Abstract
Background
Preeclampsia (PE) is one of the main causes of medical complication of pregnancy and is the main cause of perinatal mortality and morbidity. It is one of the top causes of maternal mortality in Ethiopia. Also known as transient hypertension, gestational hypertension (GH) is increased blood pressure during pregnancy without proteinuria, which is expected to return to normal by the 12th-week postpartum visit. PE is GH with proteinuria and /or other systemic manifestations. Evidence from high income countries show that GH significantly progresses towards PE. To our knowledge, this is the first study on the progression of GH towards PE in an African setting. The objective of this study is, therefore, to assess the incidence of GH, progression towards PE and factors associated with progression in Ethiopia.
Methods
This is a prospective cohort study conducted at Ayder Comprehensive Specialized Hospital (ACSH) and Mekelle General Hospital (MGH), the largest referral centers in Northern Ethiopia. Two hundred and forty women with GH were enrolled and followed up until delivery. Clinical and laboratory data at initial presentation and at follow-up were compared among women who progressed towards PE and who remained with the diagnosis of GH. Logistic regression analysis was employed to model the combined effects of the clinical and laboratory data as significant predictors of progression from GH to PE.
Result
The incidence of GH in this study was 6 % (4.9–8.5). The rate of progression was 17.1 % (13.4–23.8). Previous history of GH, anemia during pregnancy, previous second-trimester spontaneous abortion were significant predictors of progression.
Conclusions
There is a high rate of progression of GH towards PE. In a resource-limited setting where predictive and diagnostic tools are scarce, clinical profile of women should be taken into consideration for prediction and diagnosis of PE.
Similar content being viewed by others
Background
Globally hypertension is the most frequent medical complication of pregnancy, occurring in 10 % of pregnancies and includes a range of conditions, including chronic hypertension, gestational hypertension (GH), and preeclampsia (PE) [1]. In Ethiopia, the prevalence of all forms of hypertensive disorders of pregnancy varies from 1.8 to 10 % [2]. GH also known as transient hypertension is the new onset of hypertension after 20 weeks of gestation [1]. GH is expected to return to normal by the 12th-week postpartum visit. PE, on the other hand, is defined as GH with proteinuria and/or systemic manifestations( headache, right upper quadrant pain, epigastric pain, blurring of vision, organ function derangement, low platelet count) and is associated with more grave maternal and neonatal outcomes [1, 3, 4].
In a 10 year study by Walker and colleagues in a Calgary health region in Canada, the incidence of GH was found to be 6.3 % [5]. In another study in Nigeria, the incidence of GH was 5.9 % [6]. Saudan et al. reported 15–25 % of women who are initially admitted with GH progress to PE [4].
Globally, 10–15 % of maternal deaths are directly related with PE and eclampsia [7]. Complications are a lot worse in resource-limited settings which are associated with a lack of antenatal care follow up and delays in diagnosis and treatment [2, 8]. Previous studies in Ethiopia show that perinatal mortality associated with hypertensive disorders of pregnancy is one of the highest in the world, 111/1000 live births [9]. In a retrospective hospital-based study in Ethiopia, PE and eclampsia accounted for 35 % of maternal deaths, highlighting the significance of this disorder in both maternal and fetal outcomes [8].
Although hypertensive disorders of pregnancy are more common in low and middle income countries than they are in the high income ones, little is known about the incidence of GH and progression towards PE [2, 4]. To our knowledge to date, no published accessible data is available on the incidence of GH and factors affecting its progression towards PE in this country. Women with GH may be managed safely as outpatients, and it would be helpful to know both the absolute risk of progression from GH to PE and the factors at the initial presentation which predict this progression [1]. The objective of this study is, therefore, to assess the incidence of GH, progression towards PE and factors associated with progression in Ethiopia.
Methods
Operational definitions
GH was defined as the onset of hypertension (systolic BP > = 140 mmHg and /or diastolic BP > = 90 mmHg) after 20 weeks of gestation [1]. Hypertension in these women was confirmed following repeated BP measurements during a 4-6-h day assessment unit visit or during admission.
