Abstract
Background
The tcdA-negative variant (A-B+) of Clostridium difficile is prevalent in East Asian countries. However, the risk factors and clinical characteristics of A-B+ C. difficile infections (CDI) are not clearly documented. The objective of this study was to investigate these characteristics.
Methods
From September 2008 through January 2010, the clinical characteristics, medication history and treatment outcomes of CDI patients were recorded prospectively. Toxin characterization and antibiotic susceptibility tests were performed on stool isolates of C. difficile.
Results
During the study period, we identified 22 cases of CDI caused by tcdA-negative tcdB-positive (A-B+) strains and 105 cases caused by tcdA-positive tcdB-positive (A+B+) strains. There was no significant difference in disease severity or clinical characteristics between the two groups. Previous use of clindamycin and young age were identified as significant risk factors for the acquisition of A-B+ CDI (OR = 4.738, 95% CI 1.48–15.157, p = 0.009 and OR = 0.966, 95% CI 0.935–0.998, p = 0.038, respectively) in logistic regression.
Rates of resistance to clindamycin were 100% and 69.6% in the A-B+ and A+B+ isolates, respectively (p = 0.006), and the ermB gene was identified in 17 of 21 A-B+ isolates (81%). Resistance to moxifloxacin was also more frequent in the A-B+ than in the A+B+ isolates (95.2% vs. 63.7%, p = 0.004).
Conclusions
The clinical course of A-B+ CDI is not different from that of A+B+ CDI. Clindamycin use is a significant risk factor for the acquisition of tcdA-negative variant strains.
Similar content being viewed by others
Background
Clostridium difficile infection (CDI) is a major cause of healthcare-associated infections worldwide. While the hypervirulent ribotype 027 strain with binary toxin has led to outbreaks only in North America and Europe [1, 2], toxin A-negative (A-B+) C. difficile infections have been reported worldwide [3–5]. The A-B+ strain infections occur in an epidemic or sporadic form worldwide, while A-B+ CDI is more frequent in East Asian countries [4, 6–9].
Initially toxin A was considered to be the most important factor responsible for diarrheal disease [5]; however, several reports have documented that A-B+ strains can lead to similar symptoms, from mild diarrhea to severe pseudomembranous colitis [10, 11]. The risk factors and clinical characteristics of A-B+ CDI have not been clearly documented.
In this study, we investigated the presence of A-B+ strains in healthcare-associated CDI (HA-CDI) in Korea, compared the clinical characteristics of A-B+ CDI and A+B+ (tcdA-positive tcdB-positive) CDI, and analyzed the risk factors for acquisition of A-B+ CDI. We also assessed the susceptibilities to clindamycin and moxifloxacin of the A-B+ and A+B+ strains.
Methods
Setting and study design
This study was conducted at Hanyang University Hospital, a 900-bed tertiary care facility located in Seoul, South Korea. From September 2008 through January 2010, we enrolled suspected HA-CDI patients and recorded prospectively their clinical characteristics. After isolation of C. difficile from patients’ stools, microbiologic studies were performed. To compare the effects of 'toxin A and toxin B' strains and 'toxin B alone' strains on clinical characteristics, patients infected with A+B+ and A-B+ strains were enrolled, while those with binary toxin-positive isolates were excluded. This study was approved by the institutional review board of Hanyang University Hospital (HYUH IRB 2010-R-12).
Definitions
Diarrhea was defined as unformed stools more than 3 times per day on consecutive days, or 6 times within 36 h. CDI was diagnosed when patients had diarrhea and the C. difficile stool isolates contained toxin genes detected by multiplex PCR [12]. We diagnosed HA-CDI when CDI developed at least 72 h after hospitalization, or within 2 months of the last discharge if the patient did spend time in a healthcare facility [13].
Clinical cure was defined as resolution of diarrhea within the treatment period, and clinical failure as the need for treatment change to resolve the diarrhea. Resolution of diarrhea required conversion to two or fewer semi-formed or formed stools per day [14]. Recurrence was defined as growth of toxinogenic C. difficile, a positive A&B toxin assay (VIDAS® C. difficile Toxins A & B; BioMerieux SA, Marcy l’Etoile, France), or a pseudomembrane on endoscopy, with resurgence of symptoms after cessation of treatment at least 10 days after the first episode [15]. Global cure was defined as clinical cure without recurrence.
