Abstract
As a routine, karyotyping of invasive prenatal samples is performed as an adjunct to referrals for DNA mutation detection and metabolic testing. We performed a retrospective study on 500 samples to assess the diagnostic value of this procedure. These samples included 454 (90.8%) chorionic villus (CV) and 46 (9.2%) amniocenteses specimens. For CV samples karyotyping was based on analyses of both short-term culture (STC) and long-term culture (LTC) cells. Overall, 19 (3.8%) abnormal karyotypes were denoted: four with a common aneuploidy (trisomy 21, 18 and 13), two with a sex chromosomal aneuploidy (Klinefelter syndrome), one with a sex chromosome mosaicism and twelve with various autosome mosaicisms. In four cases a second invasive test was performed because of an abnormal finding in the STC. Taken together, we conclude that STC and LTC karyotyping has resulted in a diagnostic yield of 19 (3.8%) abnormal cases, including 12 cases (2.4%) with an uncertain significance. From a diagnostic point of view, it is desirable to limit uncertain test results as secondary test findings. Therefore, we recommend a more targeted assay, such as e.g. QF-PCR, as a replacement of the STC and to provide parents the autonomy to choose between karyotyping and QF-PCR.
Similar content being viewed by others
Background
Currently, there is no evidence available in the literature indicating that the prevalence of chromosomal abnormalities is higher in pregnancies with a referral for DNA mutation or metabolic testing. Although the European cytogenetic guidelines for prenatal diagnosis [1] indicate that both DNA mutation and metabolic testing do not serve as referral categories for traditional karyotyping (TK), most prenatal centres worldwide routinely offer TK as an additional test. In clinical practice, most couples referred for DNA mutation analysis also opt for TK [2].
It can be disputed, however, whether TK is required when there is no a priori increased risk for chromosomal anomalies as compared to the normal population. On the other hand, it has been argued that when a risky invasive prenatal test is performed anyway, it is unethical not to concomitantly exclude the occurrence of putative chromosomal abnormalities [3]. With TK, a wide range of chromosomal abnormalities can be detected, including alterations in copy number (aneuploidy) and structural chromosomal rearrangements such as translocations and inversions, being either balanced or unbalanced. Targeted PCR-based assays such as multiplex ligation-dependent probe amplification (MLPA) or quantitative fluorescent PCR (QF-PCR), are highly suited for rapid aneuploidy detection (RAD) of the chromosomes 21, 18, 13, X and Y [4–12]. Previously, it has been suggested that if the referral reason is an increased risk of Down's syndrome, resulting from a positive screening test result or an advanced maternal age, karyotyping could effectively be replaced by RAD, provided that no structural fetal abnormality has been detected upon ultrasound examination [5, 13–17]. The use of RAD as a targeted, standalone test instead of karyotyping when invasive prenatal testing is performed in cases with DNA mutation or metabolic test referrals, has not been studied before. This retrospective study addresses the clinical impact of TK for samples offered for prenatal diagnosis with a molecular or metabolic referral.
Methods
The data in this retrospective study were obtained from the patient database of the Department of Human Genetics, Radboud University Nijmegen Medical Centre, the Netherlands. All procedures were performed with ethical approval from the local ethical committees. In the period January 1994 to July 2010, 500 samples from pregnant women undergoing chorionic villus (CV) sampling or amniocentesis were examined. The reason for invasive diagnostic testing was a referral for fetal DNA mutation detection, metabolic diagnostics or other "non-cytogenetic" reasons, with or without advanced maternal age (AMA). The samples were from hospitals participating in the Network Prenatal Diagnostics Nijmegen (NPDN): Radboud University Nijmegen Medical Centre, Rijnstate Hospital Arnhem, St. Elisabeth Hospital and TweeSteden Hospital Tilburg, Medical Spectrum Twente Enschede, Jeroen Bosch Hospital 's-Hertogenbosch, and some other hospitals in the Netherlands.
