosis and anticoccidial resistance to anticoccidial drugs used as part of control strategies for this disease in Korean chicken farms.
Results
An overall prevalence of 75% (291/388) was found. Positive farms contained several Eimeria species (mean = 4.2). Of the positive samples, E. acervulina (98.6%), E. maxima (84.8%), and E. tenella (82.8%) were the most prevalent species. Compared with cage-fed chickens, broilers and native chickens reared in free-range management were more at risk of acquiring an Eimeria infection. Sensitivities to six anticoccidial drugs (clopidol, diclazuril, maduramycin, monensin, salinomycin, and toltrazuril) were tested using nine field samples. Compared with untreated healthy control chickens, the body weight gains of infected chickens and treated/infected chickens were significantly reduced in all groups. Fecal oocyst shedding was significantly reduced in four clopidol-treated/infected groups, three diclazuril-treated/infected groups, two toltrazuril-treated/infected groups, one monensin-treated/infected group, and one salinomycin-treated/infected group, compared with the respective untreated/infected control groups. Intestinal lesion scores were also reduced in three clopidol-treated/infected groups, one monensin-treated/infected group, and one toltrazuril-treated/infected group. However, an overall assessment using the anticoccidial index, percent optimum anticoccidial activity, relative oocyst production, and reduced lesion score index found that all field samples had strong resistance to all tested anticoccidial drugs.
Conclusion
The results of this large-scale epidemiological investigation and anticoccidial sensitivity testing showed a high prevalence of coccidiosis and the presence of severe drug resistant Eimeria species in the field. These findings will be useful for optimizing the control of coccidiosis in the poultry industry.
Similar content being viewed by others
Background
The meat and eggs from poultry are significant sources of protein. Chickens are the most essential poultry species globally; they constitute about 90% of the poultry population worldwide [1]. Coccidiosis is a major cause of immunosuppression in poultry. It is identified by the American Association of Avian Pathologists as one of the top diseases of concern affecting broiler and layer farms [2]. This disease occurs worldwide and is caused by any one of seven Eimeria species. Eimeria is an intracellular parasite that invades the intestinal epithelial cells of chickens. Infection that progresses to disease causes mortality and production losses associated with reduced feed efficiency, body weight gain, and egg production in affected chickens [3,4,5].
Coccidiosis is endemic in most tropical and subtropical regions where farm management practices mostly include use of deep litter that creates environmental conditions that favor year-round propagation of Eimeria species. In practice, increasing use of intensive farming systems and the associated high stocking densities on farms increases the likelihood of disease persistence. Based on 2016 prices, the estimated global cost of coccidiosis control is £10.36 billion [6]. Consistent year-by-year financial pressures on the poultry industry are linked to prophylactic use of anticoccidial drugs (e.g., ionophores and synthetic chemicals) in feed or water [7]. However, development of drug resistance over time in different parts of the world have reduced efficacy of anticoccidials [8, 9]. To address the growing consumer demand for antibiotic-free poultry products and appearance of drug-resistant strains, regulations implemented to ban the use of antibiotics in food-producing animals, and natural dietary supplements and probiotics are being used as alternative coccidiosis control strategies [8, 10,11,12,13]. Low-dose virulent or attenuated vaccine strains of Eimeria are also used to control and prevent infection [4, 14,15,16,17]. However, there is little cross-protection between Eimeria species, and there are between-species differences in susceptibility to anticoccidial drugs [18, 19]. Therefore, it is crucial to accurately identify Eimeria species to select more effective vaccines and control measures.
In our previous report in 2010, the prevalence of coccidiosis was 78.7% in fecal samples from 356 poultry farms. By using a species-specific PCR diagnostic method based on the internal transcribed spacer 1 sequence, E. acervulina and E. tenella were the most common in the investigated farms with 87.5% and 62.5%, respectively, followed by E. brunetti and E. praecox (59.3%, and 37.5%, respectively). The other species such as E. maxima, E. necatrix, and E. mitis had a prevalence of 31.3% [20]. There have also been studies of the use of various natural substances to control coccidiosis, such as aloe vera [13], berberine [21], galla rhois [22] and green tea [23]. The objective of this study is to provide an up-to-date status of coccidiosis in Korea by investigating the occurrence of Eimeria infection in poultry farms and comparing the resistance of field Eimeria species to commercially available anticoccidial drugs. The resulting epidemiological data can be used to develop guidelines for control measures that are more appropriate to the current status of this disease.
Results
Prevalence and distribution of coccidiosis
Of the fecal samples collected from 388 chicken farms, 291 were positive for coccidian oocysts (overall prevalence = 75%). The specific rates (prevalence rates) in chicken types were determined for broilers (84.5%), native chickens (81.48%), and breeder and laying hens (33.3–42.6%). Similarly, prevalence rates in the management types of free-range chickens (83.0%) and caged chickens (41.3%) were identified (Table 1). The numbers of oocysts in fecal samples were < 50/g (25%), 50–999/g (18.8%), 1,000–9,999/g (27.1%), and ≥ 10,000 (29.1%) (Fig. 1A). The 90.2% and 96.4% of fecal samples containing ≥ 10,000 oocysts/g were from broilers and free-range chickens, respectively (Table 2). Positive fecal samples included multiple Eimeria species (mean = 4.2). There were 3 to approximately 5 Eimeria species in 77.5% of the positive samples (Fig. 1B). The most prevalent Eimeria species were E. acervulina (98.6%), E. maxima (84.8%), and E. tenella (82.8%), followed by E. mitis (42.9%), E. praecox (36.4%), E. brunetti (14%), and E. necatrix (8.8%) (Fig. 1C).