Collection of data
The demographic and clinical data collected in the study were age, sex, length of hospital stay, medication history within the past 2 months, and underlying diseases including Charlson scores. Installation of a catheter, the use and dosage of antibiotics, probiotics, proton pump inhibitors, H2 blockers, steroids, chemotherapy, and surgical procedure within the past 2 months, were investigated as potential risk factors for CDI [16]. Past medication history was obtained from medical records. The amount of antibiotics administered was expressed as the number of defined daily doses (DDD), which means the total amount of antibiotic divided by the DDD [17].
Vital signs, stool character, stool frequency, and abdominal pain and tenderness were monitored within 24 h after inclusion. Laboratory tests such as white blood cell (WBC) count, albumin and C-reactive protein (CRP) levels were performed within 24 h of enrollment. Age >60 years, temperature >38.3°C, albumin level <2.5 mg/dL, and WBC count >15,000 cells/mm3 received 1 point each, and the scores were summed for each patient. Scores above 2 were defined as severe CDI [18, 19].
Microbiologic studies
Stool specimens were grown anaerobically on C. difficile-selective cycloserine-cefoxitin-taurocholate agar (CCFA-TA, Oxoid Ltd., Cambridge, UK), supplemented with 7% horse blood, after alcohol shock treatment [20]. Colonies of C. difficile were identified with API® Rapid ID 32A (BioMerieux SA, Marcy l’Etoile, France). ATCC 43598 and PCR-ribotype 027 (BI/NAP1/027) were used as internal controls.
Template DNA from stool isolates was used in multiplex PCR to investigate the presence of toxin genes, as described elsewhere, with minor modifications [12]. After the strains producing binary toxin were excluded by multiplex PCR, toxin typing was performed [21].
Minimum inhibitory concentrations (MICs) of clindamycin and moxifloxacin were measured on supplemented Brucella agar by the E-test (AB-BIOdisc, Solna, Sweden), as recommended by the Clinical and Laboratory Standards Institute (CLSI) [22]. C. difficile ATCC 700057 was used as quality control strain for susceptibility testing.
PCR was performed on template DNA to detect the ermB gene [23]. A PCR product of 688 base pairs on electrophoresis was considered a positive result.
Statistical methods
SPSS version 18.0 for Windows (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. Categorical variables were analysed using Pearson’s chi-square test or Fisher’s exact test. Continuous variables were analysed using an independent t-test or the Mann–Whitney U-test. Variables with p-values <0.1 on univariate analysis were included in the multivariate analysis. A p-value <0.05 in a two-tailed test of significance was considered statistically significant.
Results
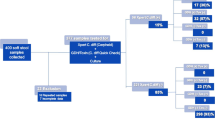
During the study period, 138 C. difficile isolates were obtained from HA-CDI patients; 11 isolates (8.0%) were confirmed by multiplex PCR to have binary toxin genes, 22 (15.9%) were A-B+ strains, and 105 (76.1%) were A+B+ strains. Consequently, 22 patients with A-B+ strains and 105 with A+B+ strains were enrolled.
Comparison of the demographic and clinical characteristics of the A-B+ and A+B+ CDI groups
Demographic and clinical characteristics were compared in the 22 A-B+ patients and the 105 A+B+ patients (Table 1). There were no differences in age, sex, length of hospital stay, history of recent surgery or Charlson score between the two groups; however, more cases of chronic obstructive pulmonary disease (COPD) as an underlying disease were identified in the A-B+ group (p = 0.049). No differences were found between the two groups with regard to factors associated with the CDI severity score. Using a definition of severe CDI adapted from Zar et al. [18, 19], severe CDI occurred at a similar rate in the A+B+ and the A-B+ group (19% and 18.2% respectively, p = 0.925). WBC counts and CRP levels were higher and the albumin level lower in the A+B+ CDI group, but these differences was not statistically significant. Clinical characteristics associated with CDI, such as stool frequency, mucoid stool, abdominal pain or tenderness, were not significantly different between the groups. The A-B+ group was 1.067 times more likely to develop pseudomembranous colitis than the A+B+ group, as measured by odds ratio.