PE was diagnosed when one or more of the following maternal systemic complications accompanied hypertension: proteinuria > = 2 + on dipstick urine analysis, renal impairment (plasma creatinine > = 1.2 mmol/L), hepatic dysfunction (aspartate transaminase > = 70 or double rise from baseline, right upper quadrant pain and/or sever epigastric pain, hematologic abnormality (platelet count < 100,000/L), neurologic features ( blurred vision, headache ,abnormal body movement ) or sever hypertension (systolic BP > = 160 mmHg and/or diastolic BP > = 110 mmHg ). Abnormal body movement is generalized tonic clonic type of seizure. Right upper quadrant pain is right sided upper abdominal pain, just beneath the ribs.
Study setting
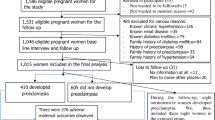
This is a prospective cohort study conducted in two public Hospitals of Mekelle City. Ayder Comprehensive Specialized Hospital (ACSH) and Mekelle General Hospital (MGH). Ayder Comprehensive Specialized Hospital, a tertiary care center for a catchment area over 8 million people in north Ethiopia. Mekelle General Hospital is the second largest Hospital in Tigrai region. High risk pregnant women including hypertensive disorders of pregnancy are referred to both hospitals for management.
Study period was between August 1, 2016 and July 31, 2017. Every case of GH seen during the study period at the hospitals were enrolled in the study Patients with previously known essential hypertension, renal disease or other secondary cause of hypertension were excluded.
Clinical and laboratory data
The data was collected by 6 trained midwives and 4 trained resident physicians at both hospitals. The following clinical and laboratory data at initial presentation were recorded in women with GH : age, parity, abortion history, gestational age, educational status, family monthly income, marital status, previous history of GH, previous history of PE, family history of diabetes mellitus, family history of chronic hypertension (obtained from patient history alone), hemoglobin, random blood sugar, platelet count.
Clinical and laboratory data were recorded at each visit and during delivery in GH women. Participants were allowed to take rest for 10 to 15 min before blood pressure had been measured. Blood pressure measurements were taken while the women was seated using mercury sphygmomanometer apparatus which covers two third of the upper arm. The measurement was taken from right arm. The cuff was inflated at a rate of 2–3 mm Hg per second and deflated at a rate of 2–3 mm. First and fifth Korotkoff sounds were taken for systole and diastole respectively [10]. The following symptoms of PE were also obtained during each follow up: headache, right upper quadrant pain, epigastric pain, blurred vision, and abnormal body movement.
The following laboratory data at initial presentation and during follow up were additionally recorded in patients with GH: creatinine, aspartate amino transferase, urine protein. Women with GH were reviewed on average every 2 weeks until delivery, which is consistent with their regular follow up. Though follow-up until 12 weeks is required to confirm that women have not developed preeclampsia after birth, women were not followed for 12 weeks postpartum.
Outcomes
The primary outcome was progression from GH to PE. Both maternal and perinatal outcomes were recorded. The maternal outcomes recorded were: number of maternal systemic complications (as mentioned above for PE under the heading” definitions”), antepartum hemorrhage (APH), mode of delivery. Additionally, the following perinatal outcomes were recorded: gestational age at delivery, birth weight, perinatal mortality, small for gestational age (SGA) rate, admission to neonatal intensive care unit (NICU).
Statistical analysis
Incidence of GH was determined by dividing the entire number of exposures by the total number of deliveries during the study period. To determine the factors which might predict the development of PE, clinical and laboratory data at initial presentation were compared among women who developed PE and who did not. An a priori power analysis was undertaken to determine an adequate sample size for our primary outcome, progression from GH towards PE. We estimated that 112 participants would provide greater than 80 % power to identify difference between early and late gestational age at presentation in the primary outcome with odds ratio of 0.65 (95 % CI ,0.53–0.79) (Saudan et al. ) and two-sided significance level < 0.001 [4].