CV samples were split into two portions, one for DNA mutation or metabolic analysis and one for TK. On CV samples, both short-term cultures (STC) and long-term cultures (LTC) were performed. Amniotic fluid (AF) cells were cultured for TK and, simultaneously, for DNA isolation. DNA mutation analyses were performed in different diagnostic centres in the Netherlands. For metabolic testing, (cell-free) AF was used.
Karyotyping was performed following standard procedures. The results were reviewed retrospectively and classified as normal or abnormal. Additional or follow-up studies, such as parental karyotyping in case of a structural rearrangement or the presence of a marker chromosome, and fluorescence in situ hybridization (FISH) in case of insufficient test results, were performed before a definite prenatal karyotype result and its interpretation were reported. In some cases a second prenatal invasive procedure was performed. The test results of DNA mutation and metabolic testing were retrieved from the patient's records.
Results
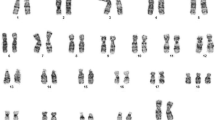
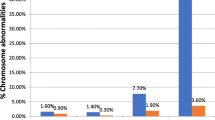
The prenatal test results of 454 (90.8%) chorionic villus (CV) and 46 (9.2%) amniotic fluid (AF) samples (500 in total) were assessed. DNA mutation analysis referrals classified in normal and abnormal karyotypes are shown in Table 1. Overall, 481 (96.2%) normal karyotypes (46, XX or 46, XY) were found. The most common reasons for DNA testing were Fragile X syndrome and Huntington disease (11.4% and 7.8%, respectively). Overall, TK resulted in 19 (3.8%) abnormal karyotypes (18 CV samples and 1 AF sample). The maternal age at sampling was ≥ 36 years in 117 (23.4%) samples and resulted in 8 (6.8%) abnormal karyotypes (mean maternal age 37.9 years, median 37.5 years). The maternal age at sampling was < 36 years in 383 (76.6%) samples, and resulted in 11 (2.9%) abnormal karyotypes (mean maternal age 29.9 years, median age 30.0 years) (Figures 1 and 2). The 19 cytogenetic abnormal samples are listed in Tables 2 and 3. These included all, except one, CV samples: 4 cases with a common aneuploidy (trisomy 21, 18 and 13), 2 cases with a sex chromosomal aneuploidy (Klinefelter syndrome), one sex chromosomal mosaicism in amniotic fluid cells and different autosomal mosaicisms in 12 CV samples. These latter samples included four aberrations present in STC but absent in LTC, 4 aberrations present in LTC but absent in STC and 4 aberrations present in both STC and LTC.
In 4 of the aberrations present in STC but not in LTC, additional testing by amniocenteses showed a normal fetal karyotype and, therefore, the mosaicism in the CV samples appeared to be a confined placental mosaicism (CPM type I).
In 3 of the 4 aberrations present in LTC but not in STC, DNA mutation analyses were positive and the respective pregnancies were terminated. Therefore no cytogenetic follow-up testing was performed. In the 4th case the percentage of tetraploid cells in the LTC was 46%. Although tetraploid cells are common in CV samples, particularly in LTC, this high percentage was a reason to report this finding and to continue pregnancy follow-up by ultrasound examination. The child was born pre-term at 33 weeks of pregnancy without congenital anomalies.
The abnormalities detected in both STC and LTC included two low-grade sex chromosome mosaicisms (cases 15 and 16). Case 15 appeared positive upon DNA mutation analysis. Therefore the pregnancy was terminated. In case 16 only 1 out of 4 metaphases in STC and 1 out of 28 metaphases in LTC showed a 45, X karyotype, all other metaphases were 46, XY. No follow-up amniocentesis was performed and the pregnancy was followed by ultrasound examination showing a normal male fetus. The 3rd sample showed a paternally inherited 45, X, inv(Y) karyotype. The 4th showed a marker chromosome in a single metaphase in both STC and LTC. The parental karyotypes were normal. The fetus appeared positive upon DNA mutation testing. The pregnancy was terminated.
Taken together, we conclude that karyotyping resulted in a diagnostic yield of 19 (3.8%) abnormal karyotypes, including 12 (2.4%) representing CV mosaicisms giving rise to uncertainty to prospective parents about the fetal prognoses.
