Abstract
Background
The Anopheles dirus complex includes efficient malaria vectors of the Asian forested zone. Studies suggest ecological and biological differences between the species of the complex but variations within species suggest possible environmental influences. Behavioural variation might determine vector capacity and adaptation to changing environment. It is thus necessary to clarify the species distributions and the influences of environment on behavioural heterogeneity.
Methods
A literature review highlights variation between species, influences of environmental drivers, and consequences on vector status and control. The localisation of collection sites from the literature and from a recent project (MALVECASIA) produces detailed species distributions maps. These facilitate species identification and analysis of environmental influences.
Results
The maps give a good overview of species distributions. If species status partly explains behavioural heterogeneity, occurrence and vectorial status, some environmental drivers have at least the same importance. Those include rainfall, temperature, humidity, shade, soil type, water chemistry and moon phase. Most factors are probably constantly favourable in forest. Biological specificities, behaviour and high human-vector contact in the forest can explain the association of this complex with high malaria prevalence, multi-drug resistant Plasmodium falciparum and partial control failure of forest malaria in Southeast Asia.
Conclusion
Environmental and human factors seem better than species specificities at explaining behavioural heterogeneity. Although forest seems essential for mosquito survival, adaptations to orchards and wells have been recorded. Understanding the relationship between landscape components and mosquito population is a priority in foreseeing the influence of land-cover changes on malaria occurrence and in shaping control strategies for the future.
Similar content being viewed by others
Background
Throughout most of their geographical distribution, species of the Anopheles dirus complex are associated with high malaria prevalence rates and the occurrence of drug resistant Plasmodium falciparum [1, 2]. The biological specificities of these efficient vectors undermine the most popular control measures and challenge the success of malaria control. Sporadic studies on sympatric sibling species from the complex suggest ecological and biological differences in types of larval habitat, seasonality and behaviour according to species [3–5] but such differences also occur for specific species within their distributions and could relate to key environmental factors. Variation in behaviour, such as early biting or ovipositing in wells, might determine vector capacity and adaptation to changing environment. It is thus of interest to clarify the geographical distributions of the species, the importance of environmental factors and the influences of ecological variation on behavioural heterogeneity.
If the biology of An.dirus s.l. (sensu lato, i.e. An. dirus complex) is well documented in the literature, no attempt has been made recently to compile this information. A major difficulty resides in the taxonomic changes which have affected the group throughout the last 50 years [4, 6, 7]. Furthermore, only recently did molecular tools allow identification of individuals from this group up to the species level and they rely on strenuous methods or require sophisticated equipment. As a result, most of the available articles do not provide specific species identification.
Fortunately, the taxonomy of the complex has recently been clarified and the species named [4, 8]. The complex belongs to the Anopheles (Cellia) leucosphyrus group in the Neomyzomyia Series [6] and now includes at least seven species: Anopheles dirus or An. dirus sensu stricto (s.s.), Anopheles crascens, Anopheles scanloni, Anopheles baimaii, Anopheles elegans ( previously known as species E), Anopheles nemophilous and Anopheles takasagoensis [4, 8]. The species previously called Anopheles elegans from Sri Lanka [9] and Southern India [10] has now been renamed Anopheles mirans and is not part of the complex [4]. Evolution of the complex and correspondence with historical names are presented in Figure 1.
Based on those taxonomic clarifications, existing distribution maps of these species [11] can now be updated using data from collection sites available in the literature and from the recent project MALVECASIA on monitoring insecticide resistance in Southeast Asia. Localisation of collection sites can facilitate identification of the species in articles lacking this information and provide the background for analysis of spatial distribution, biology, behaviour, vectorial status and key ecological factors of individual species.
This article thus aims to, 1) update the distributions of members of the An. dirus complex using literature records and personal data, 2) to provide an overview of intra- and inter- species variation of biology and behaviour and 3) to identify key ecological factors influencing the behaviour distribution, occurrence and vectorial status of An. dirus s.l.
Methods
The basis for the paper is a comprehensive analysis of more than a hundred articles examined for the following items: information on geographical dispersion, species identification methods, behaviour of adults and larvae, and environmental factors influencing occurrence and behaviour. Key papers on taxonomy and population structure were first reviewed to associate historical species records with currently recognized species.
More than five hundred collection sites were then spatially located. They include literature records but also recent data from the MALVECASIA network, a research network of eight partners that studied the distribution and insecticide resistance of malaria vectors in Vietnam, Cambodia, Laos and Thailand. This information was gathered in a table, and maps were produced to plot the collection sites on a vegetation background adapted from the global dataset Global Land Cover 2000[12]. This background represents what is thought to be a forested habitat from a mosquito's point of view. It includes the following original classes: evergreen and deciduous forest, open or closed, including mixed leaf type, flooded forest, mosaic of tree cover and other natural vegetation as well as mosaic of cropland, tree cover and other natural vegetation. The accuracy of site location depends on the available information: the maximum accuracy is obtained when the coordinates are provided in the original article. In most of the other cases, the extended Geographical Information System (SEAGIS) gathered and organized by the MALVECASIA network [13] provided the necessary tools to find an accurate location. Maps or detailed site descriptions were compared with datasets such as village databases, administrative maps, roads, rivers, vegetation and altitude. Some collection sites could not be accurately located using the above mentioned methods which made it necessary to look for locations in gazetteers [14].
For each site number, the sibling species identified in the reviewed publication is recorded, as well as the identification methods. Considerable uncertainty may result from some of these methods [15] and the distinction between An. dirus s.s. and An. scanloni remains problematic. Morphological keys based on reared adults with associated larval and pupal exuviae[16], polytene chromosomal banding patterns[17], enzyme electromorph [18], allele-specific polymerase chain reaction (ASPCR) [15, 19] and random amplified polymorphic DNA (RAPD) [20] are a few of the existing identification methods. Sallum et al. provided a complete list [4]. Species status can be extrapolated from the map, for sites located in allopatric zones.
The species spatial distribution is first discussed. Then larval and adult ecology and behaviour are analysed to highlight possible variation between species. The influence of environmental drivers and land-cover is then considered as well as the consequences for vector status and control.
Results and Discussion
Species spatial distribution
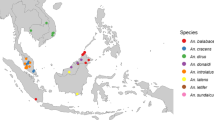
A map first shows the overall extent of the An. dirus complex distribution (Figure 2). A second map focuses on Southeast Asia, where the species diversity and the number of records are higher (Figure 3). In this area, the presence of An. dirus s.l. seems to correspond to presence of malaria cases. The additional file 1 allows better reading of the maps. It indicates the reviewed publication, the species discovered and the identification method used for each site. As it was not graphically possible to display individually the more than 500 sites, sites close to each other are represented by a single point on the map and a single reference number. The additional files (Additional file 2, 3 and 4) associated with the current article provide a description of the site, collection methods and the number of An. dirus s.l. collected, as well as the original location for each of the 500 sites organized by site number or by reviewed publication. The current article is also aimed at malaria control workers, and with this information, they can find the situation recorded at any of the collection sites in the past, as well as locate any collection site cited in the publications reviewed.
Detailed distribution of the species from the Anopheles dirus complex. Distribution of the sibling species of the Anopheles dirus complex depicted on a vegetation background and some indication of important temperature and rainfall thresholds. Each point represents one or several collection sites from the literature and from personal recent data. Details for each site are available in table 1 and in the additional file provided with this article. The details for the Southeast Asian region are shown in figure 3.
Species from the Anopheles dirus complex in the Southeast Asian region. Distribution of the sibling species of the Anopheles dirus complex depicted on a vegetation background. Each point represents one or several collection sites from the literature and from personal recent data. Details for each site are available in table 1 and in the additional file provided with this article.