Comparison of outcomes in the A-B+ and A+B+ CDI groups
Of the 127 patients with HA-CDI, 7 were discharged before diagnosis and 3 died from an underlying disease before they had begun treatment. In 32 patients, diarrhea improved or resolved without treatment, and 85 patients (16 A-B+ and 69 A+B+ CDI cases) completed treatment (16/22 vs. 69/105, p = 0.623). Treatment outcomes were compared in the two groups. The treatment regimens (metronidazole or vancomycin) did not differ significantly (p = 0.681). The rates of global cure, failure and mortality were respectively 62.3%, 5.8% and 10.1% in the A+B+ group and 75%, 12.5% and 12.5% in the A-B+ group (p = 0.398, 0.315, and 0.675, respectively) (Table 2). One case of mortality attributable to CDI occurred in the A+B+ group. Additionally 15 of 69 patients (21.7%) in the A+B+ group and none in the A-B+ group (0/16) experienced CDI recurrence after completion of treatment; however, the difference in recurrence rate between the groups did not reach statistical significance (p = 0.063). Clinical outcomes were not significantly different between the metronidazole and vancomycin treatment groups (measured in all patients, p for trend = 0.597).
Comparison of previous medication history in the A-B+ and A+B+ groups
Medication histories of the patients were obtained by retrospective review of their medical records. After excluding 5 cases of HA-CDI due to incomplete medical records, 21 cases of A-B+ CDI and 101 cases of A+B+ CDI were included in the analysis (Table 3).
Previous use of proton pump inhibitors, H2 blockers, probiotics, steroids or chemotherapy was not significantly different between the groups. The most commonly prescribed antibiotics in the A+B+ CDI group were extended-spectrum cephalosporins (ESCs) (56.4%) followed by fluoroquinolones (45.5%) and beta-lactam/beta-lactamase inhibitors (35.6%), while in the A-B+ group they were fluoroquinolones (66.7%), clindamycin (52.4%) or ESCs (52.4%). Calculating the amount of antibiotics as DDD showed that, in the A+B+ group, the order of antibiotics by quantity was similar to the order by frequency of use. In the A-B+ group, antibiotics consumed in largest quantities were fluoroquinolones, clindamycin and ESCs. Finally, a comparison of previous use and amount of antibiotics in the two groups showed that both the use and amount of clindamycin were significantly higher in the A-B+ group (p = 0.001 and p < 0.0001, respectively).
Risk factors for A-B+ strain acquisition
We performed a multivariate logistic regression analysis to identify the risk factors for acquisition of tcdA-negative strains. Age, sex, chronic obstructive pulmonary disease as an underlying disease, albumin level, and quinolone and clindamycin use were included in the analysis. Previous use of clindamycin was found to be a significant risk factor for A-B+ C. difficile infection (OR = 4.738, 95% CI 1.481–15.157, p = 0.009). Age also had a statistically significant effect (OR = 0.966, 95% CI 0.935–0.998, p = 0.038) (Table 4).
Microbiologic studies
Toxinotyping was performed on the 138 isolates of C. difficile; 104 of the 105 A+B+ isolates (99.0%) belonged to toxinotype 0, and one isolate to toxinotype I; all 22 isolates of the A-B+ strain were toxinotype VIII; 8 of 11 binary toxin-producing isolates were toxinotype IV and 3 isolates, toxinotype III.
Clindamycin and moxifloxacin susceptibility testing was performed on only 123 isolates (21 A-B+ isolates and 102 A+B+ isolates) because subculture failed in the case of 4 isolates.