Risk factor analysis and coccidiosis infection status
Based on the specific rates of prevalence in chicken types (Table 1), broilers and native chickens were about 7.3 and 5.9 times, respectively, more at risk than laying hens. Free-range chickens were about 6.9 times more at risk than caged chickens (Table 1). With fecal samples containing ≥ 10,000 oocysts/g, broilers and native chickens were about 8.2 and 4.9 times, respectively, more at risk than laying hens. Free-range chickens were about 9.4 times more at risk than caged chickens (Table 2).
Analysis of fecal oocyst concentration indicated that most samples had < 50 oocysts/g (58.6%) in fecal samples obtained from caged chickens; most samples collected from free-range chickens had ≥ 10,000 oocysts/g (34.8%) (Fig. 2A). The mean number of Eimeria species was not different between free-range (4.8 ± 0.7) and caged chickens (3.9 ± 0.6) (Fig. 2B). In general, regardless of chicken breed and management type, E. acervulina and E. maxima were the most prevalent Eimeria species (Fig. 2C).
Status of coccidiosis based on level of infection and Eimeria species. A Level of infection based on the number of oocysts per gram of fecal samples. B Mean of numbers of Eimeria species present in positive samples. C Species-specific distribution of Eimeria species based on the source of fecal samples
Anticoccidial sensitivity test of field samples
Nine field samples (A–I) collected from different provinces were used for the anticoccidial experiment against six different drugs. Parameters such as body weight gain, oocyst production, and lesion score were measured (Figs. 3, 4 and 5). The nine field samples included the most prevalent Eimeria species (E. acervulina, E. maxima, and E. tenella) with 0 to 3 different Eimeria species depending on the sample (Additional file 1).
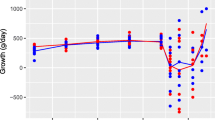
Comparison of body weight gain in field sample-infected chickens. Five-day-old ROSS 308 female chickens were orally infected with 3 × 104 sporulated oocysts of 9 different field samples (A–I). Body weight gain (n = 20) was measured at 9 days after infection. * P < 0.05, ** P < 0.01, and ***P < 0.001 indicate significant differences compared to untreated and healthy group (NC). The results represent mean ± SE values. NC, negative control; PC, untreated and infected group as a positive control; A-I, farm samples
Comparison of oocyst numbers in field sample-infected chickens. Five-day-old ROSS 308 female chickens were orally infected with 3 × 104 sporulated oocysts of 9 different field samples (A–I). Production of oocysts per group (n = 20) was obtained from the fecal samples collected from days 6 to 9 post-infection. * P < 0.05, ** P < 0.01, and ***P < 0.001 indicate significant differences compared to untreated and infected group (PC). The results represent mean ± SE values. NC, negative control; PC, untreated and infected group as a positive control; A-I, farm samples
Comparison of intestinal lesion scores in field sample-infected chickens. Five-day-old ROSS 308 female chickens were orally infected with 3 × 104 sporulated oocysts of 9 different field samples (A–I). At day 7 post-infection, five chickens from each group were randomly selected for intestinal lesion scoring. Lesions scores range from 0–4, following the Johnson and Reid [24] scoring technique. * P < 0.05 indicates significant difference compared to untreated and infected group (PC). Results represent mean ± SE values. NC, negative control; PC, untreated and infected group as a positive control; A-I, farm samples
Compared with untreated healthy control (NC) animals, body weight gains of untreated and infected chickens (PC) and treated/infected chickens were significantly reduced in all groups. There was no difference in body weight gain between PC and treated/infected chickens (Fig. 3). Fecal oocyst shedding was significantly reduced in 4 clopidol-treated/infected groups, 3 diclazuril-treated/infected groups, 2 toltrazuril-treated/infected groups, 1 monensin-treated/infected group, and 1 salinomycin-treated/infected group, compared with their respective PC groups. No oocysts were found in the NC groups (Fig. 4). Intestinal lesion scores were reduced in 3 clopidol-treated/infected groups, 1 monensin-treated/infected group, and 1 toltrazuril-treated/infected group. No lesion scores were found in the NC groups (Fig. 5).
Next, the four anticoccidial indexes, the ACI, POAA, ROP and RLS index, were evaluated with body weight gain, oocyst production and the lesion scores used above and survival rates (Additional file 2). The results for the ACI, POAA, ROP, and RLS values indicated that all field samples were resistant to all anticoccidial drugs (Additional files 3, 4, 5 and 6). Taken together, the results indicated that all field samples had severe resistance to all anticoccidial drugs tested (Table 3).