Species of the An. dirus complex can be found in forest and forest foothills from India to Taiwan and from the 30° north parallel to the Malaysian peninsular. The mean monthly temperature below 20° seems to limit the northern distribution of the complex to just beyond the border of India with Nepal and Bhutan. Rainfall is probably the limiting factor to the west with annual rainfall per year under 800 mm. The absence of species of the complex in large non-forested areas of Thailand, southern Vietnam and central India is probably linked to the lack of suitable habitat. However the species of the complex are also absent from the north of Vietnam. This is most peculiar, given that this area is still forested and that members of the complex occur at the same latitude in neighbouring countries. The possible occurrence of An. dirus s.l. in central India was investigated by Srivastava [21]. A predictive model identifying suitable factors for Anopheles survival and the distribution of wet evergreen and deciduous forest confirmed the presence of An. dirus s.l. in already well-known areas of occurrence, but also in a few suitable sites through central and North-Western India. This seems to correspond to historical collection sites in Uttar Pradesh, Kerala and Karnakata. Bhat [22] and Rajavel [7] also cite historical records in Kasauli and other regions now considered free of An. dirus s.l. Recent reports of the presence of An. dirus s.l. in the state of Jharkhand (Dr. Diwakar Dinesh, personal communication) call for further investigation into the actual distribution of the complex in India. Those areas are presented in Figure 2 as sites with presence suspected but not confirmed.
Regarding the species distributions within the range of the complex, there are no major geographic or topographic reasons that seem to justify the current species distribution pattern. Therefore, speciation probably does not result from adaptation to a specific environment but the species might have been isolated for some time. Their distribution most probably reflects geo-morphological changes which have occurred in the past and are today not evident [3]. The mountains of the Western Ghats in southern India are the most westward limit of the complex with the presence of An. elegans [10, 23] apparently isolated from the other species. The closest records to this area come then from the north eastern states in India [24] and the border of Nepal [25], and are considered to be An. baimaii based on identification in nearby Bangladesh [26]. An. baimaii seems to be the main species in Myanmar, occurring throughout the country, then giving way to An. dirus s.s. eastward from western Thailand. An. dirus s.s. is the only species recorded so far in Vietnam, Lao, Cambodia [27–29] and Hainan Island [30]. An. crascens is confined to southern Thailand and Malaysia[31], and An. takasagoensis to Taiwan [32]. An. scanloni and An. nemophilous seem to have more patchy distributions. An. scanloni is restricted to western and southern Thailand, whereas the distribution of An. nemophilous closely follows monsoon forests. The south of Thailand and the Thai – Myanmar border present various sites of high sympatry for several species of the complex [3].
Some clarifications are still called for. Variations of ITS2 rDNA usually occur only between sibling species, thus intraspecific variations between populations of An. baimaii from Thailand and site 302 in China, and An. scanloni at sites 175 and 195 suggest the possible existence of two new species [15], with a less diverse population at site 195 indicating a period of isolation[33].
There is need for some investigations at the Thai-Cambodian border, the border between Lao and China and some areas in Myanmar. Indeed, although only An. dirus s.s seems to occur in Cambodia, two specimens of An. baimaii were earlier recorded near to the border in Thailand at site 241 [34] and specimens at the border have been observed to feed on monkeys in the canopy as would An. nemophilous in nearby Thailand [35]. Only An. dirus s.s. has been recorded in Lao although An. baimaii occurs nearby in China. Unexpected records such as An. dirus s.s. at site 49 [36] and 148 [37], and An. scanloni at site 126 [37], should be taken with caution and investigated further. This is also the case for records of An. baimaii at site 241, which are based on the collection of only two specimens. Sallum et al. [4] reported the presence of An. crascens in Sumatra but gave no reference.
Larval habitats
Primary and secondary larval habitats of An. dirus s.l. have some constant characteristics: temporary, standing or slowly moving water under shade. While primary sites occur year round and tend to be associated with the stream system in deep forest, they vary in nature according to the season with drying pools in stream beds, pools connected with streams or meanders of slow moving streams in the dry season [38–40], complemented in the rainy season by pools fed by underground seepage along streams, springs, rock pools in the beds of ravines, and in deep holes or pits which form ahead of gullies [32, 39, 41]. Such primary sites are often confined to the deep forest resulting in transmission of malaria mainly to forest workers. Some of them however occur in peri-domestic area: An. dirus s.s. occurs in gem pits [42] and in water pits fed by seepage along streams [43]. If wells are generally negative[39, 44], An. baimaii occurs in wells close to houses in a particular area of Myanmar throughout the year [36, 45–47] even if densities are reduced in the dry season.
Secondary larval habitats occur in the rainy season and can be found closer to human settlement at the forest fringe. These are commonly small, shallow, temporary, shaded water-holding depressions [48]. Puddles in paths are commonly positive, whereas larger bodies of water such as ponds, large rivers, irrigation channels and marshy areas are generally negative. However, An. takasagoensis breeds in large permanent pools used by buffaloes for bathing if freshened by the rain [32]. An. dirus s.l. was also recorded in long marshy areas in the forest of Myanmar [41] and in two swamps and one rice paddy in Thailand[49]. Artificial containers [39] and natural containers are also generally negative but An. dirus s.s. is found reported in clay jars in Hainan[2], in a tin-hat[40], empty tins and in terracotta jars in Vietnam[50]. Positive natural containers include only bamboo stumps [5, 36, 40] and palm leaves [40]. Details on larval habitats and locations investigated are presented in Figure 4.
Immature stages of An. dirus s.s. and An. baimaii show adaptations to temporary habitats [39, 43]. The eggs survive on the moist floor of a drained pool up to one month after the rain stops, but not throughout the dry season, unless sporadic rains prevent the soil from drying or unless the eggs are on the wet soils of forest. Not only do the eggs survive between rainfalls but they can also partially mature on a moist surfaces thus reducing development times, with first stage larvae present in pools containing no water prior to rain [39, 43]. Larval stages can crawl to other pools in case of drying but they can also survive the desiccation of a draining pool and ant predation by getting covered in a coat of mud and becoming buried and motionless just before complete drainage. On re-flooding, only An. dirus s.l. reappears at the surface thus eliminating other competitors. An. dirus s.l. immature stages can feed on the larvae of other species and less frequently on its own larvae, in particular under crowded conditions [39]. Increased larval density results in higher larval mortality[36] and the production of smaller adults even with the same amount of food per larva, suggesting an influence on feeding efficiency rather than a shortage of food per se [51].
For a sibling species, larval habitats seem quite diverse according to location but Baimai [3] recorded a preference for vegetated limestone rock pools for An. scanloni and rocky-stony micro-environments for An. dirus s.s., An. crascens and An. baimaii. Sympatric populations of An. dirus s.s. and An. baimaii share identical larval habitat and even the same pools at site 204 [5]. An. baimaii was, however, more often found in pools that dried out in dry spells during the rainy season. This might reflect a seasonal variation in larval habitats or species, with An. dirus s.s. more abundant at the start and An. baimaii during the middle of the rainy season [3].
Adult particularities and behaviour
An. baimaii and An. dirus s.s. are extremely anthropophilic [1, 52–55], even in the presence of numerous cattle [56], but less anthropophilic behaviour has been sporadically recorded for these two species [27, 57]. In particular, higher densities were recorded on cattle in some years [46] or every year for An. baimaii [58]. Some studies show that An. nemophilous feeds primarily on monkey, and An. dirus s.s. and An. scanloni feed more often on humans than An. crascens. These experiments were however based on few observations[3]. Monkeys are natural and winter hosts [2, 32, 59] but preference for monkey was not evident from human- and monkey-bait collections in the forest. More specimens were caught on human bait on the ground. As many were caught on monkey bait in the canopy as on human bait on the ground [35]. Anecdotal records report individual mosquitoes feeding on birds [56], dogs and pigs [41], as well as mixed feeding on bovines and humans suggesting that mosquitoes do not always come back to the same host[57, 60, 61].
Females are primarily exophagic but enter open shelters to feed [62]. Variations between regions are linked to housing facilities with the highest endophagy in largely open houses built directly on the ground [53]. Most open houses in the jungle show no significant biting differences between indoors and outdoors [1, 56, 57, 63, 64]. In some cases, indoor biting is even higher [38, 43, 65].
Resting places are difficult to find and occur mainly outdoors [58, 59, 66] in holes, wells[36, 58, 63]and vegetation such as bushes, tree holes [41, 52], thick grass, under the surfaces of leaves [36, 40], branches near the ground [40] and under the foots of trees[2]. In India, an intensive search collected none on the ground but 20 from the moist, dark crevices of large tree trunks, and 2 from creepers in the forest, the nearest at 150 m from a village[66]. Day-resting can be separated from night-resting[67]. Females rest early in the evening in outdoor vegetation, fences and wood stacked around houses [52, 56] but after feeding, they fly almost immediately back to the jungle[1], with none found even at dawn around dwellings [43, 52]. Eyles [35] caught more An. dirus s.l. in the canopy than on the ground and when Wilkinson [43] released hundreds of females in predawn darkness, they flew immediately upwards into trees suggesting that they might rest in the canopy of trees.
