Abstract
Background
Public health strategies are needed to curb antimalarial drug resistance. Theoretical argument points to an association between malaria transmission and drug resistance although field evidence remains limited. Field observations, made in Zimbabwe, on the relationship between transmission and multigenic drug resistance, typified by chloroquine, are reported here.
Methods
Periodic assessments of the therapeutic response of uncomplicated falciparum malaria to chloroquine in two selectively sprayed or unsprayed health centre catchments, from 1995 – 2003. Cross-sectional analysis of in vivo chloroquine failure events for five sites in relation to natural endemicity and spraying history.
Results
During selective house spraying, the chloroquine failure rate for the sprayed catchment decreased, such that, after four years, the odds of chloroquine failure were 4× lower than before start of spraying in the area (OR 0.2, 95% CI 0.07 – 0.75, p = 0.010, n = 100). Chloroquine failure odds for the sprayed area became 4× lower than contemporaneous failure odds for the unsprayed area (OR 0.2 95% CI 0.08 – 0.65, p = 0.003, n = 156), although the likelihood of failure was not significantly different for the two catchments before selective spraying started (OR 0.5, 95% CI 0.21 – 1.32; p = 0.170, n = 88). When spraying ended, in 1999, the drug failure odds for the former sprayed area increased back 4 fold by 2003 (OR 4.2, 95%CI 1.49 – 11.78, p = 0.004, n = 146). High altitude areas with naturally lower transmission exhibited a 6× lower likelihood of drug failure than low-lying areas (OR 0.16 95% CI 0.068 – 0.353, -2 log likelihood change 23.239, p < 0.001, n = 465). Compared to sites under ongoing annual spraying, areas that were last sprayed 3–7 years ago experienced a 4-fold higher probability of chloroquine failure (OR 4.1, 95%CI 1.84 – 9.14, -2 log likelihood change 13.956, p < 0.001).
Conclusion
Reduced transmission is associated with suppressed levels of resistance to chloroquine and presumably other regimens with multigenic drug resistance. It seems the adoption of transmission control alongside combination chemotherapy is a potent strategy for the future containment of drug-resistant malaria.
Similar content being viewed by others
Background
The escalation of parasite drug resistance has persisted as a major obstacle to malaria control for decades [1–3]. Owing to dwindling options for affordable, safe and effective drugs, rising clinical failure rates exact a substantial public health toll, especially in Africa [4, 5]. In countries that recently replaced chloroquine with sulfadoxine/pyrimethamine as first line treatment, there are signs of increasing resistance to the antifolate combination [1, 2, 6–9]. Partly because of the spectre of drug resistance, pharmaceutical companies reduced investment in new antimalarial drug research. Fortunately, official calls in the mid 1990's led to renewed public-private sector initiatives for the development of new compounds, as well as the improvement of existing ones [10, 11].
However, Plasmodium falciparum has repeatedly demonstrated the ability to develop resistance to practically any drug upon wider introduction, as illustrated by multi-drug resistance, especially in South East Asia [12–15]. Thus, public health strategies that delay or minimize the escalation of drug resistance are urgently required. To date, the only approach that has been widely evaluated and is currently being introduced is the use of combination chemotherapy [11, 16, 17] which protects constituent drugs from resistance through a multigenic mechanism of resistance and strategic pharmacological properties such as short half-life. In poor countries the effectiveness of this method is hampered by increased cost of medication. Furthermore, even the new combinations are not totally protected from the development of resistance, as illustrated by the recent confirmations of clinical failure and in vitro resistance to proguanil/atovaquone [18–22]. Additional strategies are, therefore, needed to ensure the successful containment of drug-resistant malaria.
Mathematical models have been proposed suggesting a relationship between malaria transmission and the evolution of drug resistance, though some workers suggest a positive association [23, 24] while others propose a negative one [25, 26]. Major implications for control pertain to this question. It may mean that vector control programmes are counterproductive by aggravating drug resistance, or, it could be that they complement chemotherapy by alleviating resistance. Although this interaction between transmission and drug resistance is further addressed in a review [27], the exact answer still remains uncertain against a background of limited field evidence. The present paper presents observations on the field relationship between transmission variations (both natural and vector control induced) and the levels of in vivo multigenic drug resistance, typified by chloroquine.