Discussion
Worldwide, coccidiosis is an economically significant disease in the poultry sector. Therefore, regular epidemiological monitoring of Eimeria species present in the field and up-to-date status of resistance to treatments are vital for the best selection of control and prevention strategies. Overall, this study found that the current prevalence of coccidiosis in Korea is 75% (291 of 388 farms). Compared with our laboratory's previous epidemiological findings of 78.7% (280 of the 356 farms) in 2010, this finding was lower, but generally higher than the coccidiosis prevalence values reported in South Ethiopia, Pakistan, Nigeria, Iran, India, Turkey, and Algeria of 20.10%, 23.8%, 31.8%, 35.2%, 39.58%, 54.3%, and 63.26%, respectively [1, 20, 25,26,27,28,29,30]. Factors including control methods used, timing of sampling, animal husbandry, and geographical variation can account for differences in prevalence, compared with previous studies [31]. But, comparing the previous results for South Korea to this study suggests there was little progress in control for the past 10 years.
In this study, the high prevalence of coccidiosis based on poultry type and management type was associated with broilers (84.5%) and native chickens (81.48%), particularly those raised using the free-range (83.0%) management (Table 1). While these findings are contrary to the results of a study in Nigeria, which found the layer chickens have a higher prevalence rate of coccidial infection than broilers, they are consistent with the findings in Jordan and Iran poultry industries [32,33,34]. The previous studies found that the high prevalence values of coccidiosis in broilers are associated with deep-litter management with high stocking densities and poor farm management practices [33, 34]. In Korea, free-range management includes use of deep litter with high stocking densities. This study found a high degree of association between disease occurrence and management type; chickens raised using a free-range type of management had a higher coccidiosis rate. The fecal–oral route is the mode of transmission of the infective state of Eimeria species (sporulated oocysts) [35]. Use of deep litter provides conditions that promote oocyst accumulation, sporulation, and persistence in the environment. Consequently, the rate of exposure or contact of chickens to Eimeria species increases via the contaminated litter or fomites. Coccidiosis prevalence in birds raised in wire cages is lower because fecal–oral transmission is essentially stopped or reduced [36].
To reflect current infection status in the field, collected fecal samples were further assessed by determining OPG values, the numbers of Eimeria species present in a single positive sample, and identification of the specific Eimeria species in fecal samples. First, monitoring oocyst number in fecal samples provides an index of the degree of infection and the parasite's rate of reproduction in the intestines [37]. Although the OPG values in litter varied widely between samples, high numbers of samples with oocyst numbers ≥ 10,000/g were consistently found in broilers and free-range chickens; 56.2% of the fecal samples contained ≥ 1,000 oocysts/g (Fig. 1A). This result suggested that there was a considerable amount of oocyst output in the field and that oocysts would continue to accumulate, sporulate, and infect more birds. It is interesting to note that even a mild infection of about 50 E. tenella or E. brunetti oocysts can yield several hundred thousand oocysts [38]. Although this OPG monitoring revealed estimates of infection levels, it did not collectively represent the course of infection in the flock; it only provided an estimate of the numbers of oocysts present at the time of sampling. Long and Rowell found that oocyst numbers generally peak at 4–5 weeks after the introduction of birds [39]. Thus, further studies of the course of infection should include flock age and information about litter recycling at the time of sampling.
Each species of Eimeria that infects poultry has a preferred site of infection in the bird's gastrointestinal tract. Lesions can be mild or severe, depending on the magnitude of infection. While some studies found single-species infections in flocks, coccidiosis mainly results from a mix of Eimeria species that parasitize different areas of the intestine [12, 40]. Consistent with this finding, the mean number of Eimeria present in a single sample was determined to demonstrate the degree of infection in the field and the Eimeria species present on the samples were identified to discriminate proper measures for control, as some Eimeria species are predisposing factors to necrotic enteritis [41]. This study found mixed populations of Eimeria species (mean = 4.2); 77.5% of the positive samples contained 3 to 5 Eimeria species in one fecal sample (Fig. 1B). This high prevalence of mixed infection in samples was consistent with findings of > 80% in Norway, 55% in Nigeria, and 54.28% in Algeria [25, 31, 42].
RT-PCR analysis of the positive fecal samples detected all Eimeria species in the fecal samples. Eimeria acervulina (98.6%), E. maxima (84.8%), and E. tenella (82.8%) were the most prevalent species. The finding that E. acervulina was the predominant Eimeria species was consistent with previous findings in South Korea, Southwestern Nigeria, and Romania [9, 20, 43]. However, it was not consistent with the results of studies of free-range chickens in Tunisia and broiler farms in Northern Jordan, where the prevalence of E. acervulina are 1.5% and 3%, respectively [33, 44]. The high prevalence of E. acervulina and E. tenella are likely attributable to the high oocyst production rates of both species while the detected high prevalence of E. maxima can be attributed to the high potential of this species to extensively affect a large portion of the intestine, compared with other species [45]. In this study, the findings for E. brunetti (14%) and E. necatrix (8.8%) were consistent with the finding of a low species prevalence in China, but were not consistent with findings in South Korea and India [19, 46, 47]. These differences may be associated with alterations in the population dynamics of the parasite brought about by the different reproductive potential of each Eimeria species [45]. Also, E. brunetti and E. necatrix are more common in older birds [48].
