Abstract
Background
Reperfusion of ischemic tissue has adverse impact on the myocardium. Dexmedetomidine (Dex) is a α2-adrenergic receptor (α2-AR) agonist with sedative and analgesic effects. Macrophage migration inhibition factor (MIF) is a pressure-regulating cytokine and is responsible for inflammatory and immune diseases. This study aims to reveal the consequences of Dex on myocardial ischemia-reperfusion injury (IRI) in young mice.
Methods
Fifty mice were raised and examined. At the end of the experiment, all mice were euthanized. The anterior descending department of the left coronary artery in mice was under ischemia for 60 min, then the ligation line was released and reperfused for 120 min to establish the IRI model. Mice were randomly divided into Sham, control, treatment using 4,5-dihydro-3-(4-hydroxyphenyl)-5-isoxazoleacetic acid (ISO-1), Dex treatment, and Dex combined ISO-1 treatment groups. Interleukin (IL)-6, IL-10 and tumor necrosis factor (TNF-α) were determined by enzyme-linked immunosorbent assay (ELISA). Reactive oxygen species (ROS) and ATP levels were recorded. The expressions of MIF, P-adenosine monophosphate-activated kinase α (AMPKα), glucose transporter (GLUT)4, Bax and Bcl-2 were detected by Western Blot (WB). Hematoxylin and Eosin (H&E) staining was used to study cell morphology. Apoptosis was detected by terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) assay. Echocardiography was carried out at the end of reperfusion, and the infarct size was calculated by Electron microscopy.
Results
I/R + Dex group showed significantly increased IL-6 and TNF-α levels and reduced myocardial cell necrosis and apoptosis. H&E staining showed alleviated myocardial disorder, myocardial cell swelling, myocardial fiber fracture, and inflammatory cell infiltration in I/R + Dex group. Myocardial cell necrosis and apoptosis were significantly reduced in I/R + Dex group. ATP level in myocardial tissue of mice in I/R group was substantially decreased, while that in Dex group was increased. WB results showed that MIF, P-AMPK α, GLUT4 and Bcl-2 levels were increased and Bax levels were decreased in I/R + Dex group.
Conclusion
Dex may exert myocardial protection in young mice through MIF/AMPK/GLUT4 axis.
Similar content being viewed by others
Introduction
The main clinical manifestation of ischemic heart disorder (IHD) is acute myocardial infarction (AMI) [1], whose prognosis improves with the timing of revascularization. Paradoxically, myocardial reperfusion may be harmful because ischemia-reperfusion injury (IRI) is an oxidation-driven process that can damage other organs [2, 3]. At the same time, reperfusion of ischemic tissue can also cause irreversible harm to the myocardium, which is called cardiac ischemia reperfusion injury (IRI) [4]. This can lead to morbidity and mortality from cardiac intervention after a heart assault or stroke [5, 6], as well as different pathological conditions such as acute kidney injury, muscle injury, organ transplantation, hypovolemic shock and optionally available surgery [5, 7].
Dexmedetomidine (Dex) is a α2-adrenergic receptor (α2-AR) agonist with sedative and analgesic effects [8, 9]. At the same time, it also has cardioprotective effect and can reduce cardiac iron death and septic heart injury caused by sepsis. Macrophage migration inhibition factor (MIF) is a pressure-regulating cytokine acting as multiple roles in many inflammatory and immune diseases [10,11,12]. MIF regulates the activation of inflammatory cells and the launch of different pro-inflammatory cytokines [13], and its pathological effects have been stated in many inflammatory diseases such as rheumatoid arthritis, atherosclerosis, sepsis and cardiovascular disease [14, 15]. It has also been proven to prevent the heart from undergoing myocardial (IRI). However, the correlation between Dex and MIF and the mechanism of action remain unclear.