Biting can happen in daylight in the jungle[38, 59] but occurs mostly from dusk to dawn. From the feeding patterns reported in the literature and presented in Figure 5, it is not obvious whether or not An. dirus s.l. is a late night feeder or an early night feeder. Scanlon [62] emphasised that late night feeding is most common in Thailand but that occasionally local populations exhibit a striking early pattern. The peak feeding activity has also been recorded either late [52] or early [68] in the same location for different years. Species-specific patterns of outdoor biting were suggested for allopatric populations with a very early peak for An. scanloni, and An. crascens, around 22h for An. dirus s.s. and 02h for An. baimaii [3]. An. dirus s.s. and An. baimaii are, however, both often recorded as late night feeders [1, 2, 27, 35, 48, 69, 70], but peak activity was also recorded well before midnight for An. takasagoensis and An. baimaii [32, 41, 46, 57, 71]. The occurrence of a particular sibling species can thus not explain all the variation. Other influencing factors seem to include the moon phase, the season and the presence of DDT, which stimulates early biting [59, 63]. A two-year survey at site 12 recorded wide a repeated variation of the feeding pattern in accordance with the moon phase [59]. Variations have also been recorded between years and seasons [34, 36, 46, 59] with earlier peak activity in the dry season[27] or earlier peak activity in October than in June [63]. Late biting in post-monsoon in some regions might also be due to the late onset of dawn [60]. Compilation of data across years, seasons, moon phases or between indoor and outdoor biting can thus result in a wide variety of patterns that are difficult to interpret [64]. The Relative Risk (RR) of being bitten in the hour before 22h compared with an hour after 22h probably provides a better indicator of exposure. A large proportion of bites before 22h coincide with activities of people before bedtime when the bed-net protection is nil [53]. Indoor biting peaks occur often later than outdoor biting [59] but in the presence of very open houses no difference is observed [53]. Outdoor biting has been recorded as starting as early as 16h for An. scanloni [3], and 19h for An. baimaii, An. dirus s.s. and An. takasagoensis [1, 32, 38, 43, 46, 72].
Biting behaviour of species of the Anopheles dirus complex. Figure 5 lists the hourly biting pattern of sibling species by collection sites (Site + number) and reviewed publication [reference]. The number of specimens collected (when available) gives an indication of the level of confidence. The data presented in the source article were converted to hourly percentages to allow comparison of data from different sources. A shaded symbol provides an easy reading of peak period and intensity of biting with decreasing density. Information on the types of collections, i.e. Outdoor Human collection (OH), Indoor Human collection (IH), Human collection (HC) and CDC light trap (CDC), is presented in the description section.
Environmental drivers
The occurrence of different sibling species can explain part of the heterogeneity in behaviour. However, differences between individuals of the same species underlines the major role of environmental factors in determining the occurrence, distribution, seasonality, behaviour and vectorial status for An. dirus s.l. The influence of moon phase and housing facilities on biting behaviour has already been discussed. Other keys factors of importance are temperature, rainfall, topography, season, soil type, shade, water quality and land-cover. These factors interact with components of the mosquito life cycle on one hand and with different stages of the parasite cycle on the other.
The occurrence of An. dirus s.l. is mainly linked to rainfall, temperature and relative humidity [46]. Attacks started after the rain exceeded 50 mm at site 12 for An. baimaii [39]. Lower or different thresholds seemed to apply according to the soil type and topography with, for example, An .dirus s.l. in Hainan [73]. Heavy rains however, flush away larval habitats, impede mosquitoes from flying [3, 46, 74] and the flooded grounds can be unsuitable for weeks[43]. The pattern of rainfall might, thus, be more important than the amount of rainfall, and light rains, not too frequent, seem to be most favourable for larval development [32]. Kitthawee [51] observed that adult body size was positively correlated with rainfall 1–2 weeks before. She suggests that renewed rainfall may bring more particulate food onto the surface of the water where larvae feed. Conversely, excessive rain could dilute nutrients and lead to nutritionally stressed, smaller adults. The importance of the rainfall pattern can explain yearly fluctuations in populations recorded during different studies in the same region [28, 29, 64]. The month of peak densities is not constant but usually takes place during the rainy season, with up to 80 bites per man/night [75]. Variations of relative abundance of sibling species in sympatric sites show more An. crascens than An. baimaii at the beginning and less An. crascens than An. scanloni at the end of the wet season at site 175. At site 204, more An. dirus s.s. were found at the start and more An. baimaii towards the middle of the wet season [3]. Populations of An. takasagoensis also present high fluctuations in abundance. The species usually is fairly uncommon but is occasionally very abundant [32].
Temperature is rarely a limiting factor but it influences the longevity of the mosquito, the length of the sporogonic cycle and mosquito activity, thus influencing seasonally the vector status.An. dirus s.l. seems inactive when temperature falls below 15°C [27]. However, in the deep forest of northern Thailand, during the cool, dry season associated with important temperature fluctuations and minima around 10°C, An. dirus s.l. can still survive and even transmit malaria. [63]. The duration of the immature stages is reduced at higher temperatures but females are then smaller [36]. If size does not influence the number of oocysts [76], larger mosquitoes live longer and have thus a greater vectorial capacity [77]. Mean daytime temperature averages 25.4°C with up to 5°C variation in positive ground pools [43]. However, in colonised wells temperature remains almost constant (26.2°C), despite outside temperature oscillations from 19.5 to 33.1°C. The clay contained in the lateritic wall lining the wells might have a sustained cooling effect[47].
Topography, salinity, pH and shade also influence the availability and occurrence of larval habitat. Topography is a major element with sites found commonly in foothills where rain water can accumulate, next to streams or in the beds of ravines. Rosenberg observed that the appearance of waves of An. dirus s.l. after the rain is not systematic at site 12 and requires three concomitant elements: rapidly draining pools, intermittent, heavy rains and embryonated eggs [39]. If suitable pools are temporary, they should hold water for at least five to eight days [39, 66]. Suitability of a site might, thus, be a combination of the clay content of the soil with the appropriate amount and frequency of rain. In the sandy soils of site 12, suitable larval habitats occur only on the compacted soil of the path. The infiltration is too high elsewhere. Where An. baimaii breeds year-long in wells, the soil outside is not appropriate for larval habitat because it dries out very rapidly by percolation and evaporation. Even puddles in path are negative [46] and most wells dry up in summer [45]. Kitthawee [42] noticed that site such as 148 and 243 which presents the particular behaviour of year-long larval development in gem pits or wells also present a particular environmental setting with very high rainfall and infiltration rates.
Salinity, pH, shade and temperature have been extensively analysed in the context of An. bamaii in wells in Myanmar. Nitrate, iron, dissolved oxygen, sulphate, chlorine, ammonia and water hardness seem to have no influence, but larval density is negatively correlated to pH in ground pools and salinity in wells, with a threshold at 200 ppm NaCl [36, 45, 47, 49]. Larval density becomes very low when the distance between the well-water surface and the ground surface is less than 2.5 m, probably reflecting an effect of shade and temperature [36, 47]. Larvae gradually disappear if the shade is removed. Except for a few records [5, 69], most larval habitats are under the shade of trees which probably reduces the drying speed of pools, influences temperature and provides food through organic matter and leaves falling into the water. Positive wells have shade and shrub on inner walls, debris on the water surface and often abundant decaying leaves at the bottom, even if the water is clean [36]. Numerous larvae are even observed in pools often fouled by buffaloes [2], but An. dirus s.l. will not be encountered if the water is not freshened frequently by rains [48].