All statistical analyses were performed using the Statistical Package for Social Sciences for Windows 21.0 (SPSS Inc., Chicago, IL, USA). Values are expressed as mean ± SD. Continuous data (SD) were tested by analysis of variance and contingency tables were used for categorical data. Comparisons of participant characteristics were performed using a Pearson X2 test for categorical variables and two tailed student’s t test for continuous variables. P < 0.05 was considered significant. Binary logistic regression analysis was employed to assess the effects of the above clinical and laboratory data as predictors of progression from GH to PE. Then variables with P value less than or equal to 0.2 were fitted to multiple logistic regression. Moreover, clinically relevant variables were also fitted to the regression. Maternal age, gravidity, gestational age at diagnosis, history of abortion, family history of chronic hypertension, previous history of gestational hypertension, and hemoglobin were included in the model a priori. The predictive ability of the model was tested with Hosmer-Lemeshow goodness-of-fit test. Variables that were not significant were removed in a stepwise fashion. Finally variables with P-value ≤ 0.05 were considered as significant predictors of progression from GH to PE. After the purpose of the study has been informed, written informed consent was obtained from each study participants. Confidentiality of study participants was maintained.
Results
During the one year study period; the total number of deliveries was 4002. Two hundred forty women with GH were seen giving an incidence of 6 % (95 CI 4.9–8.5). The mean age of the study participants was 27.15 years and SD of 5.33. Nearly half of the participants were nulliparous, mean gestational age at diagnosis of GH was 33 weeks. Family history of PE (1 %) and previous history of PE (2.1 %) was not common (Table 1).
Of the 240 women with GH, 41 (17.1 %, 95 CI 13.4–23.8) developed PE. The mean age of women who progressed to PE, 29.6 years, was older than those who remained with the diagnosis of GH, 27.1 years (P = 0.01). Those who presented with GH but developed PE presented at earlier gestational age than those who remained with GH (32.8 ± 4.0 weeks vs. 36.7 ± 4.0, P = 0.01). Their mean gestational age at progression to PE was 37.1 ± 3. Further, they had a lower level of hemoglobin, 11.6 ± 2.0 g/dl, (P < 0.0001) at presentation (Table 1).
Women who progressed from GH to PE had comparable SBP/DBP, 145 ± 10 mmHg/ 92 ± 4 mmHg, at diagnosis with those who remained with a diagnosis of GH until delivery,140 mmHg±/91 ± 7 mmHg (P = 0.36/P = 0.37).
There was no difference in terms of gestational age at delivery and birth weight between women who progressed from GH to PE and women who remained with a diagnosis of GH until delivery (P = 0.56 and P = 0.29).
The most commonly experienced symptoms were neurologic manifestations. Twenty-six women (63.4 %) among those who developed PE had one or more of the following neurologic symptoms; headache, blurring of vision and abnormal body movement. Among the neurologic symptoms, headache and blurred vision were the two most common presenting complaints. Of the 41 women who progressed to PE from an initial diagnosis of GH, 25 of them had these two neurologic manifestations. Other symptoms experienced were liver dysfunction 27 (65.9 %), severe hypertension 25(61 %), APH 10 (24.4 %), renal impairment 5(12.2 %) and thrombocytopenia 4 (9.8 %) (Table 2). Of the 41 women who progressed to PE, 37 women (90.24 %) had symptoms and clinical signs of PE at the time of progression. Twenty two women had symptoms and clinical signs of PE while having normal laboratory result. Only 4 (9.76 %) women had abnormal laboratory result as sole evidence of progression.
Univariate analysis revealed that maternal age, gestational age, history of abortion, family history of chronic hypertension, previous history of GH, and hemoglobin were factors associated with progression at 0.2 level of significance. Gravidity was included in the multivariate analysis because of its clinical significance.
However abortion, previous history of GH, and hemoglobin level remained significantly and independently associated with progression of GH towards PE in the multivariate analysis (Table 3).
Women who had previous history GH were 26 times more likely to develop PE in the consecutive pregnancy than women who had not had previous history GH (AOR = 26.76, 95 % CI: 3.03–49.08). Women who had abortion are 3.85 times more likely to develop PE in the consecutive pregnancy than their counterparts (AOR = 3.84. 95 % CI: 1.07-25.0).
Another significant association was found between hemoglobin levels and women who developed PE. Women with a hemoglobin level between 7 and 10 g/dl are 13times more likely to develop PE than women with hemoglobin level 11 g/dl and above (AOR = 13.1, 95 % CI: 1.40-17.27).