Glucose metabolism depends on glucose uptake by cells. However, glucose cannot enter cells freely via the lipid bilayer of cell membrane, and glucose uptake can only be carried out by means of glucosetransporters (GLUT) on cell membrane. One such protein, glucose transporter4 (GLUT4), is found in fat and muscle tissue. Study has shown that the regulation of GLUT4 in muscle requires the participation of adenosine monpophosphate-activated kinase (AMPK) [16] but its exact mechanism is unclear.
In this study, we installed the cardiac IR model of male C57BL/6 mice by ligation of the left coronary artery. The objective is to find out about the impact of Dex on cardiac IR and its molecular mechanism.
Materials and methods
Mice
This study was approved by the Animal Experimental Medical Ethics Committee of our hospital (IACUC-20170214025) and carried out in accordance with ARRIVE guidelines. All laboratory procedures were in accordance with the Guidelines for the Care and Use of Laboratory Animals. Non-pathogenic C57BL/6 mice (male; 8-week-old; 18-20 g) were purchased from Beijing Weitonglihua Experimental Animal Technology Co., LTD. (Production license No. SCXK (Beijing) 2016–0010) and raised in SPF Laboratory our university. At the end of the experience, all mice were euthanized.
Myocardial ischemia/reperfusion (I/R)
The mice were fed for 1 week, and the follow-up operations were carried out after adaptation to the environment. Briefly, mice were anesthetized with the aid of intraperitoneal injection of 1% pentobarbital sodium (60 mg/kg), intubated by tracheal intubation, and ventilated by small animal ventilator. Microscopically, the anterior descending department of the left coronary artery was seen. A 6–0 silk suture was inserted 1-2 m below the root of the left atrial appendage, and the left margin of the pulmonary conus was removed. The stitching direction was parallel to the lower edge of the left atrial appendage. The mark of successful ligation was weakened movement of the myocardium tissue around that anterior wall of left ventricle and epex. In mice, the left anterior descending department was under ischemia for 60 min and reperfused for 120 min. Mice were randomized into Shan, control, treatment using treatment using 4,5-dihydro-3-(4-hydroxyphenyl)-5-isoxazoleacetic acid (ISO-1, 35 mg/kg), Dex (20 μg/kg) treatment and Dex (20 μg/kg) combined ISO-1 (35 mg/kg) treatmemt groups.
Enzyme-linked immunosorbent assay (ELISA)
The blood samples of each group were accumulated and centrifuged (5000 g, 10 min, 4 °C) to separate the serum for Interleukin (IL)-6(cat. no. 70-EK206/3–96; MultiSciences), IL-10 (cat. no. 70-EK210/4–96; MultiSciences) and tumor necrosis factor (TNF)-α (cat. no. 70-EK282/3–96; MultiSciences) testing. These kits were used according to the operation manual.
Cell reactive oxygen species (ROS) detection
Fresh isolated left ventricular myocardial tissue was ground into single cell suspension, 2 ml phosphate-buffered saline (PBS) and 2ul 2′-7’dichlorofluorescin diacetate (DCFH-DA) were added. The cells were incubated at 37 °C for 30 min, centrifuged at 1000 rpm for 5 min, then the cells were accumulated and washed twice with PBS. After centrifugation, the cells were collected and precipitated for fluorescence detection. The wavelength was 525 nm.
Adenosine triphoshpate (ATP) content analysis
As previously described [17], ATP content was quantified by measuring the luminescence produced by the ATP-dependent luciferase bioluminescence assay. Left ventricular myocardial tissue was cryopreserved at − 80 °C and thawed immediately prior to automatic addition of the luciferase mixture. The protein was quantified in accordance with the directions of the ATP detection kit (cat. no. A095–1; Nanjing Jiancheng Technology Co., LTD), and detected at 636 nm by spectrophotometer. In order to reduce potential differences between samples, three replicates and analyses were performed.