Association with forest
The most important environmental parameter is definitely land-cover. Throughout its distribution An. dirus s.l. is associated with forested foothills, forests or forest fringes. If availability of larval habitats and the presence of natural hosts, such as monkeys [35] are explanatory factors for this association, adults may also require the highest humidity and lower temperature of the jungle biotope for optimal survival [2]. Favourable environmental conditions of dense vegetation, humid soil, high relative humidity and shade, coupled with the presence of permanent suitable larval habitats or primary sites, appear to persist deep inside the forest during the dry season. Although rainfall in a tropical rain forest is unpredictable, the forest floor is humid. Even if the larval habitats loose their free water, the high humidity probably keeps the eggs viable for fairly long periods until the next rain comes[2]. The tree cover provides food for larvae, with leaves and other debris falling into larval habitats and assures stable micro-climatic conditions, even in the dry season. As the rainy season begins, conditions also become favourable at the margins of forest and An. dirus s.l. seems to spill over from the forest into secondary larval habitats.
Much larger densities of An. dirus s.s. are present in deep forest settlements or villages than in villages located at the edge of forest or in fragmented forests [78]. Variations in forest are difficult to interpret when mosquito densities are low near a village, very high at 1.5 km and moderate at 5 km[1, 35, 78, 79], Infected mosquitoes can be present at oviposition sites 1.5 to 3 km away from the village [59]. Terracotta jars provided for oviposition in the forest attracted the highest number of mosquitoes at 150 m from the village but hardly any 300 m away [29]. Specimens were captured up to 2 km away from a source during a mark-release-recapture study [80]. These complex and contradicting results show that all the parameters influencing the occurrence and density of the An. dirus s.l. populations in the forest are not totally understood but the distance to the potential host and suitability of oviposition site certainly play a major role. The association with the forest is high in any case and almost systematically results in high vector contact and malaria transmission.
Species such as An. nemophilous seem to be dependant on monsoon forest[3]. In Cholbury, Thailand, large populations of An. dirus s.l. were encountered by Scanlon [52] in 1964, but the site has been further deforested and the population has been considerably reduced. However, adaptation to other land-cover has been recorded, particularly to teak and rubber plantations and orchards. The mosquitoes can adapt to the edge of man-made clearings [39, 48, 81] and have once been recorded in rice fields [49]. However, it is not known,, if large forested areas are necessary for the survival of these vector populations or if fragmented forest or plantations might be sufficient. The particular behaviour of year-long oviposition in wells might be a consequence of an adaptation to a new type of larval habitat and resting places that provide a cooler and more constant temperature, corresponding to the forest biotope [47].
Vector status and control
The role played by An. dirus s.l. in the transmission of malaria has only been assessed during the last 50 years. It is now considered as the most important vector in Southeast Asia. Several factors contribute to making species of the An. dirus s.l. complex an exceptionally efficient vectors: they are so long lived and highly anthropophilic that only small populations are necessary to maintain high malaria endemicity [81]. Their exophilic behaviour, early biting habits and insecticide avoidance undermine the efficiency of the most common vector control measures e.g. insecticide residual spraying and insecticide impregnated nets. High human/vector contact in the typical forest biotope inhabited by species of this complex can explain the extended occurrence of what has been called "forest malaria".
The forest activities of humans play a major role in the malaria epidemiology of Southeast Asia [82]. The colonization of new land for agriculture, logging, mining and other activities, as well as resettlement of populations in the forest, expose people to high transmission risks in the most favoured biotope of An. dirus s.l Overnight stays in the open for hunting and collecting fruits in forest increase the human/vector contact, and open temporary shelters and forest huts facilitate early indoor biting when people are not yet protected by bed-nets[40]. The invasion of the jungle by human settlers most probably increases the densities of these mosquitoes by providing hosts and the small transitory pools that are preferred for oviposition [39]. During the dry season, people are mainly getting malaria in the deep forest [63] where infected vectors [43] are commonly found all year round near permanent streams [39]. Malaria attacks occur in villages only during the rainy season, when An. dirus s.l. moves back to the valley and forest fringe and where secondary larval habitats become available [59]. People move between villages and semi-permanent huts in forest and migrations between infected areas in the forest and non-infected areas trigger the start of transmission in the forest fringe and its surroundings once conditions there become favourable.
The vector density peak occurs generally one month before the malaria incidence peak [40]. However, large populations of mosquitoes are, not required for maintaining a high level of transmission [1, 29, 61, 71]. Hence, small populations of mosquitoes might not be detected in short-term surveys as abundance and presence vary greatly between years, seasons and even from one week to another. In some regions, females feed late at night and may be missed unless night-long collections are made [62]. The sporozoite rates of An. dirus s.l. vary with season and location, with the highest rates recorded in October (7.8%) at site 17 [75] and rates up to 14% in forested site 22 [41]. Rosenberg [81] found high variation between villages 800 m away from each other with a sporozoite rate three to four times greater in the site of lower abundance. Sporozoites of P.vivax and P.falciparum have been commonly detected in An. baimaii and An. dirus s.s Baimai [83] reported sporozoites in An. scanloni and An. crascens with slight differences between species in relation to the parasite. An. dirus s.s. developed Plasmodium vivax and P. falciparum oocysts more readily than An. crascens and An. scanloni. An. elegans, An. nemophilous, and An. takasagoensis probably only transmit simian malaria [83].
Foci of chloroquine resistance have been commonly associated with An. dirus s.l. [1, 2, 70, 84]. Wilkinson [85] carried out an experiment in a highly endemic area for chloroquine resistant strains of P. falciparum and showed that 66% of An. dirus s.l. and 44% of An. minimus became infected when fed on the same infected patients. When comparing the infected mosquitoes, the number of oocysts was also higher in An. dirus s.l Trung [29] recorded P.falciparum, P.vivax-210 and P.vivax-247 circumsporozoite protein (CSP) in a single An. dirus s.l. mosquito. The great longevity of An. dirus s.l., its high susceptibility to Plasmodium infections and a tendency to develop high numbers of oocysts increases the risk of recombining parasite strains in the mosquito gut and as consequence the risk for a fast spread of multi-drug resistance.
Alternative methods to human landing collection should be used where multi-drug resistance is present. CDC light traps have been used successfully on other species in Africa [86, 87] and on An. dirus s.l. indoors in India [60] but other studies show less positive results when comparing various trapping method to human landing collections [88, 89]. Alternative methods to human landing collection such as CDC light trap should be further evaluated as possible tool for monitoring vector control programmes.
An. dirus s.l. is susceptible to DDT [36, 41, 43, 50] but due to exophilic behaviour, females avoid treated walls [43, 52, 63, 90] or even avoid the sprayed huts by biting more outdoors after residual spraying [59, 91] and high sporozoite rates may persist after application of DDT [2]. Insecticide impregnated bed-nets were proved to be effective [84] if kept in good condition [92], however, early biting habits in some areas exposes people to bites before bed time. Insecticide-treated hammocks and personal protection might thus be more effective. Alternative methods such as vegetation clearing are difficult to apply to such diffuse temporary larval habitats[2]. Treating the vegetation surrounding houses to target resting females would probably fail due to rapid loss of insecticide to rain and rapid vegetation growth [62].
Conclusion
An. dirus s.s and A. bamaii of the An.dirus complex are certainly the most efficient malaria vectors in Asia. Human activities in the jungle create high human/vector contact exposing people with poor shelter conditions in forested habitat, leading to perennial transmission. The efficiency of these species, as malaria vectors is largely explained by biological particularities. These species are highly susceptible to malaria parasites, there are highly anthropophilic, and have an excellent survival rate required for the sporogonic cycle. These vectors are difficult to control regarding the dispersion of temporary larval habitats in the forest, and their exophilic behaviour by which the mosquitoes entering the house will avoid any contact with indoor insecticide treated surfaces. Their relative early biting behaviour, preferably outdoors, may hamper the efficacy of insecticide treated nets (ITN). However, as these vectors are very sensitive to pyrethroids and almost exclusively anthropophilic, scaling up of ITNs will probably affect locally the An.dirus s.l. populations.
The distributions of species of the An. dirus complex have been thoroughly analysed in Thailand, Malaysia and recently in other areas of Southeast Asia following extended surveys from the Malvecasia project, but the distributions of members of the complex in the western region rely on few records and should be further investigated. Historical records and references from the literature have been used to delimitate the maximum extent of the complex distribution, but they do not reflect the current situation as major changes in land-cover have occurred in the region.