Discussion
In case an invasive prenatal test is performed for DNA mutation or metabolic testing, most prenatal centres offer traditional karyotyping (TK) as an additional test to exclude the presence of chromosome abnormalities. There is also no evidence from the literature that the prevalence of chromosomal abnormalities is higher in pregnancies with a referral for DNA mutation or metabolic analysis. The majority of the cases included in our current study represented CV samples (91.2%) because DNA isolation for mutation detection can be performed immediately after tissue sampling and a diagnostic result can be obtained in the first trimester. In contrast, AF cells have to be cultured first and, hence, a diagnostic result can only be obtained in the second trimester. In total, 19 (3.8%) abnormal karyotypes were denoted. When no cytogenetic testing was performed in the group of pregnant women < 36 years 11 (2.9%) cytogenetic aberrations would remained undetected. In 8 of these 11 it was uncertain whether the abnormal karyotype represented the true genetic constitution of the fetus due to mosaicisms. Mosaicisms are thought to be present in ~1% of CV samples, and have been confirmed in the fetus in 5-25% of these cases [18, 19]. In our study population of 454 CV samples 12 (2.6%) showed a chromosome mosaicism with uncertain clinical impact. Uncertain diagnostic results are leading to parental anxiety and mostly require follow-up testing. For example in cases 9 and 11 (Tables 2 and 3), next to additional TK of amniotic fluid, additional DNA testing is required to exclude uniparental disomies (UPD) for chromosomes 7 and 15. Prenatal UPD(7) testing, however, is questionable considering the mild phenotype [20]. The risk of UPD(15) when trisomy 15 mosaicism has been detected, upon CV or AF analysis, has been estimated to range from 11% to 29% [21, 22].
The overall percentage of mosaicism in our study (2.4%) score slightly higher as the 1-2% reported in the literature [18, 19, 23]. A possible explanation is the use of both the STC and LTC procedure. As individual laboratories use different protocols to examine CV samples (LTC only, STC combined with LTC or QF-PCR alone), the incidence of mosaicism is likely to vary to some degree between laboratories.
With the implementation of targeted molecular-cytogenetic tests such as MLPA and QF-PCR, the question arises whether TK should remain the gold standard or could be replaced by RAD for pregnancies at risk for common aneuploidies. RAD test results are unequivocal and available within 1-2 days, which considerably reduces parental anxiety [16]. In case of thalassaemia, for example, RAD has already shown to be the best approach for the detection of chromosomal abnormalities when prenatal invasive testing is performed [3]. Next to the possibility to recognise maternal cell contamination, RAD is also cost-effective particularly when performed large scale [24]. For pregnancies with an increased risk of Down syndrome, a change of policy from full karyotype analysis to rapid molecular aneuploidy testing would result in a failure to detect chromosome abnormalities that may have clinical consequences. This residual risk has been estimated to be 0.07% [25–27]. Next to the debate of targeted RAD replacing TK there is a discussion whether the scope of diagnostic testing should be broader than karyotyping. Although broadening the scope of testing benefits in terms of clinically relevant findings that would otherwise be missed, a serious challenge is that, with the present state of knowledge, results of such testing could be difficult to interpret [28], creating parental anxiety, uncertainty and unnecessary termination of pregnancy. Our overall tendency towards broadening the scope of prenatal testing is using genome-wide microarray analysis in high-risk pregnancies (e.g. with fetal ultrasound abnormalities) and narrowing (targeting) the scope in low-risk pregnancies.
In our prenatal diagnostic service, RAD (QF-PCR) is already implemented as replacement for the STC in all CV samples and offered as stand-alone test for pregnancies with an increased risk of Down syndrome, such as a positive screening test result or an advanced maternal age, as a test of choice. With the application of a cell dissociation protocol steps are taken to prevent fully discrepant results between cytotrophoblasts and mesenchymal core cells for the chromosomes 13, 18, 21, X and Y. Herewith, the cytotrophoblast cells (analyzed in STC) and mesenchymal core cells (analyzed in LTC) can be tested for RAD separately [29, 30] and, thus, discrepancies as in cases 8 and 10 would have been detected if RAD were used. The application of RAD to replace karyotyping in both STC and LTC will prevent findings without clinical relevance and/or uncertain outcome.