Western blotting (WB)
Mouse left ventricular myocardial tissue samples were milled in RIPA buffer and centrifuged at 12000 rpm at 4 °C for 15 min. The supernatant was accumulated for total protein analysis, and the protein concentration was decided by bicinchoninic acid assay (BCA) method. An equivalent amount of protein (30 μg) was extracted from mouse heart homogenate, separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to PVDF membrane. It was then sealed with 5% (W/V) skim milk at room temperature (RT) for 1 h, and incubated overnight with primary antibody at 4 °C. Primary antibodies are as follows: against Bax (cat. no. ab32503; Abcam), Bcl-2 (cat. no. ab59348; Abcam), Phospho-AMPKα (cat. no. 2535S; Cell Signaling Technology, Inc.), GLUT4(cat. no. PA5–23052; Thermo Fisher Scientific), MIF (cat. no. ab65869; Abcam) and beta-Actin (cat. no. 100166-MM10; Sino Biological) were used at the dilution 1:1000. After washing with Tris-buffered saline with Tween detergent (TBST) three times for 10 min each, the membrane strips were incubated at RT with a 1:5000 dilution of an anti-rabbit IgG (cat. no. ab205718; Abcam) or anti-mouse IgG (cat. no. ab205719; Abcam) secondary antibody conjugated to horseradish peroxidase for 1 h. Protein bands were detected by Chemiscope 3000 (Shanghai, China) and the images were quantified with the use of ImageJ version 1.51 software (National Institutes of Health).
Hematoxylin and eosin (H&E) staining
Mouse hearts were fixed with 4% paraformaldehyde at RT overnight, embedded in paraffin, and sliced into 4 μm thick sections. At RT, the tissues were stained via hematoxylin (cat. no. CTS-1097; Fuzhou Maixin Biotechnology Development Co. LTD) for 5 min and eosin (cat. no. ZLI-9613; Beijing Zhongshan Jinqiao Biotechnology Co., LTD) for 1 min. The slides were visually examined under a microscope (cat. no. E200; Nikon; Japan).
Apoptosis
Terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) staining was used to assess apoptosis. The cells were immobilized in 4% paraformaldehyde at RT for 1 h and then treated with 0.5% TritonX-100 for 10 min. After washing with PBS, cells were incubated with cell death detection kits (cat. no. MK1025; BOSTER) in accordance with manufacturer’s instructions. The nuclei were stained with 0.1 g/mL DAPI (cat. no. D1306; Invitrogen) for 5 min.
Echocardiography
All mice were anesthetized with isoflurane (American veterinary anesthesia system) and detected through ultrasonic electrocardiogram (visualsonics VEVO 770 system, Canada) 40 MHz frequency scraper before death. The parameters of heart rate (HR), left ventricular end diastolic diameter and left ventricular end systolic diameter were collected. Then, the heart rate (HR), stroke volume (SV), ejection fraction (EF), fractional shortening (FS), cardiac output (CO), LVPWs (Left ventricle posterior wall thickness in systole), LVPWd (Left ventricle posterior wall thickness in diastole) and A (Aorta) were calculated as previously described to evaluate cardiac function [18].
Electron microscopy and ultrastructural study
Histological and ultrastructural research were carried out using the methods previously described by Loh et al [19]. Morphological changes were observed, and images were taken using an electron microscope (Leica, Germany).
Statistics
All statistical analyses of the data were processed in blind using Prism 8.0 software (GraphPad, San Diego, CA, USA). Data from each group were expressed as mean ± standard deviation (SD) and characterized by one-way ANOVA or Student’s t-test (and nonparametric test) for multiple comparisons. The behavioral records were statistically analyzed by two-way variance evaluation of double comparison. P < 0.05 was regarded statistical significant.