Discussion
According to this study, the incidence of GH was 6 % (4.9–8.5) and the likelihood of progression from GH to PE was 17.1 % (13.4–23.8). This was more likely to occur in those who had history of abortion, previous history of GH, or low hemoglobin.
The overall incidence of GH in this study (6 %) is comparable with those previously reported studies which estimated 3–10 % [5, 11,12,13].
In our study, the rate of progression from GH to PE was 17.1 %. In agreement with a study by Saudan P. et al. which reported 15–25 % [4]. But lower than reported in another study by Barton et al. 46 %. It is good to mention that in the latter study the authors used urine dipstick + 1 as opposed to + 2 in our study to diagnose progression. Furthermore, when the authors used + 3 urine dipsticks, the rate of progression dropped to 9 %. The difference in urine dipstick cut off point used for the diagnosis of progression may explain the discrepancy in findings [14].
In this study women with previous history of GH were 26 times more likely to have PE in the subsequent pregnancy. This is much higher than a report from prospective study done in UK that showed women with previous GH were more likely to develop various obstetrics complications in subsequent pregnancies including 25 % risk of PE development [15]. The discrepancy may be explained due to set up difference. In our set up, urine dipstick is used to make a diagnosis of GH, potentially increasing its diagnosis.
Multiple studies have described that women with high hemoglobin level are likely to develop hypertensive disorders of pregnancy than those with normal level [16,17,18,19]. Elevated hemoglobin level can either be cause or effect of PE. There is limited intravascular expansion in women with PE leading to hemoconcentration. On the other end, the increased incidence of PE in pregnant women with elevated hemoglobin levels could be clarified by the toxic effects of haeme deposition on the vascular endothelium [20]. Interestingly, the present study found a significant association between low hemoglobin levels and progression towards PE. Women with a hemoglobin level between 7 and 10 were 13 times more likely to develop PE than women with hemoglobin level 11 and above. Another study by Abdulaziem et al. reported that severe anemia was risk factor for PE [21]. But the authors didn’t show association between mild/moderate anemia and PE. Moreover, the hemoglobin was determined after women presented with PE, posing difficulty in making cause and effect relationship. As other causes of anemia were not studied in the present study, it is difficult to determine if the anemia was the predictor.
This study found out that previous abortion is associated with risk of progression towards PE. Women who had an abortion were 3.85 times more likely to develop PE in the consecutive pregnancy than their counterpart. This is similar to published report by S Bhattacharya et al. that states women with history of abortion were 3.3 times more likely to suffer from PE compared to women with no previous history [22]. Other studies have showed that risk of PE to be related with the number of previous abortions, Mahdi S. et al. reported that the number of previous spontaneous abortions was found to be associated with PE. In this latter study, with each previous abortion the odds having PE in subsequent pregnancy was increased by 1.2 times [23]. Additionally Saudan P. et al. reported an association between history of abortion and chance of progression from GH-PE. However in a cohort study, Gunnarsdottir et al. found relationship between three previous abortions and PE not with one or two previous abortions [24]. In the present study we were not able to quantify number of previous abortions.
This study has found out that majority of the women (90.24 %) who progressed towards PE had signs and symptoms of PE (headache, blurring of vision, right upper quadrant pain, epigastric pain, abnormal body movement, and sever hypertension) at the diagnosis of progression. The remaining 9.76 % women had abnormal laboratory result as sole evidence of progression. Management of GH requires continued monitoring to detect transformation from this relatively mild disorder to the more severe disorder of PE [11]. However, either signs and symptoms or abnormal laboratory result is enough to make a diagnosis of progression [25]. This study has shown that majority of the women might have not required lab investigations to rule in or rule out PE. Thus, in low resource setting where laboratory investigations are absent signs and symptoms could be used to diagnose progression towards PE without resorting to laboratory investigations.