The A-B+ strains had significantly higher resistance rates to clindamycin and moxifloxacin than the A+B+ strains (p = 0.006 and 0.004, respectively). In the A-B+ group, the resistance rates to clindamycin and moxifloxacin were 100% (MIC range 16 6– > 256 mg/L) and 95.2% (MIC range 2–32 mg/L), respectively, whereas in the A+B+ group they were 69.6% and 63.7%, respectively, with wide ranges of MIC (MIC range 1.5– > 256 mg/L and <0.25–128 mg/L, respectively). Resistance to moxifloxacin was correlated with resistance to clindamycin (P < 0.0001). The moxifloxacin resistance rate among clindamycin-resistant A+B+ isolates was 88.7% (63/71) compared with 6.5% (2/31) among clindamycin-susceptible A+B+ isolates. By the same token, the clindamycin resistance rate for the moxifloxacin-resistant A+B + isolates was 96.9% (63/65), but only 21.6% (8/37) for the moxifloxacin-susceptible A+B+ isolates.
ErmB gene-specific PCR revealed that 17 of the 21 A-B+ isolates (81.0%) were ermB-positive. All 17 of these isolates were resistant to clindamycin, and 16 (94.1%) showed high level resistance (MIC > 256 mg/L). Among the 102 A+B+ isolates, there were 49 (48.0%) ermB-positive isolates, 37 of which (75.5%) were highly resistant to clindamycin.
Discussion
The prevalence of toxin A-negative/toxin B-positive (A-B+) strains among CDI isolates varies depending on the country. In most of Europe and in North America, the prevalence of A-B+ strains has been reported to be only 0.2–8% [6, 24]; however, such strains are thought to have caused several outbreaks in those regions. For example, in Poland the prevalence of A-B+ strains has increased from 11% to 45% [25] since there was an outbreak. By contrast, Japan has reported a high prevalence of A-B+ strains; the peak incidence was 39% in 2000 [10], and the incidence decreased to 12.7% in 2005–2008 [8]. A report from Shanghai revealed that A-B+ strains were responsible for a third of all CDI cases [4]. In Korea, the frequency of A-B+ strains was reported to be 4.9% in 1998 [26], and it then increased to 50.9% in 2004–2005 [7, 11]. In our study, the frequency of A-B+ strains among the HA-CDI patients in 2009 was 15.9%. Even given the expected fluctuations in time and space, such a high frequency of A-B+ strains over the East Asian region is interesting. Possible explanations could be similar profiles of antibiotic use, and spread of the pathogen through common foods, as well as the heavy traffic of people across the three countries.
The role of toxin A and toxin B in the pathogenecity of CDI has been debated. Although early studies suggested that toxigenic strains of C. difficile always produce both toxin A and toxin B [5], one study showed that toxin B is the key virulence factor [27]. However, a recent study figured out that C. difficile producing either one or both toxins showed cytotoxic activity in vitro that translated directly into virulence in vivo [28]. Several studies of the clinical characteristics of A-B+ CDI [8, 29–31] have shown that A-B+ strains cause a wide spectrum of diseases from asymptomatic colonization to life-threatening colitis [31], and that there is no significant difference between the clinical manifestations and outcomes of CDI caused by A-B+ and A+B+ strains [10]. However, another study reported more cases of pseudomembranous colitis on endoscopy in A-B+ patients than in A+B+ patients (70% vs. 40%, p = 0.0016) [11]. Furthermore, severe CDI caused by PCR ribotype 017 was responsible for 5% of cases in Germany, and all these cases had lethal outcomes [30]. In another report, the mortality during a PCR ribotype 017 CDI outbreak attributable to that ribotype was similar to that attributable to PCR ribotype 027 (7.0% vs. 6.5%), and higher than that attributable to other types (7.0% vs. 1.6%) [32]. Conversely, in our study there was no significant difference in the clinical characteristics, the laboratory findings or the incidence of pseudomembranous colitis between the study groups, and there were no deaths attributable to A-B+ CDI. The clinical outcomes also did not differ in the two groups, although there was no A-B+ CDI recurrence. In previous reports, the recurrence rate of A-B+ CDI varied from 9 to 35.7% and was similar to that of A+B+ CDI [3, 29]. The reason that we observed no recurrence and no attributable death could be simply that the number of A-B+ cases in this non-epidemic setting was too low. Alternatively it could be because the A-B+ CDI developed in younger patients with lower Charlson scores, or because the shorter hospital stays among the A-B+ group prevented re-infection (however that effect was not statistically significant). Another possibility is that A-B+ CDI recurs less frequently than A+B+ CDI. To investigate this, a further study with a larger number of cases would be necessary.