Results
Effects of Dex on serum inflammatory factor levels
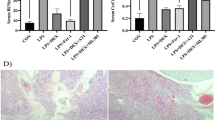
The effect of Dex on serum inflammatory cytokines was measured by ELISA. The outcomes confirmed that the production of IL-6 and TNF-α in I/R + Dex group was substantially decrease than that in Sham group (P < 0.05; Fig. 1 and Table 1), IL-10 production was considerably elevated (P < 0.05; Fig. 1 and Table 1). Further addition of an MIF inhibitor (ISO-1) substantially counteracted the effect of Dex on inflammatory levels.
Effect of Dex on myocardial histopathology in mice
H&E staining was used to assess histopathology based on morphology. In the I/R group, myocardial tissue was disordered, myocardial cells were swollen and vacuolated, some myocardial fibers were broken, and a few chronic inflammatory cells were infiltrated. Compared with I/R group, myocardial disorder, myocardial cell swelling, myocardial fiber fracture and inflammatory cell infiltration were reduced in I/R + Dex + ISO-1 and I/R + Dex groups. The lesion of I/R + Dex group was slightly milder than that of I/R + Dex + ISO-1 group (Fig. 2).
Effect of Dex on myocardial apoptosis
Immunofluorescence assay confirmed that in contrast with Sham group, myocardial cell necrosis and apoptosis were extensively enhanced in I/R group (P < 0.05; Fig. 3A, B). However, in contrast with the I/R group, necrosis and apoptosis in mice treated with Dex significantly decreased (P < 0.05). In addition, in comparison to the I/R + Dex group, cell death was significantly increased in the I/R + ISO-1 and I/R + Dex + ISO-1 groups, respectively (all P < 0.05; Fig. 3A, B). ROS detection also displayed similar trend and confirmed the above results (Fig. 3C).
Effect of Dex on ATP content in the heart
Compared with the Sham group, ATP levels in the myocardial tissue of mice in the I/R group were significantly decreased, but significantly increased after Dex treatment. ATP content in the I/R + ISO-1 group was lower than that in the I/R + ISO-1 group, and the situation improved after Dex combined with ISO-1, but ATP level was lower than that in the I/R + Dex group (Fig. 4).
Effects of Dex on ultrasound and evaluation of ultrastructure in mice
SV, EF, FS, CO and LVPWs were drastically expanded in the I/R + DEX group by cardiac ultrasound, but decreased in combination with ISO-1 (Table 2 and Fig. 5). Figure 6 shows myocardial electron microscopy images of each group. Electron micrographs of mice in the sham group showed normal cardiac structures (Fig. 6A). The I/R group showed extensive muscle necrosis, loss of lipid droplets, and giant destruction of myofilaments and Z-band structures (Fig. 6B). Similar adjustments were discovered in the ISO-1 group alone (Fig. 6C). However, the ultrastructure of I/R + Dex group and I/R + Dex + ISO-1 group were normal without swelling and vacuole formation (Fig. 6D, E).
Dex protects myocardial IRI through MIF/AMPK/GLUT4 NF-κB axis
Western blotting confirmed that in contrast with the I/R group, p-AMPKα, MIF, GLUT4 and Bcl-2 protein expressions were extensively elevated in the I/R + Dex group (P < 0.05), Bax protein was drastically diminished (P < 0.05; Fig. 7).
Discussion
IR damage causes distal organ damage, which effects in the release of reactive oxygen species (ROS) and inflammation-related molecules from ischemic tissues [20,21,22]. Tissue damage caused by ischemia is a major cause of fatal diseases such as heart attack and stroke [23]. Myocardial ischemia (MI) is reported to be one of the most vital danger elements for adverse cardiac outcomes in surgical patients with cardiovascular disease [24]. Reperfusion therapy is considered as one of the most effective method to preserve ischemic myocardium after AMI. However, restoring blood flow may lead to myocardial ischemia-reperfusion injury (MIRI) [25]. MIRI is an important issue affecting the prognosis of patients with myocardial infarction. The pathogenesis of MIRI often includes inflammatory response, apoptosis, calcium overload and the production of oxygen free radicals [26]. It can induce myocardial coma, no regurgitation, reperfusion arrhythmia, and even irreversible myocardial cell death [27]. Importantly, severe IRI to the heart can lead to everlasting incapacity or death [28]. Reperfusion injury is inevitable due to production of ROS and apoptosis of cardiomyocytes [29]. However, our study showed that Dex inhibited ROS production and myocardial cell apoptosis, which could prevent reperfusion injury to a certain extent. It has been reported that neutrophils and macrophages release ROS due to IR injury. These free radicals may lead to lipid peroxidation of cell membranes, increase in excitatory amino acids, and destruction of nucleic acids and enzymes [30].