An. dirus s.l. is strongly associated with deep forest larval habitats and probably also requires deep forest for adult survival. It can survive year round wherever evergreen forest occurs. Drastic deforestation in recent decades has considerably reduced suitable habitats but adaptation of the species to man-made habitats such as orchards and plantations greatly increases the human/vector contact and suggests high plasticity in habitat requirements. However, such adaptations might only occur in areas where some of the environmental conditions, particularly micro-climatic conditions (e.g. wells, orchards) are still suitable.
High heterogeneity in behaviour has been recorded for mosquitoes of the An. dirus complex. The assumption that the recent discovery of seven cryptic species would explain most of this heterogeneity is challenged by the high behavioural differences recorded for different populations of the same species. An. baimaii specimens are developing in wells or forest habitats. Biting peaks vary from early to late within individual species and seasonality seems more linked to environmental factors than to species distribution. This could be a consequence of incomplete characterization of the group due to imperfect identification methods. Indeed, new molecular tools suggest the existence of two more species within the complex. Population history and phylogenetic relationships between the species are not straightforward and are sometimes even contradictory [33, 93]. Behavioural differences between sibling species can only be analysed when they occur in sympatry, and very few sympatric populations have been studied. Allopatric species such as An. dirus s.s. and An. crascens are impossible to compare.
Environmental factors play a major role in intraspecific heterogeneity by interacting with the immature and adult stages. The most important factors are rainfall, which provides larval habitats and keeps growth conditions optimal by refreshing the sites and providing food, and the land-cover with conditions being are optimal year-long in the forest and seasonally in forest fringes. Temperature, topography, soil type, salinity and drainage also have an influence.
Environmental proxies might be relevant factors in a preliminary approach to establish approximate limits to the possible extension in the flexible distribution range of An. dirus s.l. However, variation in transmission dynamic occurs at very small spatial and temporal scales and can only be understood by studying micro-environmental parameters in details and in relation to human factors such as housing, settlement location in relation to the forest, occupations and migrations. This highlights the importance of micro-environmental variations on mosquito populations in a region that is currently undergoing major land-cover changes. Forest fragmentation and changes in land occupation influence habitat suitability for members of the complex. Although forest seems to be essential for mosquito survival, adaptations to orchards and wells have been recorded. Understanding the relationship between landscape components and mosquito population is thus a priority in foreseeing the impact of the land-cover changes on malaria occurrence and in shaping control strategies for the future.
References
Gould DJ, Scanlon JE, Ward RA: Anopheles vectors of malaria in Southeast Asia. Army Science Conference Proceedings. 1966, 1: 361-373.
Yang TH: A review of literature on Anopheles balabacensis balabacensis. World Health Organization. WHO/MAL/83.999, WHO/MAL/83.873. 1983, 30-
Baimai V, Kijchalao U, Sawadwongporn P, Green CA: Geographic distribution and biting behaviour of four species of the Anopheles dirus complex (Diptera: Culicidae) in Thailand. Southeast Asian J Trop Med Pub Hlth. 1988, 19: 151-161.
Sallum MAM, Peyton EL, Wilkerson RC: Six new species of the Anopheles leucosphyrus group, reinterpretation of An. elegans and vector implications. Med Vet Entomol. 2005, 19: 158-199. 10.1111/j.0269-283X.2005.00551.x.
Rattanarithikul R, Green CA, Panyim S, Noigamol C, Chanaimongkol S, Mahapibul P: Larval habitats of malaria vectors and other anopheles mosquitoes around a transmission focus in northwestern Thailand. J Am Mosq Control Assoc. 1995, 11: 428-433.
Peyton EL: A new classification for the leucosphyrus group of Anopheles (Cellia). Mosq Syst. 1989, 21: 197-205.
Rajavel AR, Das PK: A review of Leucosphyrus Group with particular reference to the Anopheles dirus complex (Diptera: Culicidae) in India. J Commun Dis. 1998, 30: 57-67.
Harbach RE: The classification of genus Anopheles (Diptera: Culicidae): a working hypothesis of phylogenetic relationships. Bull Entomol Res. 2004, 94: 537-553. 10.1079/BER2004321.
Mendis KN, Ihalamulla RL, Peyton EL, Nanayakkara S: Biology and descriptions of the larva and pupa of Anopheles (Cellia) elegans James (1903). Mosq Syst. 1983, 15: 318-324.
Tewari SC, Hirikan J, Reuben R: Survey of the anopheline fauna of the Western Ghats in Tamil Nadu, India. Indian J Malariol. 1987, 24: 21-28.
Baimai V: Heterochromatin Accumulation and karyotypic evolution in some dipteran insects. Zoological studies. 1998, 37: 75-88.
Stibig H-J, Upik R, Beuchle R, Hildanus H, Mubareka S: The land cover map for Southeast Asia in the year 2000. GLC2000 database, European Commission Joint Research Centre. 2003
MALVECASIA Network. [http://www.itg.be/malvecasia/]
Global gazetteer version 2.1. [http://www.fallingrain.com/world/]
Walton C, Handley JM, Kuvangkadilok C, Collins FH, Harbach RE, Baimai V, Butlin RK: Identification of five species of the Anopheles dirus complex from Thailand, using allele-specific polymerase chain reaction. Med Vet Entomol. 1999, 13: 24-32. 10.1046/j.1365-2915.1999.00142.x.
Peyton EL, Ramalingam S: Anopheles (Cellia) nemophilous, a new species of the Leucosphyrus Group from peninsular Malaysia and Thailand (Diptera: Culicidae). Mosq Syst. 1988, 20: 273-299.
Baimai V, Harrison BA, Somchit L: Karyotype differentiation of three anopheline taxa in the Balabacensis Complex of Southeast Asia (Diptera: Culicidae). Genetica. 1981, 57: 81-86. 10.1007/BF00131231.
Green CA, Munstermann LE, Tan SG, Panyim S, Baimai V: Population genetic evidence for species A, B, C and D of the Anopheles dirus complex in Thailand and enzyme electromorphs for their identification. Med Vet Entomol. 1992, 6: 29-36.
Audtho M, Tassanakajon A, Boonseng V, Tpiankijagum S, Panyim S: Simple nonradioactive DNA hybridization method for identification of sibling species of Anopheles dirus (Diptera: culicidae) complex. J Med Entomol. 1995, 32: 107-111.
Manguin S, Kengne P, Sonnier L, Harbach RE, Baimai V, Trung HD, Coosemans M: SCAR markers and multiplex PCR-based identification of isomorphic species in the Anopheles dirus complex in Southeast Asia. Med Vet Entomol. 2002, 16: 46-54. 10.1046/j.0269-283x.2002.00344.x.
Srivastava A, Nagpal BN, Saxena R, Subbarao SK: Predictive habitat modelling for forest malaria vector species An. dirus in India – A GIS-based approach. Current Sciences India. 2001, 80: 1129-1134.
Bhat HR: A note on Anopheles dirus Peyton and Harrison, 1979 [An. balabacensis (sensu lato) Baisas, 1936] in India. Indian J Malariol. 1988, 25: 103-105.
Sawadipanich Y, Baimai V, Harrison BA: Anopheles dirus species E: chromosomal and crossing evidence for another member of the dirus complex. J Am Mosq Control Assoc. 1990, 6: 477-481.
Nandi J, Rao JS, Dasgupta RK, Sharma RS: Ecological observations on the anopheline mosquitoes of Jalpaiguri Duars, West Bengal. J Commun Dis. 1996, 28: 279-286.
Darsie RF, Pradhan JR, Pradhan SP: The mosquitoes of Nepal: their identification, distribution and biology. Mosq Syst. 1990, 22: 69-130.
Baimai V, Thu MM, Paing M, Maheswary NP: Distribution and chromosomal polymorphism of the malaria vector Anopheles dirus species D. Southeast Asian J Trop Med Pub Hlth. 1988, 19: 661-665.
Toma T, Miyagi I, Okazawa T, Kobayashi J, Saita S, Tuzuki A, Keomanila H, Nambanya S, Phompida S, Uza M, Takakura M: Entomological surveys of malaria in Khammouane Province, Lao PDR, in 1999 and 2000. Southeast Asian J Trop Med Pub Hlth. 2002, 33: 532-546.