In our observation, comparison of perinatal outcome in between those who remained with GH alone till delivery and those who progressed to PE in terms of; birth weight, intrauterine growth restriction, admission to N-ICU, and 5th minute Apgar score and stillbirth was not statistically significantly different. This is similar to previous studies done in different centers which showed late onset PE had favorable perinatal outcome. Most common poor perinatal outcomes related to early onset PE include; fetal growth restriction, preterm delivery, poor Apgar score and stillbirth [26].
Study done in turkey by Riza M. et al. showed that incidences of small-for-gestational age, Apgar score < 7 at 5 min, and stillbirth were significantly higher in women with early onset PE compared to late onset PE [27]. Similarly, another cohort study by Wojtowicz et al. et al showed that women with late onset PE had better perinatal outcome as compared to early onset PE [28]. In our study the mean gestational age at progression towards PE was 37.1 ± 3 weeks. This late gestational age at progression might have led to comparable perinatal outcome among women who did and who did not progressed towards PE.
Conclusions
The present study highlighted the incidence and rate of progression is similar to other studies done in low, middle, and high income countries.
Previous history of GH, anemia during pregnancy, previous second-trimester spontaneous abortion were significant predictors of progression towards PE.
The present study underlined that, in a resource-limited setting where predictive and diagnostic means are limited, the clinical picture of women should be taken into consideration for prediction and diagnosis of PE. Owing to the significant association found in-between rate of progression and low hemoglobin, further prospective research is required to identify if anemia was the cause or effect of PE.
Limitation of the study
Definite diagnosis of GH is made retrospectively when the patient doesn’t develop PE and if blood pressure returns to normal by 12-week postpartum visit. In this study, subjects were followed until delivery only. Thus, hampering the true rate of progression of GH towards PE. Though BMI is reported in other studies as a risk factor for preeclampsia [29], we have not assessed it in this study; as most of the women did not recall their pre-pregnancy weight. Moreover, there are other clinical risk factors reported in other studies which were not assessed in this study. These clinical risk factors are hypercholesterolemia, Antiphospholipid syndrome, and thrombophilia [30, 31]. We were not able to assess these factors due to limitation in set up. Due to these limitations, we cannot state these risk factors are not important in the progression of PE. Although all cases seen during the study period were enrolled, smaller sample size has limited us from doing further analysis of variables with less frequent observations.
Availability of data and materials
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- APH:
-
Antepartum hemorrhage
- ACSH :
-
Ayder Comprehensive Specialized Hospital
- CS:
-
Cesarean section
- DBP:
-
Diastolic blood pressure
- GH:
-
Gestation hypertension
- HDP:
-
Hypertensive disorders of pregnancy
- HTN:
-
Hypertension
- NICU:
-
Neonatal intensive care unit
- PE:
-
Preeclampsia
- SBP:
-
Systolic blood pressure
References
Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000;183(1). Available from: https://pubmed.ncbi.nlm.nih.gov/10920346/.
Berhe A, Kassa G, Fekadu G, Muche A. Prevalence of hypertensive disorders of pregnancy in Ethiopia: a systemic review and meta-analysis. BMC Pregnancy Childbirth. 2018;18(1). Available from: https://bmcpregnancychildbirth.biomedcentral.com/articles/10.1186/s12884-018-1667-7.
Dolea C, Abouzahr C. Global burden of environmental disease. Epidemiology. 2000;11(4):S159. Available from: https://www.researchgate.net/publication/265222498.
Saudan P, Brown M, Buddle M, Jones M. Does Gestational Hypertension Become Pre-Eclampsia?. Obstetrical & Gynecological Survey [Internet]. 1999;54(5):287–288. Available from: https://obgyn.onlinelibrary.wiley.com/doi/full/10.1111/j.1471-0528.1998.tb09971.x.
Walker R, Hemmelgarn B, Quan H. Incidence of gestational hypertension in the Calgary Health Region from 1995 to 2004. Canad JCardiol. 2009;25(8):e284-e287. Available from: https://pubmed.ncbi.nlm.nih.gov/19668790/.
Umegbolu E, Ogamba J. Incidence of gestational hypertension among pregnant women (2006–2015) in Enugu State, Southeast Nigeria: a retrospective study. International J Commun Med Public Health. 2017;4(2):357. Available from: https://www.ijcmph.com/index.php/ijcmph/article/view/575.