Dex is a potent α2-AR agonist that is clinically appropriate for sedation in patients undergoing intubation and ventilator use during intensive care treatment. Currently, Dex is increasingly being introduced into the perioperative care of patients undergoing surgery [31]. In animal studies, Dex reduces organ damage. In clinical studies, Dex has been associated with improved heart surgery and transplant outcomes [31]. It has also been suggested that post-treatment of Dex leads to a significant reduction in concentration-dependent infarct size [32]. Kip G et al. [33] found that Dex could protect lung damage after myocardial ischemia-reperfusion in diabetic rats. Meanwhile, Dex has been found to have myocardial protection against hypertensive hypertrophic myocardial IRI [34]. Zhao et al. [35] found that Dex alleviated cerebral IRI in rats by inhibiting c-un N-terminal Kinase (JNK) pathway. Moreover, Dex reduces Nod-like receptor family pyrin domain containing 3 (NLRP3) through adenosine monophosphate-activated kinase (AMPK), thereby improving NLRP3 inflammasome activation in alveolar macrophages mediated by renal ischemia-reperfusion [36]. Dex alleviates endotoxin-induced acute kidney damage via enhancing autophagy and inhibiting NLRP3 inflammatory body activation through the 2-AR/AMPK/mammalian target of rapamycin (mTOR) pathway [37]. Similar to these studies, our study found that Dex protected myocardial cells from IRI through the MIF/AMPK/GLUT4 axis.
Cardiac IR triggers a massive inflammatory response, exacerbating cardiac damage and dysfunction. It has been reported that MIF expression is enhanced after ischemic heart injury in animal models [38]. A clinical research performed by Yu et al. also confirmed that plasma MIF level was intently associated with the severity of myocardial injury [39]. Activation of the MIF-AMPK pathway has been found to increase glucose transporters and glucose metabolism, reducing IRI [40]. In addition, the shielding role of MIF in acute kidney injury after cardiac surgery has been investigated [41]. Several research have reported the protective function of MIF in cardiac IR injury, including promoting glucose uptake through AMPK activation [42], inhibiting oxidative stress [43] or inhibiting JNK-mediated apoptosis [44]. Our study also found that ROS and myocardial cell apoptosis increased after inhibition of MIF, indicating that MIF could protect myocardium from reperfusion injury.
There are some limitations to this study. Although Dex has been shown in vivo to be succesful of lowering myocardial ischemia-reperfusion damage in young mice through MIF/AMPK/GLUT4 axis, it has not been tested in vitro and further studies are needed.
Conclusion
Dex reduces myocardial IRI in young mice by MIF/AMPK/GLUT4 axis. Future research evaluating the cardioprotective role of Dex in the scientific putting of MIRI is necessary.