Limitations of this retrospective study should be addressed. First, this study does not represent all pregnant women primary referred for molecular or metabolic testing. Some of the pregnancies were terminated because of a positive DNA mutation result and, therefore, the impact of the respective mosaicisms could not be evaluated. Secondly, although we already implemented QF-PCR as stand-alone test in our routine prenatal practice as a test of choice for pregnant women with an increased risk for Down syndrome, the QF-PCR test was not performed on the cases presented in this study.
Taken together, we conclude that there is additional diagnostic value of TK. However, there is also a need to limit uncertain test results as secondary findings. In this population with a low risk for a chromosomal aneuploidy, we recommend to implement QF-PCR as replacement of the STC and to give parents the autonomy to choose between karyotyping and QF-PCR.
The results of this study are in line with those reported by Tse et al [3], showing that RAD seems to be the best approach for the detection of chromosomal abnormalities when invasive prenatal testing is performed for the diagnosis of thalassaemia. The broader application of this study may create awareness and reconsideration of national standards for prenatal cytogenetic testing, a step towards international harmonized procedures and prenatal care.
Abbreviations
- AF:
-
amniotic fluid
- AMA:
-
advanced maternal age
- CPM:
-
confined placental mosaicism
- CV:
-
chorionic villus
- FISH:
-
fluorescence in situ hybridization
- LTC:
-
long-term culture
- MLPA:
-
multiplex ligation-dependent probe amplification
- NPDN:
-
Network Prenatal Diagnostics Nijmegen
- QF-PCR:
-
quantitative fluorescent polymerase chain reaction
- RAD:
-
rapid aneuploidy detection
- STC:
-
short-term culture
- TK:
-
traditional karyotyping
- UPD:
-
uniparental disomy.
References
Cytogenetic Guidelines and Quality Assurance: A common European framework for quality assessment for constitutional and acquired cytogenetic investigations[http://e-c-a.eu/]
Eason J: Prenatal diagnosis of single gene disorders. Obstet Gynecol Reprod Med 2010, 20: 155–160. 10.1016/j.ogrm.2010.02.004
Tse KY, Leung WC, Leung KY, Lee CP, Ng LK, Lau ET, Chan V, Tang MH: Full karyotyping rapid aneuploidy diagnosis or both when invasive prenatal testing is performed for diagnosis of thalassaemia? Mol Hum Reprod 2006, 12: 55–59. 10.1093/molehr/gal003
Gerdes T, Kirchhoff M, Lind AM, Vestergaard LG, Kjaergaard S: Multiplex ligation-dependent probe amplification (MLPA) in prenatal diagnosis- experience of a large series of rapid testing for aneuploidy of chromosomes 13 18 21 X and Y. Prenat Diagn 2008, 28: 1119–1125. 10.1002/pd.2137
Kooper AJA, Faas BHW, Kater-Baats E, Feuth T, Janssen JC, van der Burgt I, Lotgering FK, Geurts van Kessel AG, Smits APT: Multiplex ligation- dependent probe amplification (MLPA) as a stand-alone test for rapid aneuploidy detection in amniotic fluid cells. Prenat Diagn 2008, 28: 1004–1010. 10.1002/pd.2111
Fauret AL, Bilan F, Patri S, Couet D, Marechaud M, Pierre F, Gilbert- Dussardier B, Kitzis A: Molecular biology usefulness for rapid diagnosis of Down's syndrome and common aneuploidies. Gynecol Obstet Fertil 2009, 37: 611–619. 10.1016/j.gyobfe.2009.05.007
Van Opstal D, Boter M, de Jong D, van den Berg C, Bruggenwirth HT, Wildschut HI, de Klein A, Galjaard RJ: Rapid aneuploidy detection with multiplex ligation-dependent probe amplification: a prospective study of 4000 amniotic fluid samples. Eur J Hum Genet 2009, 17: 112–12. 10.1038/ejhg.2008.161
Cirigliano V, Voglino G, Ordonez E, Marongiu A, Paz Canadas M, Ejarque M, Rueda L, Lloveras E, Fuster C, Adinolfi M: Rapid prenatal diagnosis of common chromosome aneuploidies by QF-PCR results of 9 years of clinical experience. Prenat Diagn 2009, 29: 40–49. 10.1002/pd.2192
Wang J, Cui YX: Advances in rapid prenatal detection of fetal chromosome abnormalities. Zhonghua Nan Ke Xue 2010, 16: 359–363.