Vythilingam I, Keokenchanh K, Phommakot S, Nambanya S, Inthakone S: Preliminary studies of Anopheles mosquitos in eight provinces in Lao PDR. Southeast Asian J Trop Med Pub Hlth. 2001, 32: 83-87.
Trung HD, Van Bortel W, Sochantha T, Keokenchanh K, Quang NT, Cong LD, Coosemans M: Malaria transmission and major malaria vectors in different geographical areas of Southeast Asia. Trop Med Int Health. 2004, 9: 230-237. 10.1046/j.1365-3156.2003.01179.x.
Xu JN, Qu FY: Ribosomal DNA difference between species A and D of the Anopheles dirus complex of mosquitoes from China. Med Vet Entomol. 1997, 11: 134-138.
Baimai V, Andre RG, Harrison BA, Kijchalao U, Panthusiri L: Crossing and chromosomal evidence for two additional sibling species within the taxon Anopheles dirus Peyton and Harrison (Diptera: Culicidae) in Thailand. Proc Entomol Soc Wash. 1987, 89: 157-166.
Peyton EL, Harrison BA: Anopheles (Cellia) takasagoensis Morishita 1946, an additional species in the Balabacensis Complex of Southeast Asia (Diptera: Culicidae). Mosq Syst. 1980, 12: 335-347.
Walton C, Handley JM, Collins FH, Baimai V, Harbach RE, Deesin V, Butlin RK: Genetic population structure and introgression in Anopheles dirus mosquitoes in Southeast Asia. Mol Ecol. 2001, 10: 569-580. 10.1046/j.1365-294x.2001.01201.x.
Gingrich JB, Weatherhead A, Sattabongkot J, Pilakasiri C, Wirtz RA: Hyperendemic malaria in a Thai village: dependence of year-round transmission on focal and seasonally circumscribed mosquito (Diptera: Culicidae) habitats. J Med Entomol. 1990, 27: 1016-1026.
Eyles DE, Wharton RH, Cheong WH, Warren M: Studies on malaria and Anopheles balabacensis in Cambodia. Bull World Health Organ. 1964, 30: 7-21.
Paing M, Sebastian A, Tun-Lin W: Anopheline mosquitoes of Myanmar. I. Anopheles (Cellia) dirus Peyton and Harrison, 1979. Myanmar Health Sci Res J. 1989, 1: 122-129.
May K: Karyological identification of two taxa of the Anopheles balabacensis complex from Burma. Genome. 1987, 29: 886-887.
Klein J-M: La faune des moustiques du Cambodge. Cahiers de l'ORSTOM – série Entomologie Médicale et Parasitologie. 1977, XV: 107-122.
Rosenberg R: Forest malaria in Bangladesh. III. Breeding habits of Anopheles dirus. Am J Trop Med Hyg. 1982, 31: 192-201.
Phan VT: Epidémiologie du paludisme et lutte antipaludique au Vietnam. 1998, Editions Médicales Vietnam, Hanoi
Kyi KM: Malaria vectors in Burma 2. Anopheles balabacensis balabacensis Baisas, 1936. Union of Burma J Life Science. 1970, 3: 217-225.
Kitthawee S, Edman JD, Upatham ES: Mosquito larvae and associated macroorganisms occurring in gem pits in southern Tha Mai District, Chanthaburi Province, Thailand. Southeast Asian J Trop Med Pub Hlth. 1993, 24: 143-151.
Wilkinson RN, Gould DJ, Boonyakanist A, Segal HE: Observations on Anopheles balabacensis (Diptera, Culicidae) in Thailand. J Med Entomol. 1978, 14: 666-671.
Khan SA, Handique R, Tewari SC, Dutta P, Narain K, Mahanta J: Larval ecology and mosquito fauna of upper Brahmaputra Valley, Northeast India. Indian J Malariol. 1998, 35: 131-145.
Oo TT, Storch V, Becker N: Studies on the bionomics of Anopheles dirus (Culicidae: Diptera) in Mudon, Mon State, Myanmar. J Vector Ecol. 2002, 27: 44-54.
Aung H, Minn S, Thaung S, Mya MM, Than SM, Hlaing T, Soe S, Druilhe P, Queuche F: Well-breeding Anopheles dirus and their role in malaria transmission in Myanmar. Southeast Asian J Trop Med Pub Hlth. 1999, 30: 447-453.
Tun-Lin W, Htay A, Moe M, Sebastian A, Paing M, Thu MM: Some environmental factors influencing the breeding of Anopheles balabacensis complex (dirus) in domestic wells in Burma. J Commun Dis. 1987, 19: 291-299.
Peyton EL, Harrison BA: Anopheles (Cellia) dirus, a new species of the Leucosphyrus Group from Thailand (Diptera: Culicidae). Mosq Syst. 1979, 11: 40-52.
Kengluecha a, Singhasivanon P, Tiensuwan M, Jones JW, Sithiprasasna R: Water quality and breeding habitats of anopheline mosquito in northwestern Thailand. Southeast Asian J Trop Med Pub Hlth. 2005, 36: 46-53.
Trung HD: Malaria vectors in Southeast Asia: identification, malaria transmission, behaviour and control, phD. 2003, Prince Leopold Institute of Tropical Medicine Antwerp, Department of Parasitology, University of Antwerp, Faculty of Sciences, Department of Biology
Kitthawee S, Edman JD, Upatham ES: Anopheles dirus size and fecundity: relationship to larval density and protein accumulation. Southeast Asian J Trop Med Pub Hlth. 1992, 23: 128-131.
Scanlon JE, Sandhinand U: The distribution and biology of Anopheles balabacensis in Thailand (Diptera: Culicidae). J Med Entomol. 1965, 47: 61-69.
Trung HD, Van Bortel W, Sochantha T, Keokenchanh K, Briët OJT, Coosemans M: Behavioural heterogeneity of Anopheles species in ecologically different localities in Southeast Asia: a challenge for vector control. Trop Med Int Health. 2005, 10: 251-262. 10.1111/j.1365-3156.2004.01378.x.
Nandi J, Misra SP, Rajagopal R, Narasimham MV: Present perspectives of malaria transmission in Boko Area of Assam. J Commun Dis. 1993, 25: 18-26.
Socheath S, Seng CM, Rath TS, Deesin V, Deesin T, Apiwathanasorn C: Study on bionomics of principal malaria vectors in Kratie Province, Cambodia. Southeast Asian J Trop Med Pub Hlth. 2000, 31 (Suppl 1): 106-110.
Dutta P, Bhattacharyya DR, Khan SA, Sharma CK, Mahanta J: Feeding patterns of Anopheles dirus, the major vector of forest malaria in north east India. Southeast Asian J Trop Med Pub Hlth. 1996, 27: 378-381.
Tun-Lin W, Thu MM, Than SM, Mya MM: Hyperendemic malaria in a forested, hilly Myanmar village. J Am Mosq Control Assoc. 1995, 11: 401-407.
Oo TT, Storch V, Becker N: Anopheles dirus and its role in malaria transmission in Myanmar. J Vector Ecol. 2003, 28: 175-183.
Rosenberg R, Maheswary NP: Forest malaria in Bangladesh. II. Transmission by Anopheles dirus. Am J Trop Med Hyg. 1982, 31: 183-191.
Prakash A, Bhattacharyya DR, Mohapatra PK, Mahanta J: Seasonal prevalence of Anopheles dirus and malaria transmission in a forest fringed village of Assam, India. Indian J Malariol. 1997, 34: 117-125.
Somboon P, Aramrattana A, Lines J, Webber R: Entomological and epidemiological investigations of malaria transmission in relation to population movements in forest areas of North West Thailand. Southeast Asian J Trop Med Pub Hlth. 1998, 29: 3-9.
Scanlon JE, Peyton EL, Gould DJ: The Anopheles (cellia) leucosphyrus Dönitz, 1901 group in Thailand. Proc Pap Annu Conf Calif Mosq Control Assoc. 1967, 35: 78-83.