Availability of data and materials
The data used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- Dex:
-
Dexmedetomidine
- α2-AR:
-
α2-adrenergic receptor
- MIF:
-
Macrophage migration inhibition factor
- IRI:
-
Ischemia reperfusion injury
- WB:
-
Western Blot
- IHD:
-
Ischemic heart disorder
- AMI:
-
Acute myocardial infarction
- IR:
-
Ischemia reperfusion
- GLUT:
-
Glucosetransporters
- GLUT4:
-
Glucosetransporter 4
- ELISA:
-
Enzyme-linked immunosorbent assay
- ROS:
-
Reactive Oxygen Species
- RT:
-
Room temperature
- H&E:
-
Hematoxylin and eosin
- HR:
-
Heart rate
- SV:
-
Stroke volume
- EF:
-
Ejection fraction
- FS:
-
Fractional shortening
- CO:
-
Cardiac output
- LVPWs:
-
Left ventricle posterior wall thickness in systole
- LVPWd:
-
Left ventricle posterior wall thickness in diastole
- A:
-
Aorta
- MI:
-
Myocardial ischemia
- MIRI:
-
Myocardial ischemia-reperfusion injury
References
Haidarali S, Patil CR, Ojha S, Mohanraj R, Arya DS, Goyal SN. Targeting apoptotic pathways in myocardial infarction: attenuated by phytochemicals. Cardiovasc Hematol Agents Med Chem. 2014;12(2):72–85.
Kuznetsov AV, Javadov S, Margreiter R, Grimm M, Hagenbuchner J, Ausserlechner MJ. The role of mitochondria in the mechanisms of cardiac ischemia-reperfusion injury. Antioxidants (Basel). 2019;8(10):454.
Yang M, Linn BS, Zhang Y, Ren J. Mitophagy and mitochondrial integrity in cardiac ischemia-reperfusion injury. Biochim Biophys Acta Mol basis Dis. 2019;1865(9):2293–302.
Marin W, Marin D, Ao X, Liu Y. Mitochondria as a therapeutic target for cardiac ischemia-reperfusion injury (review). Int J Mol Med. 2021;47(2):485–99.
Chouchani ET, Pell VR, James AM, Work LM, Saeb-Parsy K, Frezza C, et al. A unifying mechanism for mitochondrial superoxide production during ischemia-reperfusion injury. Cell Metab. 2016;23(2):254–63.
Lesnefsky EJ, Chen Q, Tandler B, Hoppel CL. Mitochondrial dysfunction and myocardial ischemia-reperfusion: implications for novel therapies. Annu Rev Pharmacol Toxicol. 2017;57:535–65.
Eltzschig HK, Eckle T. Ischemia and reperfusion--from mechanism to translation. Nat Med. 2011;17(11):1391–401.
Keating GM. Dexmedetomidine: a review of its use for sedation in the intensive care setting. Drugs. 2015;75(10):1119–30.
Coursin DB, Coursin DB, Maccioli GA. Dexmedetomidine. Curr Opin Crit Care. 2001;7(4):221–6.
Li JH, Tang Y, Lv J, Wang XH, Yang H, Tang PMK, et al. Macrophage migration inhibitory factor promotes renal injury induced by ischemic reperfusion. J Cell Mol Med. 2019;23(6):3867–77.
Calandra T, Roger T. Macrophage migration inhibitory factor: a regulator of innate immunity. Nat Rev Immunol. 2003;3(10):791–800.
Baugh JA, Bucala R. Macrophage migration inhibitory factor. Crit Care Med. 2002;30(1 Supp):S27–35.
Calandra T, Bernhagen J, Metz CN, Spiegel LA, Bacher M, Donnelly T, et al. MIF as a glucocorticoid-induced modulator of cytokine production. Nature. 1995;377(6544):68–71.
Morand EF, Leech M, Bernhagen J. MIF: a new cytokine link between rheumatoid arthritis and atherosclerosis. Nat Rev Drug Discov. 2006;5(5):399–410.
Zernecke A, Bernhagen J, Weber C. Macrophage migration inhibitory factor in cardiovascular disease. Circulation. 2008;117(12):1594–602.
Ojuka EO, Jones TE, Nolte LA, Chen M, Wamhoff BR, Sturek M, et al. Regulation of GLUT4 biogenesis in muscle: evidence for involvement of AMPK and ca(2+). Am J Physiol Endocrinol Metab. 2002;282(5):E1008–13.