Compared with the results of a previous study in South Korea performed 10 years ago, this study found that the prevalence of coccidiosis remains high. The mean number of Eimeria species per sample increased from 3.4 to 4.2, accompanied by evidence of a change in population dynamics of the infecting species present in the field (Fig. 1). The varying resistance of Eimeria species to anticoccidials used in other countries is well-documented [42, 49, 50]. However, in South Korea, the drug-resistant strains of Eimeria species are poorly understood and documented. We also evaluated the sensitivities of nine field samples to anticoccidials. We used six anticoccidial drugs used in the field (clopidol, diclazuril, maduramycin, monensin, salinomycin, and toltrazuril). Parameters such as body weight gain, intestinal lesion scores, and oocyst production along with mortality rates were assessed. Treatments are considered protective when birds maintain weight gain and lesion scores are zero or at a low number [51]. Our trials found that all infected chickens had reduced body weight gain (Fig. 3), which is a clinical manifestation of coccidiosis. But, while oocyst production (Fig. 4) and lesion scores (Fig. 5) were reduced in some medicated groups, limited levels of reduction in lesion scores and oocyst production were found in most medicated groups. In general, assessment of drug efficacy based on a single parameter may result in inconsistent findings [52]. Thus, we used a standard method for an overall assessment of drug sensitivity (i.e., the ACI, POAA, ROP, and RLS indices). Overall, the results indicated that all field samples collected in this study were severely drug resistant to all anticoccidials evaluated, but particularly to diclazuril, maduramycin, and toltrazuril. Likewise, varying levels of drug resistance to clopidol [53], diclazuril [49, 54], maduramycin [55], monensin [56, 57], salinomycin [56,57,58], and toltrazuril [42, 49] were previously reported. Similar to the findings in this study, presence of field isolates with multi-drug resistance to anticoccidials has also been reported [49, 50, 59].
Chemical anticoccidials and ionophores generally negatively affect the metabolism and ion transport, respectively, of the parasites [35]. However, some anticoccidials are not entirely efficient in all the developmental stages of Eimeria species [18]. The development of Eimeria species resistance to ionophores is linked to a change in the biochemical composition of the parasite membrane [8]. In the field, husbandry practices that include the intensive use of anticoccidials is often the cause of development of resistance. The use of anticoccidials in Korea has decreased significantly, from about 52,612 kg in 2014 to 23,682 kg in 2019. However, the poultry industry still uses anticoccidials such as clopidol, diclazuril, maduramycin, monensin, and salinomycin as prophylactic medications [60].
Conclusion
Taken together, the results of this study indicated that the prevalence of coccidiosis in South Korea remains high, and that over time the population dynamics and infection levels of Eimeria species have changed. Furthermore, there are Eimeria species in the field with severe levels of multi-drug resistance to anticoccidials. Therefore, it is critical to look for alternative control methods that consider drug-sensitivity profiles of field samples found in this study. Periodic monitoring of resistance, field status, and prevalence of Eimeria species is critical, because knowing the patterns will be helpful for planning and implementation of an optimized control program.
Methods
Fecal sample collection and oocyst counting
Fecal samples were collected from 388 randomly selected poultry farms in Korea between December 2019 and May 2021. All sampled farms have no history with the use of coccidiosis vaccine in their programs. The 388 fecal samples were collected from 278 broiler farms, 61 laying hen farms, 27 native chicken farms, 12 broiler breeder farms and 10 laying breeder farms. Additionally, farms consisted of 75 cage and 313 free-ranged types of management (Table 1). Each sample consisted of a pool of fresh manure collected from several chickens or a pooled fecal sample from areas distributed in the litter of randomly selected poultry farm buildings. The collected samples were put into sealable plastic bags or screw-cap containers and were sent to the laboratory. One-gram fecal samples were homogenized in 5 ml saturated NaCl solution using a vortex mixer. All fecal samples were first examined for Eimeria oocysts using a standard McMaster technique, as previously described [20]. The number of oocysts was presented as the number of oocysts per gram of feces (OPG). The lower detection limit of the fecal examination was less than 50 OPG.
Genomic DNA extraction from oocysts
Before genomic DNA was extracted from fecal oocysts, positive fecal samples were diluted fivefold with phosphate-buffered saline (PBS, pH 7.4), homogenized using a vortex mixer, and then filtered using a mesh sieve. The materials that did not pass through the sieve were discarded. The filtrate was then transferred to a plastic centrifuge container and centrifuged for 10 min at 1,000 × g. The supernatant was discarded, while the sediment containing the oocysts was resuspended with PBS and washed two times using centrifugation (1,000 × g, for 10 min). After washing, the sediment was resuspended with 2.5% potassium dichromate solution (Samchun Chemicals, Pyongtack, Korea) and incubated at 28 °C for 1–3 days with shaking and aeration to achieve sporulation. Oocysts sporulation was checked using microscopy at 100 × magnification. The samples were washed twice using PBS (pH 7.4) by centrifugation to remove the potassium dichromate solution. They were then resuspended in PBS. Genomic DNA extraction was performed using the QIAamp® Fast DNA Stool Mini Kit according to the manufacturer’s protocol (Qiagen, Germany).
Molecular characterization of Eimeria species using PCR
Eimeria species was determined using PCR primers (Table 4) targeting the internal transcribed spacer 1 (ITS-1) of nuclear ribosomal DNA of each Eimeria species. Each PCR reaction was performed in a 20 μl PCR premix (Bioneer, South Korea) containing 1 μl genomic DNA template and 10 p mol each for the forward and reverse primers, as previously described [20]. The cycling program for the primary PCR using ITS-1 primers consisted of a denaturation step for 5 min at 95 °C, followed by 30 cycles at 95 °C for 45 s, 55 °C for 45 s, and 72 °C for 1 min, then a final extension of 72 °C for 4 min in a thermal cycler. The secondary PCR reaction with the primary PCR product was performed as previously described [61]. Briefly, initial denaturation was performed for 5 min at 95 °C, then 30 cycles at 95 °C for 30 s, 58 or 65 °C for 30 s, and 72 °C for 1 min, followed by a final extension at 72 °C for 5 min. Each PCR product was loaded to a 1.5% agarose gel with ethidium bromide and was run in TAE buffer for 25 min. The DNA fragments were visualized using ultraviolet light.