Methods
Study areas and population
Zimbabwe, on the southern fringes of malaria in Africa, experiences seasonal and potentially epidemic transmission characterized by a non-immune population with high probability of drug treatment [28–30]. The country has sustained a national malaria vector control programme for decades, based on intradomicilliary application of residual insecticide. From the early 1990s selective vector control was introduced, in which areas with moderate transmission are of less priority and spraying is focused in zones of high transmission/high malarial incidence. Chloroquine has remained the first line treatment for uncomplicated malaria, although a combination of chloroquine and sulfadoxine/pyrimethmanine is currently being introduced in some areas. A tiered drug distribution policy has been implemented in the country, so that, until 1997, chloroquine was the only antimalarial available at the peripheral level. Thereafter, policy revisions allowed wider distribution of sulfadoxine/pyrimethamine to treat chloroquine failure cases.
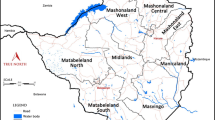
The study was based at five health centres located in the low-lying (<600 m above sea level) hyperendemic transmission zone as well as those in the higher altitude (600 – 1200 m asl) mesoendemic transmission zone bordering the malaria-free central watershed (Table 1, Fig 1). All the study locations experience seasonal, single peak (February-May) malaria transmission typical for Zimbabwe. Acute symptoms and complications occur across all ages in this non-immune population, where asymptomatic carriage of asexual parasitaemia is rare [28, 29]. The study was conducted on uncomplicated falciparum malaria cases of all age groups presenting at the health centres for treatment.
Study design
The study was a prospective assessment of the therapeutic response of P. falciparum malaria to chloroquine from 1995–2003. Consecutive assessments of therapeutic response were conducted in two mesoendemic sites during the presence or absence of selective indoor residual insecticide spraying (house spraying). Transverse assays for Pfmdr1 and Pfcrt mutations associated with chloroquine resistance were carried out in these two sites during the 1998–99 transmission season. Further assessments of in vivo chloroquine therapeutic response were carried out cross-sectionally in another three sites where treatment change to chloroquine (CQ) + sulfadoxine/pyrimethamine (SP) was not yet being implemented due to temporary unavailability of SP. Malarial incidence was determined retrospectively for all sites using available health centre records.
In vivo antimalarial therapeutic efficacy assessments
The in vivo therapeutic efficacy of chloroquine was assessed using the standard WHO (1996) protocol [31]. Since this protocol was primarily targeted for regions of intense malaria transmission, two modifications were adopted to suit the seasonal/epidemic conditions of Zimbabwe. These were (i) inclusion of febrile patients of all age groups and (ii) adoption of radical asexual parasite elimination as a criterion for adequate response to treatment.
Inclusion of all age groups was on the rationale that there is no premunition in the population. Recruited patients were thus a representative sample of the symptomatic population which presents for treatment with chloroquine in the primary health care system. The radical asexual parasite elimination criterion was adopted because persistent asexual parasitaemia poses a risk of complications in non-immunes.
Molecular detection of Pfmdr1 and Pfcrt polymorphisms
Amino acid polymorphisms at codons 86 and 1246 of the P. falciparum Pfmdr 1 gene and at codon 76 of the P. falciparum chloroquine resistance transporter gene (Pfcrt), which are associated with chloroquine resistance [32, 33], were detected by PCR and codon-specific restriction enzyme digestion [34, 35]. Appropriate positive and negative control strains were used in interpretation and, except for the Pfcrt codon, additional restriction sites were included in the target PCR product to serve as internal controls for complete digestion.
Ethics
The study was approved by respective provincial medical health authorities and by the Medical Research Council of Zimbabwe. Patient participation was by the informed consent of the patients themselves or guardians, in the case of children.