Duley L. The global impact of pre-eclampsia and eclampsia. Semin Perinatol.2009; 33(3):130–137. Available from: https://pubmed.ncbi.nlm.nih.gov/19464502/.
Terefe W, Getachew Y, Hiruye A, Deribew M, Haile Mariam D, Mammo D, et al. Patterns of hypertensive disorders of pregnancy and associated factors at Debre Berhan Referral Hospital, North Shoa, Amhara Region. Ethiop Med J. 2015;(Suppl 2):57–65. Available from: https://pubmed.ncbi.nlm.nih.gov/26591284/.[cited 20 June 2020].
Asseffa NA, Demissie BW. Perinatal outcomes of hypertensive disorders in pregnancy at a referral hospital, Southern Ethiopia. PLoS ONE. 2019;14(2):e0213240. https://doi.org/10.1371/journal.pone.0213240.
Ogedegbe G, Pickering T. Principles and techniques of blood pressure measurement. Cardiol Clin. 2010;28(4):571–586. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3639494/.
Wallis A, Saftlas A, Hsia J, Atrash H. Secular Trends in the Rates of Preeclampsia, Eclampsia, and Gestational Hypertension, United States, 1987–2004. Am J Hypertens. 2008;21(5):521–526. Available from: https://pubmed.ncbi.nlm.nih.gov/18437143/.
Sadanandan K, Kurian S, Valliot B, Sasidharan A, Sherin N, Madhu R, et al. Prevalence of gestational hypertension and factors influencing selection of anti-hypertensive drugs in pregnancy. J Hypertens. 2019;37:e239-e240. Available from: https://journals.lww.com/jhypertension/Abstract/2019/07001.
Liu C, Cheng P, Chang S. Maternal Complications and Perinatal Outcomes Associated with Gestational Hypertension and Severe Preeclampsia in Taiwanese Women. J Formosan Med Assoc. 2008;107(2):129–138. Available from: https://pubmed.ncbi.nlm.nih.gov/18285245/.
Barton JR, O’brien JM, Bergauer NK, Jacques DL, Sibai BM. Mild gestational hypertension remote from term: progression and outcome. Am J Obstet Gynecol. 2001;184(5):979 – 83. Available from: https://pubmed.ncbi.nlm.nih.gov/11303208/.
Nzelu D, Dumitrascu-Biris D, Hunt K, Mark C, Kametas N. P 17 Pregnancy outcomes in women with previous gestational hypertension – A cohort study to guide counselling and management. Pregnancy Hypertens. 2017;9:44–45. Available from: https://pubmed.ncbi.nlm.nih.gov/29113718/.
Aghamohammadi A, Zafari M, Tofighi M. High maternal hemoglobin concentration in first trimester as risk factor for pregnancy induced hypertension. Caspian J Intern Med. 2011;2(1):194–197. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3766933/.
Phaloprakarn C, Tangjitgamol S. Impact of high maternal hemoglobin at first antenatal visit on pregnancy outcomes: a cohort study. J Perinat Med. 2008; 36(2):115-9. Available from: https://pubmed.ncbi.nlm.nih.gov/18331205/.
Wang C, Lin L, Su R, Zhu W, Wei Y, Yan J, Feng H, Li B, Li S, Yang H. Hemoglobin levels during the first trimester of pregnancy are associated with the risk of gestational diabetes mellitus, pre-eclampsia and preterm birth in Chinese women: a retrospective study. BMC Pregnancy Childbirth. 2018;18(1):263. Available from: https://pubmed.ncbi.nlm.nih.gov/29940874/.
Huisman A, Aarnoudse JG. Increased 2nd trimester hemoglobin concentration in pregnancies later complicated by hypertension and growth retardation. Early evidence of a reduced plasma volume. Acta Obstet Gynecol Scand. 1986; 65:605–8. Available from: https://pubmed.ncbi.nlm.nih.gov/3799157/.
Balla G, Vercellotti GM, Muller-Eberhard U, Eaton J, Jacob HS. Exposure of endothelial cells to free heme potentiates damage mediated by granulocytes and toxic oxygen species. Lab Invest. 1991; 64(5):648 – 55. Available from: https://pubmed.ncbi.nlm.nih.gov/2030579/.