Ismail IA, Notananda V, Schepens J: Studies on malariaand responses of Anopheles balabacensis balabacensis and Anopheles minimus to DDT residual spraying in Thailand. World Health Organization – WHO/MAL/75.847 or WHO/VBC/75.515. 1975
Vythilingam I, Phetsouvanh R, Keokenchanh K, Yengmala V, Vanisaveth V, Phompida S, Hakim SL: The prevalence of Anopheles (Diptera: Culicidae) mosquitoes in Sekong Province, Lao PDR in relation to malaria transmission. Trop Med Int Health. 2003, 8: 525-535. 10.1046/j.1365-3156.2003.01052.x.
Dutta P, Bhattacharyya DR, Sharma CK, Dutta P: Anopheline fauna of parts of Tirap district, Arunachal Pradesh with reference to malaria transmission. Indian J Med Res. 1992, 95: 245-249.
Prakash A, Bhattacharyya DR, Mohapatra PK, Mahanta J: Breeding and day resting habitats of Anopheles dirus in Assam, India. Southeast Asian J Trop Med Pub Hlth. 1997, 28: 610-614.
Elias M, Dewan ZAR: Vectors of malaria in Bangladesh. J Prev Social Med. 1982, 1: 20-28.
Rosenberg R, Andre RG, Somchit L: Highly efficient dry season transmission of malaria in Thailand. Trans R Soc Trop Med Hyg. 1990, 84: 22-28. 10.1016/0035-9203(90)90367-N.
Dutta P, Dev V, Bhattacharyya DR: Anopheline fauna and malaria incidence in Changlang District (Arunachal Pradesh). Indian J Malariol. 1993, 30: 135-143.
Das SC, Baruah I: Incrimination of Anopheles minimus Theobald and Anopheles balabacensis balabacensis Baisa (An. dirus) as malaria vector in Mizoram. Indian J Malariol. 1985, 22: 53-55.
Rattanarithikul R, Konishi E, Linthicum KJ: Observations on nocturnal biting activity and host preference of anophelines collected in Southern Thailand. J Am Mosq Control Assoc. 1996, 12: 52-57.
Rahman WA, Che'Rus A, Ahmad AH: Malaria and Anopheles mosquitos in Malaysia. Southeast Asian J Trop Med Pub Hlth. 1997, 28: 599-605.
Lin CF, He YJ, Tang LH, Ji WQ: Surveillance on Anopheles dirus in malaria hyperendemic areas in Hainan Province (1990–1994). Chinese J Parasitic Dis Control. 2000, 13: 5-7.
Teng T, Chien HL, Hwnag YC, Yeng HS, Yeng HS: Morphological description of Anopheles balabacensis balabacensis Baisas. Acta Entomologica Sinica. 1974, 17: 463-473.
Prakash A, Bhattacharyya DR, Mohapatra PK, Mahanta J: Estimation of vectorial capacity of Anopheles dirus (Diptera: Culicidae) in a forest-fringed village of Assam (India). Vector Borne Zoonotic Dis. 2001, 1: 231-237. 10.1089/153036601753552594.
Ichimori K: Correlation of mosquito size, blood meal size and malarial oocyst production. Jpn J Sanit Zool. 1989, 40: 81-85.
Kitthawee S, Edman JD, Upatham ES: Relationship between female Anopheles dirus (Diptera: Culicidae) body size and parity in a biting population. J Med Entomol. 1992, 29: 921-926.
Paing M, Tun-Lin W, Min S, Myint Z: Malaria situation in a forested foot-hill area of Pegu. Burma Health Sci Res J. 1989, 1: 52-54.
Dutta P, Bhattacharyya DR, Dutta P: Epidemiological observations on malaria in some parts of Tengakhat PHC, Dibrugarh District, Assam. Indian J Malariol. 1991, 28: 121-128.
Marchand RP, Hai NS, Quang NT, Vien NT: Mark-release-recapture studies with Anopheles dirus A in deep forest in central Vietnam to understand its role in highly efficient malaria transmission. 40th annual scientific seminar of Malaysian society of parasitology and tropical medicine (MSPTM) Tropical Diseases and vectors: Management and Control. 2004, 10:
Rosenberg R, Andre RG, Ngampatom S, Hatz C, Burge R: A stable, oligosymptomatic malaria focus in Thailand. Trans R Soc Trop Med Hyg. 1990, 84: 14-21. 10.1016/0035-9203(90)90366-M.
Erhart A, Thang ND, Hung NQ, Toi LV, Hung LX, Tuy TQ, Cong LD, Speybroeck N, Coosemans M, D'alessandro U: Forest malaria in Vietnam: a challenge for control. Am J Trop Med Hyg. 2004, 70: 110-118.
Baimai V, Green CA, Andre RG, Harrison BA, Peyton EL: Cytogenetic studies of some species complexes of Anopheles in Thailand and Southeast Asia. Southeast Asian J Trop Med Pub Hlth. 1984, 15: 536-546.
Dutta P, Bhattacharyya DR, Dutta P: Incrimination of Anopheles dirus as a vector of malaria in Dibrugarh District, Assam. Indian J Malariol. 1989, 26: 149-152.
Wilkinson RN, Gould DJ, Boonyakanist A: Comparative susceptibility of Anopheles balabacensis and An. minimus to naturally occurring Plasmodium falciparum in central Thailand. Proc Helminthol Soc Wash. 1972, 39: 423-427.
Mathenge EM, Misiani GO, Oula DO, Lucy WI, Ndegwa PN, Smith TA: Comparative performance of the Mbita trap, CDC light trap and the human landing catch in the sampling of Anopheles arabiensis, An. funestus and culicine species in a rice irrigation in western Kenya. Malaria J. 2005, 4:
Davis JR, Hall T, Chee EM, Majala A, Minjas J, Shiff CJ: Comparison of sampling anopheline mosquiotes by light-trap and human-bait collections indoors at Bagamoyo, Tanzania. Med Vet Entomol. 1995, 9: 249-255.
Sithiprasasna R, Jaichapor B, Chanaimongkol S, Khongtak P, Lealsirivattanakul T, Tian-Trong S, Burkett DA, Perich MJ, Wirtz RA, Coleman RE: Evaluation of candidates traps as tools for conducting surveillance for Anopheles mosquitoes in a malaria-endemic area in western Thailand. J Med Entomol. 2004, 41: 151-157.
Moore SJ, Zunwei D, Hongning Z, Xuezhong W, Hongbing L, Yujiang X, Hill N: The efficacy of different mosquito trapping methods in a forest-fringe village, Yunnan Province, southern China. Southeast Asian J Trop Med Pub Hlth. 2001, 32: 282-289.
Cheong WH, Warren M, Omar AH, Mahadevan S: Anopheles balabacensis balabacensis identified as vector of simian malaria in Malaysia. Science. 1965, 150: 1314-1315. 10.1126/science.150.3701.1314.
Suwonkerd W, Ung B, Rimwangtrakul K, Wongkattiyakul S, Kattiyamongkool B, Chitprarop U, Takagi M: A field study on the response of Anopheles dirus to DDT and Fenitrothion sprayed to huts on Petchabun Province, Thailand. Trop Med. 1990, 32: 1-5.
Uza M, Phompida S, Toma T, Takakura M, Manivong K, Bounyadeth S, Kobayashi J, Koja Y, ozasa Y, Miyagi I: Knowledge and behavior relating to malaria in malaria endemic villages of Khammouane Province, Lao PDR. Southeast Asian J Trop Med Pub Hlth. 2002, 33: 246-254.
Walton C, Handley JM, Collins FH, Harbach RE, Baimai V, Butlin RK: Population structure and population history of Anopheles dirus mosquitoes in Southeast Asia. Mol Biol Evol. 2000, 17: 962-974.
Kulkarni SM, Naik PS: Breeding habitats of mosquitoes in Goa. Indian J Malariol. 1989, 26: 41-44.
Oo TT, Storch V, Becker N: Review of the anopheline mosquitoes of Myanmar. J Vector Ecol. 2004, 29: 21-40.
Rosenberg R, Maheswary NP: Forest malaria in Bangladesh. I. Parasitology. Am J Trop Med Hyg. 1982, 31: 175-182.
Baimai V, Harbach RE, Kijchalao U: Cytogenetic evidence for of fifth species within the taxon Anopheles dirus in Thailand. J Am Mosq Control Assoc. 1988, 4: 333-338.
Misra SP, Nandi J, Narasimham MV, Rajagopal R: Malaria transmission in Nagaland, India. Part I: Anophelines and their seasonality. J Commun Dis. 1993, 25: 62-66.