Results
Association of house-spraying with reduced levels of chloroquine resistance
(i) Burma Valley and Sahumani follow-up study
On the grounds of low malarial incidence, the catchments of Sahumani clinic, in Mutasa district and Burma Valley clinic, in Mutare district (Fig 1), were removed from the spraying programme in 1992, with the advent of selective control to save on inseciticide. However, the Burma catchment, which is situated on commercial farms, was re-allocated to annual spraying from 1995 – 1999 when, for economic reasons, local farmers agreed to supply the malaria control authorities with insecticide. The Burma catchment subsequently reverted to no spraying after the 1999 spraying operation, due to disagreements between commercial farmers and the government. In contrast, the Sahumani catchment, which is located in villages, remained unsprayed from 1992.
There were no malaria statistics for the two health centres prior to 1998, (1997 for Sahumani). However, during the selective annual spraying, the risk of contracting malaria in the sprayed Burma Valley catchment was at least 2.6 fold lower than for Sahumani (Fig 2) from 1998 – 2000. After the selective spraying operation ended in 1999, the malarial incidence became uniform for the two catchments by 2001 (Fig 2).
The therapeutic failure rate of Burma Valley decreased during selective spraying (Table 2) such that by 1999, the odds of chloroquine failure were 4× lower than they were before spraying resumed in this area (OR (95% CI) 0.233 (0.072 – 0.747), for 1999 compared to 1995; 0.482 (0.270 – 0.861), for each year of spraying: -2 log likelihood ratio change 6.432, df = 1, p = 0.011). Therapeutic failure rates were not significantly different in the Burma Valley and Sahumani catchments (1995 season) prior to selective spraying of Burma Valley (Table 2). However, by 1999 the odds of drug failure had become 4× lower in the annually sprayed Burma catchment (Table 2). The failure rate in Sahumani did not significantly change during the 4-year period (OR (95%CI): 0.62 (0.25 – 1.57), for 1998 compared to 1995; 0.53 (0.26 – 1.08) for 1999 compared to 1995; 0.85 (0.71 – 1.02) for each successive year, -2 × log likelihood ratio change, 3.080, df, 1, p = 0.0793).
After selective spraying ceased in 1999, the odds of drug failure in Burma valley increased back 4-fold by 2003 (OR (95%): 4.18 (1.485 – 11.782), p = 0.004, n = 146) Chloroquine efficacy assessments for 2003 were not conducted in Sahumani as the treatment was changed that year to chloroquine plus sulfadoxine/pyrimethamine.
(ii) In vivo prevalence of mutations in Pfmdr1 and Pfcrt genes
Amino acid polymorphisms on Pfmdr 1 and Pfcrt codons associated with chloroquine resistance were examined in pre-treatment patient samples from 1998 and 1999 in Burma Valley and Sahumani (i.e. 3–4 years after re-start of spraying in Burma Valley). Resistance-associated mutations at amino acid codons 86 and 1246 of Pfmdr 1, and codon 76 of Pfcrt, were more prevalent in the Sahumani area (Table 3, Fig 3). Interestingly, mixed infections containing both mutant and wild type variants tended to be more frequent in the Burma Valley area (Fig 3), despite lower transmission in this catchment. The same distribution pattern observed with individual codons was mirrored in mutated haplotypes comprising two or more amino acid codons (Table 3). Three-codon haplotypes from the sprayed area exhibited significantly more mixed mutant and wild variants at one or more codons than corresponding haplotypes from the unsprayed area (odds ratio 5.4, 95 % CI: 1.89 – 15.54, p = 0.001, n = 131).
Drug failure as a function of transmission
A scatterplot of chloroquine therapeutic failure rate with malarial incidence suggested a positive association (Fig 4). In stead of using parametric tests on arcsine transformed data (perhaps better done with more data points), the probability of chloroquine failure was examined as a function of transmission zone, and spraying history, using a logistic model. The health centre catchments naturally falling in the mesoendemic zone according to altitudinal classifications [28], did exhibit significantly lower malarial incidence, for at least the previous 10 years, than those in the hyperendemic zone (Fig 5, Table 4). In the logistic regression, the probability of therapeutic failure was 6.4-fold lower in these mesoendemic catchments than in hyperendemic ones (Table 5, 6, 7). At any time point, catchments that were under ongoing annual spraying experienced 4-fold lower likelihood of drug failure than those that were last sprayed 3–7 years ago (Table 5).