In order to confirm that clindamycin use is an important risk factor for A-B+ CDI in a clinical setting, we compared the antimicrobial susceptibilities of the A-B+ and A+B+ strains. As expected [33], all 21 A-B+ strains were resistant to clindamycin. High level resistance to clindamycin (MIC > 256 mg/L) in C. difficile is usually linked to the ermB gene, encoding resistance to macrolide-lincosamide-streptogramin B (MLSB) [31]. In this study, the percentage of ermB-positive strains was 53.7% among all C. difficile isolates and 81.0% among the A-B+ isolates. Among the A-B+ strains, 4 of the 21 isolates were ermB-negative, and all of these were highly resistant to clindamycin. Clindamycin resistance in these strains could also be induced by other mechanisms such as other erm genes, efflux mechanisms, or nucleotide substitutions in other genes [34]. Further study of these resistance mechanisms would be desirable.
In previous studies, moxifloxacin resistance was closely associated with clindamycin resistance (95% CI 68–97%) [34, 35]. It was reported that, of isolates resistant to fluoroquinolones, 98% were resistant to either erythromycin or clindamycin, although fluoroquinolone resistance in the absence of MLSB resistance was rare [35]. Similarly, in our study, resistance to moxifloxacin was highly correlated with clindamycin resistance (p < 0.0001). An efflux pump may be responsible for the co-resistance to the MLSB drugs and fluoroquinolone.
Conclusions
The prevalence of A-B+ strains among HA-CDI patients in Korea was 15.9%. Disease severity and clinical characteristics were not significantly different between the A-B+ and the A+B+ patients. Previous use of clindamycin is a risk factor for acquisition of an A-B+ strain, and all tested A-B+ isolates were highly resistant to clindamycin. Heavy use of clindamycin appears to facilitate A-B+ strain infection in our hospitals.
References
Warny M, Pepin J, Fang A, Killgore G, Thompson A, Brazier J, Frost E, McDonald LC: Toxin production by an emerging strain of Clostridium difficile associated with outbreaks of severe disease in North America and Europe. Lancet. 2005, 366: 1079-1084. 10.1016/S0140-6736(05)67420-X.
Clements AC, Magalhaes RJ, Tatem AJ, Paterson DL, Riley TV: Clostridium difficile PCR ribotype 027: assessing the risks of further worldwide spread. Lancet Infect Dis. 2010, 10: 395-404. 10.1016/S1473-3099(10)70080-3.
Goorhuis A, Legaria MC, van den Berg RJ, Harmanus C, Klaassen CH, Brazier JS, Lumelsky G, Kuijper EJ: Application of multiple-locus variable-number tandem-repeat analysis to determine clonal spread of toxin A-negative Clostridium difficile in a general hospital in Buenos Aires, Argentina. Clin Microbiol Infect. 2009, 15: 1080-1086. 10.1111/j.1469-0691.2009.02759.x.
Huang H, Wu S, Wang M, Zhang Y, Fang H, Palmgren AC, Weintraub A, Nord CE: Clostridium difficile infections in a Shanghai hospital: antimicrobial resistance, toxin profiles and ribotypes. Int J Antimicrob Agents. 2009, 33: 339-342. 10.1016/j.ijantimicag.2008.09.022.
Lyerly DM, Krivan HC, Wilkins TD: Clostridium difficile: its disease and toxins. Clin Microbiol Rev. 1988, 1: 1-18.
Bauer MP, Notermans DW, van Benthem BH, Brazier JS, Wilcox MH, Rupnik M, Monnet DL, van Dissel JT, Kuijper EJ: Clostridium difficile infection in Europe: a hospital-based survey. Lancet. 2011, 377: 63-73. 10.1016/S0140-6736(10)61266-4.
Kim H, Riley TV, Kim M, Kim CK, Yong D, Lee K, Chong Y, Park JW: Increasing prevalence of toxin A-negative, toxin B-positive isolates of Clostridium difficile in Korea: impact on laboratory diagnosis. J Clin Microbiol. 2008, 46: 1116-1117. 10.1128/JCM.01188-07.