Anticoccidial drug sensitivity test
Experimental design and infection
The anticoccidial drug sensitivity test was carried out for a total of 16 days in an experimental animal facility in Gyeongsang National University. Day-old ROSS 308 female broiler chicks (Samhwa, Korea) were housed in cages in a coccidiosis-free environment and were given ad libitum access to water and feed throughout the experiment. To make similar and balanced groups before treatment, the weights of 5-day-old chicks were measured and the chicks weighing 110-125 g were randomly assigned to groups (n = 40/group): uninfected/untreated control group, infected/untreated groups and infected/treated groups. Treatment and infection was randomized to each group using = RANDBETWEEN () function in Microsoft Excel and the groups were randomly allocated to cages.
Anticoccidial drug sensitivity testing of nine field samples (Additional file 1) to the six anticoccidials clopidol (125 ppm), diclazuril (1 ppm), maduramycin (5 ppm), monensin (100 ppm), salinomycin (60 ppm), and toltrazuril (25 ppm) was performed. At 7 days old, the chicks were weighed and orally inoculated with 3 X 104 sporulated oocysts of the field samples to their corresponding group and fed a standard diet supplemented with anticoccidial drugs beginning 2 days before infection and continued throughout the entire experimental period. Oocysts used for infection were cleaned using flotation on 5.25% sodium hypochlorite; they were then washed three times with PBS. The untreated/infected control birds (positive control, PC) were given their respective inoculums, while the uninfected untreated control birds (negative control, NC) were given PBS. A dose titration trial on the nine different field samples was performed to determine the inoculation dose to cause a weight gain reduction of 15–25% in infected and unmedicated birds [63].
Birds were observed daily and the mortality was monitored. The following parameters were measured: body weight gain, oocyst number/g and lesion scores. Body weight gain (n = 20) was measured each bird at 9 days post-infection. Fecal samples were collected from 6 to 9 days post-infection and were homogenized using a blade grinder. Two 20-ml samples were collected from each suspension. Samples were diluted in saturated NaCl, and oocysts were counted microscopically using a McMaster counting chamber. Total oocyst numbers were calculated using the formula: [total oocyst number = oocyst count × dilution factor × (fecal sample volume/counting chamber volume)/number of birds per cage]. Intestinal lesion score (n = 5, randomly picked) was evaluated from each group at 7 days post-infection, based on previously described scoring techniques [24]. The lesion score for each chicken was assigned a numerical value from 0 to 4 and was carried out blindly by three individuals.
Evaluation of drug resistance
Anticoccidial drug resistance of the Eimeria species in the farm samples was evaluated using the anticoccidial index (ACI), percent optimum anticoccidial activity (POAA), relative oocyst production (ROP), and reduced lesion score (RLS).
The ACI was calculated as previously described [42]: ACI = (rate of relative body weight gain + survival rate) – (lesion score + oocyst value). Oocyst value = (fecal oocyst number of treated and infected group ÷ fecal oocyst number of PC group) X 100. An ACI value ≥ 160 indicated sensitivity and an ACI value < 160 indicated resistance. POAA values were computed as previously described [42, 49]: POAA = [(average GSR of treated and infected group—average GSR of PC group) ÷ (average GSR of NC group—average GSR of PC group)] X 100. GSR = (total weight at end of experiment + weight of dead birds) ÷ total weight at start of experiment. A POAA value ≥ 50% indicated sensitivity and a POAA value < 50% indicated resistance. The ROP value was computed as previously described [49]: ROP = (oocyst output of treated and infected group ÷ oocyst output of PC group) X 100. An ROP value < 15% indicated sensitivity and a value ≥ 15% indicated resistance. RLS were computed as previously described [64] = (mean lesion score of PC group ÷ mean lesion score of treated and infected group)/mean lesion score of PC group X 100. An RLS > 50% indicated sensitivity and RLS ≤ 50% indicated resistance.
Each index was assigned a point system in the presence of resistance. The overall assessment of drug resistance was adapted from Lan et. al [49]. Briefly, field samples were considered to be severely resistant when all or three indexes showed resistance (3 or 4), moderately resistant when 2 of the indexes showed resistance, slightly drug resistant if 1 showed resistance, and no drug resistance (0) in the absence of indexes indicating resistance.