Discussion
The build up of drug-resistant P. falciparum malaria calls for public health strategies to maximize the useful life of antimalarials. According to the findings of the present study, reduced transmission, due to vector control or high altitude, was associated with suppressed levels of in vivo therapeutic failure and genotypic resistance to chloroquine. Assuming that chloroquine resistance has a multigenic mechanism, as is the general consensus [12, 36, 37], this association between transmission and drug resistance presumably governs other drugs or drug combinations that have polygenically encoded resistance.
From the Burma Valley and Sahumani cross-sectional assays, there was, in the sprayed catchment, a higher likelihood of infections carrying mixed mutated and wild type codons, for both Pfcrt and Pfmdr1, despite the lower transmission. This paradoxical result suggests that the sprayed area probably favoured more genetic out-crossing, resulting in recombination break down of drug-resistant haplotypes. The genetic out-crossing may partly explain the association of low drug resistance with the house spraying. Further studies are needed to verify this relationship in more areas.
In Burma Valley, despite drug pressure, the proportion of resistant parasites decreased during spraying, and subsequently resurged after the spraying stopped. This is reminiscent of the decrease in proportion of chloroquine-resistant parasites reported in China [38], and more recently in Malawi [39, 40], following suspension of chloroquine use. From these observations it would seem that chloroquine-resistant parasites bear a fitness cost as drug selection advantage is removed or counteracted.
What is distinct about the current results is that the fitness cost for resistance appeared to occur in the sporogonic phase, as distinguished from an in vivo fitness burden that is thought to ensue following cessation of drug use. In the present results, drug selection advantage for the resistant parasites appeared to be directly counteracted by independent survival limiting factors, such as vector control and high altitude. This has important implications for control as it means that drug-resistant P. falciparum can be contained during drug use. Furthermore, costly acquisition of immunity in the resident population is, presumably, not the only prerequisite for curbing drug resistance.
The present results afford field evidence supporting the continuation of sustainable malaria vector control programmes. Similar findings were reported for Uganda [41], although the same workers found a difference between chloroquine (multigenic resistance) and sulfadoxine/pyrimethamine (monogenic resistance) below a critical threshold of transmission [37]. These papers may further support the findings of the present study. It has been cautioned that resistance might exacerbate as eradication is approached [42]. However, in the current study, the low transmission levels associated with high altitude and spraying showed no signs of this counterproductive effect. Moreover, in poor countries, which are the de facto stronghold for malaria, eradication so far remains only an academic prospect, as the malaria burden continues to increase [43]. It seems that the adoption of sustainable transmission control with combination chemotherapy is a potent approach for the future containment of drug-resistant malaria.
Conclusions
Reduced transmission due to house spraying or high altitude is associated with suppressed levels of phenotypic and genotypic resistance to chloroquine and presumably other multigenically encoded drug regimens. Transmission control implemented with combination chemotherapy seems a potent approach for the future containment of drug-resistant malaria.
References
Wernsdorfer WH, Payne D: The dynamics of drug resistance in Plasmodium falciparum. Pharmacol Ther. 1991, 50: 95-121. 10.1016/0163-7258(91)90074-V.
Wernsdorfer WH: Epidemiology of drug resistance in malaria. Acta Trop. 1994, 56: 143-156. 10.1016/0001-706X(94)90060-4.
D'Alessandro U, Buttiens H: History and importance of antimalarial drug resistance. Trop Med Int Health. 2001, 6: 845-848. 10.1046/j.1365-3156.2001.00819.x.
Trape JF: The public health impact of chloroquine resistance in Africa. Am J Trop Med Hyg. 2001, 64: 12-17.