Ali AA, Rayis DA, Abdallah TM, Elbashir MI, Adam I. Severe anaemia is associated with a higher risk for preeclampsia and poor perinatal outcomes in Kassala hospital, eastern Sudan. BMC Res Notes. 2011;4:311. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3224576/.
Bhattacharya S, Townend J, Shetty A, Campbell D. Does miscarriage in an initial pregnancy lead to adverse obstetric and perinatal outcomes in the next continuing pregnancy? BJOG. 2008;115:1623–1629. Available from: https://pubmed.ncbi.nlm.nih.gov/18947339/.
Sepidarkish M, Almasi-Hashiani A, Maroufizadeh S, Vesali S, Pirjani R, Samani R. Association between previous spontaneous abortion and pre-eclampsia during a subsequent pregnancy. Int J Gynecol Obstet. 2016;136(1):83–86. Available from: https://pubmed.ncbi.nlm.nih.gov/28099708/.
Gunnarsdottir J, Stephansson O, Cnattingius S, Åkerud H, Wikström A: Risk of placental dysfunction disorders after prior miscarriages: a population-based study. Am J Obstet Gynecol. 2014, 211:34.e1-34.e8. Available from: https://www.ajog.org/article/S0002-9378(14)00112-4/fulltext.
Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol . 2003; 102(1):181 – 92. Available from: https://pubmed.ncbi.nlm.nih.gov/12850627/.
Pairu J, KN B, George K. Maternal and perinatal outcome in pregnancy induced hypertension and preeclampsia. Int J Reprod Contraception Obstet Gynecol. 2016;:2166–2170. Available from: https://www.ijrcog.org/index.php/ijrcog/article/view/1320.
Madazli R, Yuksel M, Imamoglu M, Tuten A, Oncul M, Aydin B, Demirayak G. Comparison of clinical and perinatal outcomes in early- and late-onset preeclampsia. Arch Gynecol Obstet. 2014; 290(1): 53–57. Available from: https://doi.org/10.1007/s00404-014-3176-x.
Wójtowicz A, Zembala-Szczerba M, Babczyk D, Kołodziejczyk-Pietruszka M, Lewaczyńska O, Huras H. Int J Hypertens. 2019: 1–9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6766116/.
K vander Tuuk, et al. Blood pressure patterns in women with gestational hypertension or mild preeclampsia. Eur J Obstet Gynecol Reproductive Biol. 2017; 210: 360 – 36. Available from: https://pubmed.ncbi.nlm.nih.gov.
Van Pampus M, Aarnoudse J. Long term outcomes after preeclampsia. Clinical Obstet Gynecol. 2005,45:489–494. Available from: https://europepmc.org.
Catov JM, Roberta B, Kevin E, Jorn. Risk of early or sever preeclampsia related to preexisting conditions. Int J Epidemiol. 2007;36(2): 412–419. Available from https://academic.oup.com.
Acknowledgements
We are grateful for Center for International Reproductive Health Training (CIRHT) for generously funding the study.
We would also like to thank all women who participated in this study for their cooperation in taking part in this study.
An abstract from this study was presented as an oral presentation during the International Federation of Gynecology and Obstetrics (FIGO) XXII World Congress 2018 at RioCentro, Brazil. https://obgyn.onlinelibrary.wiley.com/doi/abs/10.1002/ijgo.12582
Funding
Center for International Reproductive Health Training (CIRHT) funded the study. The funding agency had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
AY was the principal investigator; AY conceived the idea. AY, HT1, and SA designed the study, collected data, and wrote first and final drafts of the manuscript. SA and HT2 assisted with data collection, analysis, and manuscript development. EL assisted with design and manuscript development. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The ethics and research committee of Mekelle University approved the study (ERC0757/2016). Informed written consent was obtained from all study participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Yemane, A., Teka, H., Ahmed, S. et al. Gestational hypertension and progression towards preeclampsia in Northern Ethiopia: prospective cohort study. BMC Pregnancy Childbirth 21, 261 (2021). https://doi.org/10.1186/s12884-021-03712-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12884-021-03712-w