Data analysis
Associations between risk factors and infection were evaluated using two-tailed Fisher’s exact tests or Chi-square (X2) tests. Body weight gain, lesion scores, and oocyst production were analyzed using Student’s t-tests or one-way ANOVA and Dunnett’s multiple comparison tests. All analyses were performed using Graphpad Prism version 9.0 and InStat statistical software (GraphPad, USA). Differences were considered statistically significant at P < 0.05. Results were expressed as mean ± standard error values.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ACI:
-
Anticoccidial Index
- CI:
-
Confidence Interval
- ITS-1:
-
Internal Transcribed Spacer 1
- NC:
-
Negative Control
- NS:
-
Not significant
- OPG:
-
The number of oocysts per gram of feces
- OR:
-
Odds Ratio
- PBS:
-
Phosphate-Buffered Saline
- PC:
-
Positive Control
- POAA:
-
Percent Optimum Anticoccidial Activity
- RLS:
-
Reduced Lesion Score
- ROP:
-
Relative Oocyst Production
References
Lawal JR, Jajere SM, Ibrahim UI, Geidam YA, Gulani IA, Musa G, et al. Prevalence of coccidiosis among village and exotic breed of chickens in Maiduguri, Nigeria. Vet World. 2016;9:653–9.
Armour N, Burleson M, Gingerich E, Genger S, TSchaal T, Glisson J, Ferguson-Noel N and Smith J. 2019 Research priorities of the American Association of Avian Pathologists. 2019;1–33. s
Blake DP, Tomley FM. Securing poultry production from the ever-present Eimeria challenge. Trends Parasitol. 2014;30:12–9.
Chapman HD. Milestones in avian coccidiosis research: a review. Poult Sci. 2014;93:501–11.
Joyner LP, Long PL. The specific characters of the Eimeria, with special reference to the coccidia of the fowl. Avian Pathol. 1974;3:145–57.
Blake DP, Knox J, Dehaeck B, Huntington B, Rathinam T, Ravipati V, et al. Re-calculating the cost of coccidiosis in chickens. Vet Res. 2020;51:115.
Allen PC, Fetterer RH. Recent advances in biology and immunobiology of Eimeria species and in diagnosis and control of infection with these coccidian parasites of poultry. Clin Microbiol Rev. 2002;15:58–65.
Abbas RZ, Iqbal Z, Blake D, Khan MN, Saleemi MK. Anticoccidial drug resistance in fowl coccidia: the state of play revisited. Worlds Poult Sci J. 2011;67:337–49.
Györke A, Pop L, Cozma V. Prevalence and distribution of Eimeria species in broiler chicken farms of different capacities. Parasite. 2013;20:50.
Lan L, Zuo B, Ding H, Huang Y, Chen X, Du A. Anticoccidial evaluation of a traditional Chinese medicine-Brucea javanica-in broilers. Poult Sci. 2016;95:811–8.
Pop LM, Varga E, Coroian M, Nedisan ME, Mircean V, Dumitrache MO, et al. Efficacy of a commercial herbal formula in chicken experimental coccidiosis. Parasit Vectors. 2019;12:1–9.
Quiroz-Castañeda RE, Dantán-González E. Control of avian coccidiosis: future and present natural alternatives. Biomed Res Int. 2015;2015:430610.
Yim D, Kang SS, Kim DW, Kim SH, Lillehoj HS, Min W. Protective effects of Aloe vera-based diets in Eimeria maxima-infected broiler chickens. Exp Parasitol. 2011;127:322–5.
Belli SI, Mai K, Skene CD, Gleeson MT, Witcombe DM, Katrib M, et al. Characterisation of the antigenic and immunogenic properties of bacterially expressed, sexual stage antigens of the coccidian parasite Eimeria maxima. Vaccine. 2004;22:4316–25.
Shirley MW, Smith AL, Tomley FM. The biology of avian Eimeria with an emphasis on their control by vaccination. Adv Parasitol. 2005;60:285–330.
Shirley MW, Bedrník P. Live attenuated vaccines against Avian coccidiosis: success with precocious and egg-adapted lines of Eimeria. Parasitol Today. 1997;13:481–4.
Williams RB. Anticoccidial vaccines for broiler chickens: Pathways to success. Avian Pathol. 2002;31:317–53.
El-Ghany WAA. Intervention strategies for controlling poutry coccidiosis: current knowledge. J World’s Poult Res. 2021;11:487–505.
Jenkins M, Klopp S, Ritter D, Miska K, Fetterer R. Comparison of Eimeria species distribution and salinomycin resistance in commercial broiler operations utilizing different coccidiosis control strategies. Avian Dis. 2010;54:1002–6.
Lee BH, Kim WH, Jeong J, Yoo J, Kwon YK, Jung BY, et al. Prevalence and cross-immunity of Eimeria species on Korean chicken farms. J Vet Med Sci. 2010;72:985–9.
Nguyen BT, Flores RA, Cammayo PLT, Kim S, Kim WH, Min W. Anticoccidial activity of berberine against Eimeria-infected chickens. Korean J Parasitol. 2021;59:403–8.
Lee HA, Hong S, Chung YH, Song KD, Kim O. Anticoccidial effects of Galla rhois extract on Eimeria tenella-infected chicken. Lab Anim Res. 2012;28:193–7.
Jang SI, Jun MH, Lillehoj HS, Dalloul RA, Kong IK, Kim S, et al. Anticoccidial effect of green tea-based diets against Eimeria maxima. Vet parasitol. 2007;144:172–5.
Johnson J, Reid WM. Anticoccidial drugs: Lesion scoring techniques in battery and floor-pen experiments with chickens. Exp Parasitol. 1970;28:30–6.
Debbou-Iouknane N, Benbarek H, Ayad A. Prevalence and aetiology of coccidiosis in broiler chickens in Bejaia province, Algeria. Onderstepoort J Vet Res. 2018;85:4–9.