Prakash A, Bhattacharyya DR, Mohapatra PK, Mahanta HC: Indoor biting behaviour of Anopheles dirus Peyton and Harrison, 1979 in upper Assam, India. Mosq Borne Dis Bull. 1997, 4: 31-37.
Dutta P, Bhattacharyya DR, Sharma CK, Dutta P: The importance of Anopheles dirus (An. balabacensis) as a vector of malaria in northeast India. Indian J Malariol. 1989, 26: 95-101.
Yu Y, Denghong C: Revision and description of Anopheles (Cellia) balabacensis Baisas from Yunnan, China. Entomotaxonomia. 1984, 6: 323-324.
Thu MM, Tun-Lin W, Sebastian A, Kanda T, May K: Preliminary biological study on two taxa of the Anopheles balabacensis complex in Burma. Jpn J Genet. 1985, 60: 313-380.
Rattanarithikul R, Linthicum KJ, Konishi E: Seasonal abundance and parity rates of anopheles species in southern Thailand. J Am Mosq Control Assoc. 1996, 12: 75-83.
Rattanarithikul R, Konishi E, Linthicum KJ: Detection of Plasmodium vivax and Plasmodium falciparum circumsporozoite antigen in anopheline mosquitoes collected in southern Thailand. Am J Trop Med Hyg. 1996, 54: 114-121.
Tun-Lin W, Moe M, Htay-Aung , Paing M: Larval density estimation of well-breeding anophelines by mark-release-recapture technique in Burma. Mosq Borne Dis Bull. 1986, 1-4.
Kobayashi J, Somboon P, Keomanila H, Inthavongsa S, Nambanya S, Inthakone S, Sato Y, Miyagi I: Malaria prevalence and a brief entomological survey in a village surrounded by rice fields in Khammouan Province, Lao PDR. Trop Med Int Health. 2000, 5: 17-21. 10.1046/j.1365-3156.2000.00516.x.
Kobayashi J, Nambanya S, Miyagi I, Vanachone B, Manivong K, Koubouchan T, Amano H, Nozaki H, Inthakone S, Sato Y: Collection of anopheline mosquitos in three villages endemic for malaria in Khammouane, Lao PDR. Southeast Asian J Trop Med Pub Hlth. 1997, 28: 615-620.
Gould DJ, Esah S, Pranith U: Relation of Anopheles aconitus to malaria in the central plain of Thailand. Trans R Soc Trop Med Hyg. 1967, 61: 441-442. 10.1016/0035-9203(67)90024-7.
Baker EZ, Beier JC, Meek SR, Wirtz RA: Detection and quantification of Plasmodium falciparum and P. vivax infections in Thai-Kampuchean Anopheles (Diptera:culicidae) by enzyme-linked immunosorbent assay. J Med Entomol. 1987, 24: 536-541.
Rosenberg R, Andre RG, Ketrangsee S: Seasonal fluctuation of Plasmodium falciparum gametocytaemia. Trans R Soc Trop Med Hyg. 1990, 84: 29-33. 10.1016/0035-9203(90)90369-P.
Harbach RE, Gingrich JB, Pang LW: Some entomological observations on malaria transmission in a remote village in northwestern Thailand. J Am Mosq Control Assoc. 1987, 3: 296-301.
Marchand RP, Quang NT, Hoanh NQ, Vien NT: The Khanh PhuMalaria Research Project. Review meeting on two year baseline studies in an area of intense malaria transmission, Central Vietnam. Medisch Comité Nederland – Vietnam and Institute of Malariology Parasitology and Entomology. 1997, Medisch Comité Nederland – Vietnam and Institute of Malariology Parasitology and Entomology, Medical Publishing House, Hanoi, 1-69.
Somboon P, Lines J, Aramrattana A, Chitprarop U, Prajakwong S, Khamboonruang C: Entomological evaluation of community-wide use of lambdacyhalothrinimpregnated bed nets against malaria in a border area of north-west Thailand. Trans R Soc Trop Med Hyg. 1995, 89: 248-254. 10.1016/0035-9203(95)90525-1.
Suwonkerd W, Tsuda Y, Overgaard HJ, Chawprom S, Tuno N, Prajakwong S, Takagi M: Change in malaria vector densities over a twenty-three year period in Mae Hong Son Province northern Thailand. Southeast Asian J Trop Med Pub Hlth. 2004, 35: 316-324.
Overgaard HJ, Ekbom B, Suwonkerd W, Takagi M: Effect of landscape structure on anopheline mosquito density and diversity in northern Thailand: Implications for malaria transmission and control. Landscape Ecol. 2003, 18: 605-619. 10.1023/A:1026074910038.
Suwonkerd W, Takagi M, Ung BA, Prajakwong S: Seasonal and spatial distribution of three malaria vectors at three mountainous villages in North Thailand. Jpn J Trop Med Hyg. 1995, 23: 183-187.
Sidavong B, Vythilingam I, Phetsouvanh R, Chan ST, Phonemicay T, Hakim SL, Phompida S: Malaria transmission by Anopheles dirus in Attapeu Province, Lao PDR. Southeast Asian J Trop Med Pub Hlth. 2004, 35: 309-315.
Tang LH: Study on malaria epidemic potential in Sanya developing area, Hainan Province. Chinese J Parasitic Dis Control. 1991, 4: 163-164.
Tang LH, Qian HL, Xu SH: Malaria and its control in the People's Republic of China. Southeast Asian J Trop Med Pub Hlth. 1991, 22: 467-476.
Acknowledgements
The authors of the manuscript are grateful to the members of the MALVECASIA network which provided fresh information on the distribution of the Anopheles dirus complex in the Southeast Asian region, including Dr Trung and Dr Thuan from the National Institute of Malariology, Parasitology and Entomology of Vietnam, Dr Tho and Dr Socheat from the National Centre for Malaria Control, Parasitology and Entomology of Cambodia, Dr Kalounna, Dr Bouasy and Dr Samlane from the Centre of Malariology, Parasitology and Entomology of Laos and Dr Visut Baimai from the Mahidol University of Thailand. Thanks to Ralph Harbach and Yvonne Linton from the Natural History Museum of London, who supervised the quality of mosquito identification in the network. Thanks to Wim van Bortel from the Institute for Tropical Medicine of Belgium for gathering and supervising the MALVECASIA collection site database, and Katrijn Verhaegen for sharing the identification of sibling species in Cambodia and Vietnam. The authors of the manuscript are also grateful to Dr Diwakar Dinesh for recent information on the distribution of An. dirus s.l. in India. Many thanks to the European Union for financing the MALVECASIA network through an INCO-DEV project (ERBIC18.CT.970211) and the MALVECASIA project (IC4-CT-2002-10041).
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors' contributions
VO analysed the more than a hundred publications, designed the database of collection sites and the maps, and drafted the manuscript. PD contributed to the interpretation, presentation of the results and critically reviewed the manuscript. MC contributed his expertise in malaria vector control in Southeast Asia as well as the MALVECASIA database of more than a hundred newly investigated sites in Southeast Asia. He critically reviewed the manuscript. All authors read and approved the final manuscript.
Electronic supplementary material
12936_2006_331_MOESM1_ESM.pdf
Additional file 1: Map Key. Correspondence between collection sites number and reviewed publications: sibling species and identification methods. (PDF 47 KB)
12936_2006_331_MOESM2_ESM.xls
Additional file 2: Collection sites description. The table lists 537 sites with geographical coordinates used for the display on the maps and true coordinates as well as details on mosquito collections and site description (XLS 253 KB)
12936_2006_331_MOESM3_ESM.xls
Additional file 3: Collection sites listed by reviewed publications. The table lists 130 reviewed publications with associated collection sites, species identified and identification method. (XLS 36 KB)
12936_2006_331_MOESM4_ESM.xls
Additional file 4: Abbreviations and descriptions used in the other additional files. lists information and description for each column and each table as well as abbreviations included in the file. (XLS 19 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under license to BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Obsomer, V., Defourny, P. & Coosemans, M. The Anopheles dirus complex: spatial distribution and environmental drivers. Malar J 6, 26 (2007). https://doi.org/10.1186/1475-2875-6-26
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1475-2875-6-26