Hills A, Donaghue C, Waters J, Waters K, Sullivan C, Kulkarni A, Docherty Z, Mann K, Ogilvie CM: QF-PCR as a stand-alone test for prenatal samples: the first 2 years' experience in the London region. Prenat Diagn 2010, 30: 509–517.
Boormans EM, Birnie E, Oepkes D, Galjaard RJ, Schuring-Blom GH, van Lith JM: Comparison of multiplex ligation-dependent probe amplification and karyotyping in prenatal diagnosis. Obstet Gynecol 2010, 115: 297–303. 10.1097/AOG.0b013e3181cbc652
Gekas J, van den Berg DG, Durand A, Vallee M, Wildschut HI, Bujold E, Forest JC, Rousseau F, Reinharz D: Rapid testing versus karyotyping in Down's syndrome screening: cost-effectiveness and detection of clinically significant chromosome abnormalities. Eur J Hum Genet 2011, 19: 3–9. 10.1038/ejhg.2010.138
Leung WC, Lau ET, Lao TT, Tang MHY: Can amnio-polymerase chain reaction alone replace conventional cytogenetic study for women with positive biochemical screening for fetal Down syndrome? Obstet Gynecol 2003, 101: 856–861. 10.1016/S0029-7844(03)00222-9
Leung WC, Lao TT: Rapid aneuploidy testing traditional karyotyping or both? Lancet 2005, 366: 97–98. 10.1016/S0140-6736(05)66791-8
Ogilvie CM: Prenatal diagnosis for chromosome abnormalities: past present and future. Pathol Biol 2003, 51: 156–160. 10.1016/S0369-8114(03)00017-8
Ogilvie CM, Lashwood A, Chitty L, Waters J, Scriven FF: future of prenatal diagnosis: rapid testing or full karyotype? An audit of chromosome abnormalities and pregnancy outcomes for women referred for Down's syndrome testing. BJOG 2005, 112: 1369–1375. 10.1111/j.1471-0528.2005.00695.x
Boormans EM, Birnie E, Oepkes D, Bilardo CM, Wildschut HI, Creemers J, Bonsel GJ, van Lith JM: The impact of rapid aneuploidy detection (RAD) in addition to karyotyping versus karyotyping on maternal quality of life. Prenat Diagn 2010, 30: 425–433.
Ledbetter DH, Zachary JM, Simpson JL, Golbus MS, Pergament E, Jackson L, Mahoney MJ, Desnick RJ, Schulman J, Copeland KL: Cytogenetic results from the US Collaborative Study on CVS. Prenat Diagn 1992, 12: 317–345. 10.1002/pd.1970120503
Stetten G, Escallon CS, South ST, McMichael JL, Saul DO, Blakemore KJ: Reevaluating confined placental mosaics. Am J Med Genet A 2004, 131: 232–239.
Kotzot D: Prenatal testing for uniparental disomy: indications and clinical relevance. Ultrasound Obstet Gynecol 2008, 31: 100–105. 10.1002/uog.5133
EUCROMIC: European Collaborative Research on Mosaicism in CVS Trisomy 15 CPM: probable origins pregnancy outcome and risk for fetal UPD. Prenat Diag 1999, 19: 29–35.