Iwashima Y, Nakamura A, Kato H, Wakimoto Y, Wakiyama N, Kaji C, Ueda R: A retrospective study of the epidemiology of Clostridium difficile infection at a University Hospital in Japan: genotypic features of the isolates and clinical characteristics of the patients. J Infect Chemother. 2010, 16: 329-333. 10.1007/s10156-010-0066-4.
Elliott B, Squire MM, Thean S, Chang BJ, Brazier JS, Rupnik M, Riley T: New types of toxin A-negative, toxin B-positive strains among clinical isolates of Clostridium difficile in Australia. J Med Microbiol. 2011, 10: 10-
Komatsu M, Kato H, Aihara M, Shimakawa K, Iwasaki M, Nagasaka Y, Fukuda S, Matsuo S, Arakawa Y, Watanabe M, Iwatani Y: High frequency of antibiotic-associated diarrhea due to toxin A-negative, toxin B-positive Clostridium difficile in a hospital in Japan and risk factors for infection. Eur J Clin Microbiol Infect Dis. 2003, 22: 525-529. 10.1007/s10096-003-0992-5.
Shin BM, Kuak EY, Yoo SJ, Shin WC, Yoo HM: Emerging toxin A-B + variant strain of Clostridium difficile responsible for pseudomembranous colitis at a tertiary care hospital in Korea. Diagn Microbiol Infect Dis. 2008, 60: 333-337. 10.1016/j.diagmicrobio.2007.10.022.
Persson S, Torpdahl M, Olsen KE: New multiplex PCR method for the detection of Clostridium difficile toxin A (tcdA) and toxin B (tcdB) and the binary toxin (cdtA/cdtB) genes applied to a Danish strain collection. Clin Microbiol Infect. 2008, 14: 1057-1064. 10.1111/j.1469-0691.2008.02092.x.
Mulvey MR, Boyd DA, Gravel D, Hutchinson J, Kelly S, McGeer A, Moore D, Simor A, Suh KN, Taylor G, et al: Hypervirulent Clostridium difficile strains in hospitalized patients, Canada. Emerg Infect Dis. 2010, 16: 678-681.
Louie T, Miller M, Donskey C, Mullane K, Goldstein EJ: Clinical outcomes, safety, and pharmacokinetics of OPT-80 in a phase 2 trial with patients with Clostridium difficile infection. Antimicrob Agents Chemother. 2009, 53: 223-228. 10.1128/AAC.01442-07.
Barbut F, Richard A, Hamadi K, Chomette V, Burghoffer B, Petit JC: Epidemiology of recurrences or reinfections of Clostridium difficile-associated diarrhea. J Clin Microbiol. 2000, 38: 2386-2388.
O'Connor JR, Johnson S, Gerding DN: Clostridium difficile infection caused by the epidemic BI/NAP1/027 strain. Gastroenterology. 2009, 136: 1913-1924. 10.1053/j.gastro.2009.02.073.
World Health Organisation: The anatomical therapeutic chemical (ATC) and defined daily dosing (DDD) system index. 2010, [http://www.whocc.no/]
Zar FA, Bakkanagari SR, Moorthi KM, Davis MB: A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007, 45: 302-307. 10.1086/519265.
Fujitani S, George WL, Murthy AR: Comparison of Clinical Severity Score Indices for Clostridium difficile Infection. Infect Control Hosp Epidemiol. 2011, 32: 220-228. 10.1086/658336.
Clabots CR, Gerding SJ, Olson MM, Peterson LR, Gerding DN: Detection of asymptomatic Clostridium difficile carriage by an alcohol shock procedure. J Clin Microbiol. 1989, 27: 2386-2387.
Rupnik M, Avesani V, Janc M, von Eichel-Streiber C, Delmee M: A novel toxinotyping scheme and correlation of toxinotypes with serogroups of Clostridium difficile isolates. J Clin Microbiol. 1998, 36: 2240-2247.