Van Blerkom J, Davis PW, Lee J. ATP content of human oocytes and developmental potential and outcome after in-vitro fertilization and embryo transfer. Hum Reprod. 1995;10(2):415–24.
Nagel E, Lehmkuhl HB, Bocksch W, Klein C, Vogel U, Frantz E, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation. 1999;99(6):763–70.
Loh HK, Sahoo KC, Kishore K, Ray R, Nag TC, Kumari S, et al. Effects of thalidomide on isoprenaline-induced acute myocardial injury: a haemodynamic, histopathological and ultrastructural study. Basic Clin Pharmacol Toxicol. 2007;100(4):233–9.
Tural K, Ozden O, Bilgi Z, Kubat E, Ermutlu CS, Merhan O, et al. The protective effect of betanin and copper on heart and lung in end-organ ischemia reperfusion injury. Bratisl Lek Listy. 2020;121(3):211–7.
Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524–51.
Yang M, Chen J, Zhao J, Meng M. Etanercept attenuates myocardial ischemia/reperfusion injury by decreasing inflammation and oxidative stress. PLoS One. 2014;9(9):e108024.
Dalen JE, Alpert JS, Goldberg RJ, Weinstein RS. The epidemic of the 20(th) century: coronary heart disease. Am J Med. 2014;127(9):807–12.
Mangano DT, Browner WS, Hollenberg M, London MJ, Tubau JF, Tateo IM. Association of perioperative myocardial ischemia with cardiac morbidity and mortality in men undergoing noncardiac surgery. The study of perioperative ischemia research group. N Engl J Med. 1990;323(26):1781–8.
Zhao Z, Sun W, Guo Z, Liu B, Yu H, Zhang J. Long noncoding RNAs in myocardial ischemia-reperfusion injury. Oxidative Med Cell Longev. 2021;2021:8889123.
Vincent A, Covinhes A, Barrère C, Gallot L, Thoumala S, Piot C, et al. Acute and long-term cardioprotective effects of the traditional Chinese medicine MLC901 against myocardial ischemia-reperfusion injury in mice. Sci Rep. 2017;7(1):14701.
Zhang C, Sun A, Zhang S, Yao K, Wu C, Fu M, et al. Efficacy and safety of intracoronary autologous bone marrow-derived cell transplantation in patients with acute myocardial infarction: insights from randomized controlled trials with 12 or more months follow-up. Clin Cardiol. 2010;33(6):353–60.
Li X, Liu M, Sun R, Zeng Y, Chen S, Zhang P. Protective approaches against myocardial ischemia reperfusion injury. Exp Ther Med. 2016;12(6):3823–9.
Zhao T, Wu W, Sui L, Huang Q, Nan Y, Liu J, et al. Reactive oxygen species-based nanomaterials for the treatment of myocardial ischemia reperfusion injuries. Bioact Mater. 2022;7:47–72.
Weigand K, Brost S, Steinebrunner N, Büchler M, Schemmer P, Müller M. Ischemia/reperfusion injury in liver surgery and transplantation: pathophysiology. HPB Surg. 2012;2012:176723.
Yuki K. The immunomodulatory mechanism of dexmedetomidine. Int Immunopharmacol. 2021;97:107709.
Bunte S, Behmenburg F, Majewski N, Stroethoff M, Raupach A, Mathes A, et al. Characteristics of Dexmedetomidine Postconditioning in the field of myocardial ischemia-reperfusion injury. Anesth Analg. 2020;130(1):90–8.
Kip G, Çelik A, Bilge M, Alkan M, Kiraz HA, Özer A, et al. Dexmedetomidine protects from post-myocardial ischaemia reperfusion lung damage in diabetic rats. Libyan J Med. 2015;10:27828.
Yoshikawa Y, Hirata N, Kawaguchi R, Tokinaga Y, Yamakage M. Dexmedetomidine maintains its direct Cardioprotective effect against ischemia/reperfusion injury in hypertensive hypertrophied myocardium. Anesth Analg. 2018;126(2):443–52.