Karaer Z, Guven E, Akcay A, Kar S, Nalbantoglu S, Cakmak A. Prevalence of subclinical coccidiosis in broiler farms in Turkey. Trop Anim Health Prod. 2012;44:589–94.
Mokhtar HP, Yagoob G. Prevalence of coccidiosis in broiler chicken farms in and around Marand City, Iran. J Entomol Zool Stud. 2016;4:174–7.
Negash A, Mohamed A, Wondimu K. Study on prevalence and risk factors associated with poultry coccidiosis in and around Hawassa Town, South Ethiopia. Br Poult Sci. 2015;4:34–43.
Sharma S, Iqbal A, Azmi S, Mushtaq I, Wani ZA, Ahmad S. Prevalence of poultry coccidiosis in Jammu Region of Jammu & Kashmir State. J Parasit Dis. 2015;39:85–9.
Yousaf A. Prevalence of coccidiosis in different broiler poultry farms in Potohar region (distract Rawalpindi) of Punjab-Pakistan. J Dairy Vet Anim Res. 2018;7:87–90.
Haug A, Gjevre AG, Thebo P, Mattsson JG, Kaldhusdal M. Coccidial infections in commercial broilers: epidemiological aspects and comparison of Eimeria species identification by morphometric and polymerase chain reaction techniques. Avian Pathol. 2008;37:161–70.
Agishi G, Luga II, Rabo JS. Prevalence of coccidiosis and Eimeria species in layers and broilers at slaughter houses in Makurdi, Benue State. Int J Eng Sci. 2016;5:8–11.
Al-Natour MQ, Suleiman MM, Abo-Shehada MN. Flock-level prevalence of Eimeria species among broiler chicks in northern Jordan. Prev Vet Med. 2002;53:305–10.
Nematollahi A, Moghaddam G, Niyazpour F. Prevalence of Eimeria sp. among broiler chicks in Tabriz (Northwest of Iran). J Poult Sci. 2008;2:72–4.
Mesa-Pineda C, Navarro-Ruíz JL, López-Osorio S. Chicken coccidiosis : from the parasite lifecycle to control of the disease. Front Vet Sci. 2021;8:787653.
Lunden A, Thebo P, Gunnarsson S, Hooshmand-Rad P, Tauson R, Uggla A. Eimeria infections in litter-based, high stocking density systems for loose-housed laying hens in Sweden. Br Poult Sci. 2000;41:440–7.
Chasser KM, Duff AF, Wilson KM, Briggs WN, Latorre JD, Barta JR, et al. Research note: evaluating fecal shedding of oocysts in relation to body weight gain and lesion scores during Eimeria infection. Poult Sci. 2020;99:886–92.
Brackett S, Bliznick A. The reproductive potential of five species of coccidia of the chicken as demonstrated by oocyst production. J Parasitol. 1952;38:133–9.
Long PL, Rowell JG. Sampling broiler house litter for coccidial oocysts. Br Poult Sci. 1975;16:583–92.
Graat EAM, Van Der Kooij E, Frankena K, Henken AM, Smeets JFM, Hekerman MTJ. Quantifying risk factors of coccidiosis in broilers using on-farm data based on a veterinary practice. Prev Vet Med. 1998;33:297–308.
Williams RB, Marshall RN, La Ragione RM, Catchpole J. A new method for the experimental production of necrotic enteritis and its use for studies on the relationships between necrotic enteritis, coccidiosis and anticoccidial vaccination of chickens. Parasitol Res. 2003;90:19–26.
Ojimelukwe AE, Emedhem DE, Agu GO, Nduka FO, Abah AE. Populations of Eimeria tenella express resistance to commonly used anticoccidial drugs in southern Nigeria. Int J Vet Sci Med. 2018;6:192–200.
Ola-Fadunsin SD. Investigations on the occurrence and associated risk factors of avian coccidiosis in Osun State Southwestern Nigeria. J Parasitol Res. 2017;2017:9264191.
Kaboudi K, Umar S, Munir MT. Prevalence of coccidiosis in free-range chicken in Sidi Thabet, Tunisia. Scientifica (Cairo). 2016;2016:7075195.
Williams RB. Quantification of the crowding effect during infections with the seven Eimeria species of the domesticated fowl: its importance for experimental designs and the production of oocyst stocks. Int J Parasitol. 2001;31:1056–69.
Aarthi S, Dhinakar Raj G, Raman M, Gomathinayagam S, Kumanan K. Molecular prevalence and preponderance of Eimeria species among chickens in Tamil Nadu, India. Parasitol Res. 2010;107:1013–7.
Sun XM, Pang W, Jia T, Yan WC, He G, Hao LL, et al. Prevalence of Eimeria Species in broilers with subclinical signs from fifty farms. Avian Dis. 2009;53:301–5.
Mattiello AR, Boviez JD, Mcdougald LR, Mattiello R, Boviez AJD, Mcdougald LR. Eimeria brunetti and Eimeria necatrix in chickens of Argentina and confirmation of seven species of Eimeria. Avian Dis. 2018;44:711–4.
Lan LH, Sun BB, Zuo BXZ, Chen XQ, Du AF. Prevalence and drug resistance of avian Eimeria species in broiler chicken farms of Zhejiang province, China. Poult Sci. 2017;96:2104–9.