Trape JF, Pison G, Spiegel A, Enel C, Rogier C: Combating malaria in Africa. Trends Parasitol. 2002, 18: 224-230. 10.1016/S1471-4922(02)02249-3.
Gorissen E, Ashruf G, Lamboo M, Bennebroek J, Gikunda S, Mbaruku G, Kager PA: In vivo efficacy study of amodiaquine and sulfadoxine/ pyrimethamine in Kibwezi, Kenya and Kigoma, Tanzania. Trop Med Int Health. 2000, 5: 459-463. 10.1046/j.1365-3156.2000.00570.x.
Bijl HM, Kager J, Koetsier DW, van der Werf TS: Chloroquine- and sulfadoxine-pyrimethamine-resistant Falciparum malaria in vivo - a pilot study in rural Zambia. Trop Med Int Health. 2000, 5: 692-695. 10.1046/j.1365-3156.2000.00629.x.
Bwijo B, Kaneko A, Takechi M, Zungu IL, Moriyama Y, Lum JK, Tsukahara T, Mita T, Takahashi N, Bergqvist Y, Bjorkman A, Kobayakawa T: High prevalence of quintuple mutant dhps/dhfr genes in Plasmodium falciparum infections seven years after introduction of sulfadoxine and pyrimethamine as first line treatment in Malawi. Acta Trop. 2003, 85: 363-373. 10.1016/S0001-706X(02)00264-4.
White N: Sulfadoxine-pyrimethamine for uncomplicated falciparum malaria: sulfadoxine-pyrimethamine is not working in Malawi. Bmj. 2004, 328: 1259; author reply 1260-
Olliaro PL, Trigg PI: Status of antimalarial drugs under development. Bull World Health Organ. 1995, 73: 565-571.
Olliaro PL, Taylor WR: Developing artemisinin based drug combinations for the treatment of drug resistant falciparum malaria: A review. J Postgrad Med. 2004, 50: 40-44.
Foote SJ, Cowman AF: The mode of action and the mechanism of resistance to antimalarial drugs. Acta Trop. 1994, 56: 157-171. 10.1016/0001-706X(94)90061-2.
Dua VK, Dev V, Phookan S, Gupta NC, Sharma VP, Subbarao SK: Multi-drug resistant Plasmodium falciparum malaria in Assam, India: timing of recurrence and anti-malarial drug concentrations in whole blood. Am J Trop Med Hyg. 2003, 69: 555-557.
Giao PT, de Vries PJ, Hung le Q, Binh TQ, Nam NV, Kager PA: CV8, a new combination of dihydroartemisinin, piperaquine, trimethoprim and primaquine, compared with atovaquone-proguanil against falciparum malaria in Vietnam. Trop Med Int Health. 2004, 9: 209-216. 10.1046/j.1365-3156.2003.01180.x.
Wattanakoon Y, Chittamas S, Pornkulprasit V, Kanda T, Thimasarn K, Rojanawatsirivej C, Looareesuwan S, Bunnag D: Six-years monitoring the efficacy of the combination of artesunate and mefloquine for the treatment of uncomplicated falciparum malaria. Southeast Asian J Trop Med Public Health. 2003, 34: 542-545.
Peters W: The prevention of antimalarial drug resistance. Pharmacol Ther. 1990, 47: 499-508. 10.1016/0163-7258(90)90067-C.
White N: Antimalarial drug resistance and combination chemotherapy. Philos Trans R Soc Lond B Biol Sci. 1999, 354: 739-749. 10.1098/rstb.1999.0426.
David KP, Alifrangis M, Salanti A, Vestergaard LS, Ronn A, Bygbjerg IB: Atovaquone/proguanil resistance in Africa: a case report. Scand J Infect Dis. 2003, 35: 897-898.
Schwartz E, Bujanover S, Kain KC: Genetic confirmation of atovaquone-proguanil-resistant Plasmodium falciparum malaria acquired by a nonimmune traveler to East Africa. Clin Infect Dis. 2003, 37: 450-451. 10.1086/375599.