Zhao B, Li D, Zhang S, He L, Ai Y. Dexmedetomidine attenuates cerebral ischemia-reperfusion injury in rats by inhibiting the JNK pathway. Ann Palliat Med. 2021;10(6):6768–78.
Chen Y, Huang Y, Xiong B, Luo H, Song X. Dexmedetomidine ameliorates renal ischemia reperfusion-mediated activation of the NLRP3 inflammasome in alveolar macrophages. Gene. 2020;758:144973.
Yang T, Feng X, Zhao Y, Zhang H, Cui H, Wei M, et al. Dexmedetomidine enhances autophagy α2-AR/AMPK/mTOR pathway to inhibit the activation of NLRP3 Inflammasome and subsequently alleviates lipopolysaccharide-induced acute kidney injury. Front Pharmacol. 2020;11:790.
Yu C-M, Lai KW-H, Chen Y-X, Huang X-R, Lan HY. Expression of macrophage migration inhibitory factor in acute ischemic myocardial injury. J Histochem Cytochem. 2003;51(5):625–31.
Yu CM, Lau CP, Lai KW, Huang XR, Chen WH, Lan HY. Elevation of plasma level of macrophage migration inhibitory factor in patients with acute myocardial infarction. Am J Cardiol. 2001;88(7):774–7.
Wang J, Tong C, Yan X, Yeung E, Gandavadi S, Hare AA, et al. Limiting cardiac ischemic injury by pharmacological augmentation of macrophage migration inhibitory factor-AMP-activated protein kinase signal transduction. Circulation. 2013;128(3):225–36.
Stoppe C, Averdunk L, Goetzenich A, Soppert J, Marlier A, Kraemer S, et al. The protective role of macrophage migration inhibitory factor in acute kidney injury after cardiac surgery. Sci Transl Med. 2018;10(441):eaan4886.
Miller EJ, Li J, Leng L, McDonald C, Atsumi T, Bucala R, et al. Macrophage migration inhibitory factor stimulates AMP-activated protein kinase in the ischaemic heart. Nature. 2008;451(7178):578–82.
Koga K, Kenessey A, Powell SR, Sison CP, Miller EJ, Ojamaa K. Macrophage migration inhibitory factor provides cardioprotection during ischemia/reperfusion by reducing oxidative stress. Antioxid Redox Signal. 2011;14(7):1191–202.
Qi D, Hu X, Wu X, Merk M, Leng L, Bucala R, et al. Cardiac macrophage migration inhibitory factor inhibits JNK pathway activation and injury during ischemia/reperfusion. J Clin Invest. 2009;119(12):3807–16.
Acknowledgements
None.
Funding
This work was funded by the National Natural Science Foundation of China (Grant number 81760044 & 81960053).
Author information
Authors and Affiliations
Contributions
SC guaranteed the integrity of the entire study and contributed to the study concepts, design, manuscript preparation and editing. AL together with TZ contributed to the interpretation of data. JW dedicated to the concept and intellectual content. YH carried out the literature research and contributed to the conception of the study. TT dedicated to the data acquisition and analysis. JW reviewed the manuscript. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Experiments were performed under a project license (IACUC-20170214025) granted by Animal Experimental Medical Ethics Committee of The First Affiliated Hospital of Xinjiang Medical University in compliance with institutional guidelines of The First Affiliated Hospital of Xinjiang Medical University for the care and use of animals.
Consent for publication
Not applicable.
Competing interests
The authors declare that there is no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Chen, S., Li, A., Wu, J. et al. Dexmedetomidine reduces myocardial ischemia-reperfusion injury in young mice through MIF/AMPK/GLUT4 axis. BMC Anesthesiol 22, 289 (2022). https://doi.org/10.1186/s12871-022-01825-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12871-022-01825-z