Christian SL, Smith AC, Macha M, Black SH, Elder FF, Johnson JM, Resta RG, Surti U, Suslak L, Verp MS, Ledbetter DH: Prenatal diagnosis of uniparental disomy 15 following trisomy 15 mosaicism. Prenat Diagn 1996, 16: 323–232. 10.1002/(SICI)1097-0223(199604)16:4<323::AID-PD856>3.0.CO;2-5
Grati FR, Grimi B, Frascoli G, Di Meco AM, Liuti R, Milani S, Trotta A, Dulcetti F, Grosso E, Miozzo M, Maggi F, Simoni G: Confirmation of mosaicism and uniparental disomy in amniocytes after detection of mosaic chromosome abnormalities in chorionic villi. Eur J Hum Genet 2006, 14: 282–288. 10.1038/sj.ejhg.5201564
Grimshaw GM, Szczepura A, Hultén M, MacDonald F, Nevin NC, Sutton F, Dhanjal S: Evaluation of molecular tests for prenatal diagnosis of chromosome abnormalities. Health technol assess 2003, 7: 1–100.
Ogilvie CM, Lashwood A, Chitty L, Waters JJ, Scriven PN, Flinter F: The future of prenatal diagnosis: rapid testing or full karyotype? An audit of chromosome abnormalities and pregnancy outcomes for women referred for Down's Syndrome testing. BJOG 2005, 112: 1369–1375. 10.1111/j.1471-0528.2005.00695.x
Shaffer LG, Bui TH: Molecular cytogenetic and rapid aneuploidy detection methods in prenatal diagnosis. Am J Med Genet C Semin Med Genet 2007, 145C: 87–98. 10.1002/ajmg.c.30114
Boormans EM, Birnie E, Oepkes D, Galjaard RJ, Schuring-Blom GH, van Lith JM: MLPA and Karyotyping Evaluation (M.A.K.E.) Study Group. Comparison of multiplex ligation-dependent probe amplification and karyotyping in prenatal diagnosis. Obstet Gynecol 2010, 115: 297–303. 10.1097/AOG.0b013e3181cbc652
de Jong A, Dondorp WJ, Frints SG, de Die-Smulders CE, de Wert GM: Advances in prenatal screening: the ethical dimension. Nat Rev Genet 2011, 12: 657–663. 10.1038/nrg3036
Mann K, Kabba M, Donaghue C, Hills A, Ogilvie CM: Analysis of a chromosomally mosaic placenta to assess the cell populations in dissociated chorionic villi: implications for QF-PCR aneuploidy testing. Prenat Diagn 2007, 27: 287–289. 10.1002/pd.1663
Kooper AJA, Feuth T, Creemers JW, Zondervan HH, Boekkooi PF, Quartero RW, Rijnders RJ, van der Burgt I, Geurts van Kessel AG, Smits APT: Detection of Chromosome Aneuploidies in Chorionic Villus Samples by Multiplex Ligation-Dependent Probe Amplification. J Mol Diagn 2009, 11: 17–24. 10.2353/jmoldx.2009.070140
Acknowledgements
The authors wish to thank their colleagues from the Network Prenatal Diagnosis Nijmegen (NPDN) in Arnhem (Rijnstate Hospital), Tilburg (St. Elizabeth Hospital, TweeSteden Hospital), 's-Hertogenbosch (Jeroen Bosch Hospital), Enschede (Medical Spectrum Hospital Twente) and Nijmegen (Radboud University Nijmegen Medical Centre) for the prenatal material and the relevant follow up data, and Prof. dr A. Geurts van Kessel for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors' contributions
AK and AS drafted the manuscript. AS, BF and AK evaluated the cytogenetic results, LH the DNA diagnostic test results. AS, BF, LH, HZ and JP have critically reviewed and approved the manuscript. HZ clinically examined the pregnancy and performed the majority of the invasive procedures. IvdB referred and counselled most of the prospective parents and clinically examined the fetus. All authors read and approved the manuscript.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Kooper, A.J., Pieters, J.J., Faas, B.H. et al. Is routine karyotyping required in prenatal samples with a molecular or metabolic referral?. Mol Cytogenet 5, 7 (2012). https://doi.org/10.1186/1755-8166-5-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1755-8166-5-7