Farnert A, Lindberg J, Gil P, Swedberg G, Berqvist Y, Thapar MM, Lindegardh N, Berezcky S, Bjorkman A: Evidence of Plasmodium falciparum malaria resistant to atovaquone and proguanil hydrochloride: case reports. Bmj. 2003, 326: 628-629. 10.1136/bmj.326.7390.628.
Fivelman QL, Butcher GA, Adagu IS, Warhurst DC, Pasvol G: Malarone treatment failure and in vitro confirmation of resistance of Plasmodium falciparum isolate from Lagos, Nigeria. Malar J. 2002, 1: 1-10.1186/1475-2875-1-1.
Crabb C: Plasmodium falciparum outwits Malarone, protector of travellers. Bull World Health Organ. 2003, 81: 382-383.
Hastings IM: A model for the origins and spread of drug-resistant malaria. Parasitology. 1997, 115 ( Pt 2): 133-141. 10.1017/S0031182097001261.
Hastings IM, Mackinnon MJ: The emergence of drug-resistant malaria. Parasitology. 1998, 117 ( Pt 5): 411-417. 10.1017/S0031182098003291.
Ariey F, Robert V: The puzzling links between malaria transmission and drug resistance. Trends Parasitol. 2003, 19: 158-60; author reply 160-1. 10.1016/S1471-4922(03)00054-0.
Schmidt KF: Malaria research. Inbred parasites may spur resistance. Science. 1995, 269: 1670-
Molyneux DH, Floyd K, Barnish G, Fevre EM: Transmission control and drug resistance in malaria: a crucial interaction. Parasitol Today. 1999, 15: 238-240. 10.1016/S0169-4758(99)01453-2.
Taylor P, Mutambu SL: A review of the malaria situation in Zimbabwe with special reference to the period 1972-1981. Trans R Soc Trop Med Hyg. 1986, 80: 12-19. 10.1016/0035-9203(86)90185-9.
Herd N, Jordan T: An investigation of malaria during pregnancy in Zimbabwe. Cent Afr J Med. 1981, 27: 62-68.
Taylor P, A. Taputaira: Forty years of malaria control in Zimbabwe. Proceedings of the First National Symposium of Science and Technology. 1988, Harare
WHO: Assessment of the Therapeutic Efficacy of Antimalarial Drugs for the Treatment of Uncomplicated Malaria in Africa in Areas with Intense Transmission. 1996, Geneva, World Health Organization
Foote SJ, Kyle DE, Martin RK, Oduola AM, Forsyth K, Kemp DJ, Cowman AF: Several alleles of the multidrug-resistance gene are closely linked to chloroquine resistance in Plasmodium falciparum. Nature. 1990, 345: 255-258. 10.1038/345255a0.
Fidock DA, Nomura T, Talley AK, Cooper RA, Dzekunov SM, Ferdig MT, Ursos LMB, Naude B, Deitsch KW, Su XZ, Wootton JC, Roepe PD, Wellems TE: Mutations in the Plasmodium falciparum digestive vacuole transmembrane protein Pfcrtand evidence for their role in chloroquine resistance. Molecular Cell. 2000, 6: 861-871.
Frean JA, el Kariem FM, Warhurst DC, Miles MA: Rapid detection of pfmdr1 mutations in chloroquine-resistant Plasmodium falciparum malaria by polymerase chain reaction analysis of blood spots. Trans R Soc Trop Med Hyg. 1992, 86: 29-30. 10.1016/0035-9203(92)90426-D.
Duraisingh MT, Drakeley CJ, Muller O, Bailey R, Snounou G, Targett GA, Greenwood BM, Warhurst DC: Evidence for selection for the tyrosine-86 allele of the pfmdr 1 gene of Plasmodium falciparum by chloroquine and amodiaquine. Parasitology. 1997, 114 ( Pt 3): 205-211. 10.1017/S0031182096008487.
Dye C, Williams BG: Multigenic drug resistance among inbred malaria parasites. Proc R Soc Lond B Biol Sci. 1997, 264: 61-67. 10.1098/rspb.1997.0009.