Stephan B, Rommel M, Daugschies A, Haberkorn A. Studies of resistance to anticoccidials in Eimeria field isolates and pure Eimeria strains. Vet Parasitol. 1997;69:19–29.
Allen PC, Danforth HD, Vinyard BL. Development of a protective index to rank effectiveness of multiple treatments within an experiment: Application to a cross-protection study of several strains of Eimeria maxima and a live vaccine. Avian Dis. 2004;48:370–5.
Arabkhazaeli F, Modrisanei M, Nabian S, Mansoori B, Madani A. Evaluating the resistance of Eimeria species field isolates to anticoccidial drugs using three different indices. Iranian J Parasitol. 2013;8:234–41.
Mathis GF, McDougald LR. Drug responsiveness of field isolates of chicken coccidia. Poult Sci. 1982;61:38–45.
Abbas RZ, Iqbal Z, Nisar Khan M, Hashmi N, Hussain A. Prophylactic efficacy of diclazuril in broilers experimentally infected with three field isolates of Eimeria tenella. Int J Agric Biol. 2009;11:606–10.
Zhang JJ, Wang LX, Ruan WK, An J. Investigation into the prevalence of coccidiosis and maduramycin drug resistance in chickens in China. Vet Parasitol. 2013;191:29–34.
Bedrník P, Jurkovic P, Kucera J, Firmanová A. Cross resistance to the ionophorous polyether anticoccidial drugs in Eimeria tenella isolates from Czechoslovakia. Poult Sci. 1989;68:89–93.
Martin AG, Danforth HD, Barta JR, Fernando MA. Analysis of immunological and cross-protection and sensitivities to anticoccidial drugs among five geographical and temporal strains of Eimeria maxima. Int J Parasitol. 1997;27:527–33.
Abbas RZ, Iqbal Z, Sindhu ZD, Khan MN, Arshad M. Identification of cross-resistance and multiple resistance in Eimeria tenella field isolates to commonly used anticoccidials in Pakistan. J Appl Poult Res. 2008;17:361–8.
Greif G, Stephan B, Haberkorn A. Intraspecific polymorphisms of Eimeria species due to resistance against an anticoccidial drugs. Parasitol Res. 1996;82:706–14.
Ministry of Agriculture, Food and Rural Affairs and National Institute of Food and Drug Safety Evaluation. Monitoring national antimicrobial consumption and resistance. 2019; 7–17.
Haug A, Thebo P, Mattsson JG. A simplified protocol for molecular identification of Eimeria species in field samples. Vet Parasitol. 2007;146:35–45.
Lew AE, Anderson GR, Minchin CM, Jeston PJ, Jorgensen WK. Inter- and intra strain variation and PCR detection of the internal transcribed spacer 1 (ITS-1) sequences of Australian isolates of Eimeria species form chickens. Vet Parasitol. 2003;112:33–50.
Holdsworth PA, Conway DP, McKenzie ME, Dayton AD, Chalpman HD, Mathis GR, et al. World Association for the Advancement of Veterinary Parasitology (WAAVP) guidelines for evaluating the efficacy of anticoccidial drugs in chickens and turkeys. Vet Parasitol. 2004;121:189–212.
Fei C, Fan C, Zhao Q, Lin Y, Wang X, Zheng W, et al. Anticoccidial effects of a novel triazine nitromezuril in broiler chickens. Vet Parasitol. 2013;198:39–44.
Acknowledgements
Not applicable.
Funding
This work was supported by IPET through the Animal Disease Management Technology Development Program funded by MAFRA (320062–2). The funder did not have any roles in the design and conduct of the experiment, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
RF, WK, and WM designed the experiments; RF, BTN, PC, TCV, and HN conducted the experiments and analyzed the data; RF and WM drafted the original manuscript; RF, WK, SK, BKN, and WM reviewed and edited the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
According to the institutional review board of Gyeongsang National University (GNU), permission is unnecessary to collect chicken fecal samples. All animal maintenance and experimental procedures were performed according to GNU Guidelines for the Care and Use of Experimental Animals and were approved by the Institutional Animal Care and Use Committee (IACUC) (GNU-191111-C0084). Humane endpoint criteria were set for all animals such that severely moribund animals with severe weight loss, tremors, and unresponsiveness or unawareness of stimuli were euthanized immediately using atlanto-occipital dislocation. All remaining animals were euthanized at specific post-inoculation timepoints. The present study were carried out in compliance with the ARRIVE guidelines.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflicts of interest related to this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1.
Specific Eimeria species present in each farm sample.
Additional file 2.
Survival rates of birds infected with farm samples.
Additional file 3.
Anticoccidial Index (ACI) of each farm sample to different anticoccidials.
Additional file 4.
Percent Optimum Anticoccidial Activity (POAA) of each farm sample to different anticoccidials.
Additional file 5.
Relative Oocyst Production (ROP) of each farm sample to different anticoccidials.
Additional file 6.
Reduction of Lesion Score (RLS) of each farm sample to different anticoccidials.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Flores, R.A., Nguyen, B.T., Cammayo, P.L.T. et al. Epidemiological investigation and drug resistance of Eimeria species in Korean chicken farms. BMC Vet Res 18, 277 (2022). https://doi.org/10.1186/s12917-022-03369-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12917-022-03369-3