Talisuna AO, Langi P, Mutabingwa TK, Van Marck E, Speybroeck N, Egwang TG, Watkins WW, Hastings IM, D'Alessandro U: Intensity of transmission and spread of gene mutations linked to chloroquine and sulphadoxine-pyrimethamine resistance in falciparum malaria. Int J Parasitol. 2003, 33: 1051-1058. 10.1016/S0020-7519(03)00156-5.
Liu DQ, Liu RJ, Ren DX, Gao DQ, Zhang CY, Qui CP, Cai XZ, Ling CF, Song AH, Tang X: Changes in the resistance of Plasmodium falciparum to chloroquine in Hainan, China. Bull World Health Organ. 1995, 73: 483-486.
Mita T, Kaneko A, Lum JK, Bwijo B, Takechi M, Zungu IL, Tsukahara T, Tanabe K, Kobayakawa T, Bjorkman A: Recovery of chloroquine sensitivity and low prevalence of the Plasmodium falciparum chloroquine resistance transporter gene mutation K76T following the discontinuance of chloroquine use in Malawi. Am J Trop Med Hyg. 2003, 68: 413-415.
Kublin JG, Cortese JF, Njunju EM, Mukadam RA, Wirima JJ, Kazembe PN, Djimde AA, Kouriba B, Taylor TE, Plowe CV: Reemergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine use in Malawi. J Infect Dis. 2003, 187: 1870-1875. 10.1086/375419.
Talisuna AO, Langi P, Bakyaita N, Egwang T, Mutabingwa TK, Watkins W, Van Marck E, D'Alessandro U: Intensity of malaria transmission, antimalarial-drug use and resistance in Uganda: what is the relationship between these three factors?. Trans R Soc Trop Med Hyg. 2002, 96: 310-317. 10.1016/S0035-9203(02)90108-2.
Hastings IM: Malaria control and the evolution of drug resistance: an intriguing link. Trends Parasitol. 2003, 19: 70-73. 10.1016/S1471-4922(02)00017-X.
Yamey G: Roll Back Malaria: a failing global health campaign. Bmj. 2004, 328: 1086-1087. 10.1136/bmj.328.7448.1086.
Acknowledgements
Mr V. Rusinga (Manicaland Provincial Medical Office), provided vital information on history of provincial selective vector control strategies. Dr. B. Mabaera commented on the manuscript. The authors are thankful to medical staff of Sahumani, Burma Valley, Chitakatira, Madhuku and Mola clinics, for their unwavering support to the study. Acknowledgement is also due to the residents of the study areas for participating in follow-up intensive drug efficacy assessments and sample collections. Mr A.C. Murahwa and J. Karisa carried out much of the microscopy for the study and played a central role in field data collection. Dr. Tim Anderson gave invaluable advice on molecular aspects of the study and edited the manuscript. Control strains were kindly donated by Dr. M.T. Duraisingh in Professor D.C. Warhurst's group and Dr. Lisa Ranford-Cartwright from Professor David Walliker's labs. This study was sponsored by WHO/TDR and the EU INCO-DC programme. The Beit Trust contributed to funding of field data collection and travel. SM was a Beit Fellow. Molecular work and part of travel expenses were met by The Wellcome Trust.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors' contributions
SM was the principal investigator responsible for the study design, data collection, analysis and drafting of the manuscript. SLM carried out essential co-ordination of project activities. RM and TC afforded technical input on the manuscript and facilitated field data collection. SKC made vital contributions in original proposal development, seeking of funding, and edited the manuscript. KPD provided crucial inputs in the study design, genetic analysis, general direction and co-ordination of the study and the writing up of the manuscript.
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Mharakurwa, S., Mutambu, S.L., Mudyiradima, R. et al. Association of house spraying with suppressed levels of drug resistance in Zimbabwe. Malar J 3, 35 (2004). https://doi.org/10.1186/1475-2875-3-35
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1475-2875-3-35