Clinical and Laboratory Standards Institute: Methods for antimicrobial susceptibility testing of Anaerobic bacteria; Approved standard, 7th edition. 2007, CLSI document M11-A7, Wayne, PA, USA
Johnson S, Samore MH, Farrow KA, Killgore GE, Tenover FC, Lyras D, Rood JI, DeGirolami P, Baltch AL, Rafferty ME, et al: Epidemics of diarrhea caused by a clindamycin-resistant strain of Clostridium difficile in four hospitals. N Engl J Med. 1999, 341: 1645-1651. 10.1056/NEJM199911253412203.
Cheknis AK, Sambol SP, Davidson DM, Nagaro KJ, Mancini MC, Hidalgo-Arroyo GA, Brazier JS, Johnson S, Gerding DN: Distribution of Clostridium difficile strains from a North American, European and Australian trial of treatment for C. difficile infections: 2005–2007. Anaerobe. 2009, 15: 230-233. 10.1016/j.anaerobe.2009.09.001.
Pituch H, Obuch-Woszczatynski P, Wultanska D, Nurzynska G, Harmanus C, Banaszkiewicz A, Radzikowski A, Luczak M, van Belkum A, Kuijper E: Characterization and antimicrobial susceptibility of Clostridium difficile strains isolated from adult patients with diarrhoea hospitalized in two university hospitals in Poland, 2004–2006. J Med Microbiol. 2011, 60: 1200-1205. 10.1099/jmm.0.029801-0.
Chung Y, Chung GT, Seong WK, Oh HB: Molecular Analysis of Clostridium difficile Isolates by Arbitrarily Primed-Polymerase Chain Reaction and Polymerase Chain Reaction-Ribotyping. Korean J Infect Dis. 2002, 34: 167-175. 10.1086/338234.
Lyras D, O'Connor JR, Howarth PM, Sambol SP, Carter GP, Phumoonna T, Poon R, Adams V, Vedantam G, Johnson S, et al: Toxin B is essential for virulence of Clostridium difficile. Nature. 2009, 458: 1176-1179. 10.1038/nature07822.
Kuehne SA, Cartman ST, Heap JT, Kelly ML, Cockayne A, Minton NP: The role of toxin A and toxin B in Clostridium difficile infection. Nature. 2010, 467: 711-713. 10.1038/nature09397.
Alfa MJ, Kabani A, Lyerly D, Moncrief S, Neville LM, Al-Barrak A, Harding GK, Dyck B, Olekson K, Embil JM: Characterization of a toxin A-negative, toxin B-positive strain of Clostridium difficile responsible for a nosocomial outbreak of Clostridium difficile-associated diarrhea. J Clin Microbiol. 2000, 38: 2706-2714.
Arvand M, Hauri AM, Zaiss NH, Witte W, Bettge-Weller G: Clostridium difficile ribotypes 001, 017, and 027 are associated with lethal C. difficile infection in Hesse, Germany. Euro Surveill. 2009, 14: 19403-
Drudy D, Fanning S, Kyne L: Toxin A-negative, toxin B-positive Clostridium difficile. Int J Infect Dis. 2007, 11: 5-10. 10.1016/j.ijid.2006.04.003.
Goorhuis A, Debast SB, Dutilh JC, van Kinschot CM, Harmanus C, Cannegieter SC, Hagen EC, Kuijper EJ: Type-Specific Risk Factors and Outcome in an Outbreak With 2 Different Clostridium difficile Types Simultaneously in 1 Hospital. Clin Infect Dis. 2011, 53: 860-869. 10.1093/cid/cir549.
Pituch H, Van Belkum A, Van Den Braak N, Obuch-Woszczatynski P, Verbrugh H, Meisel-Mikolajczyk F, uczak M: Recent emergence of an epidemic clindamycin-resistant clone of Clostridium difficile among Polish patients with C. difficile-associated diarrhea. J Clin Microbiol. 2003, 41: 4184-4187. 10.1128/JCM.41.9.4184-4187.2003.
Ackermann G, Degner A, Cohen SH, Silva J, Rodloff AC: Prevalence and association of macrolide-lincosamide-streptogramin B (MLS(B)) resistance with resistance to moxifloxacin in Clostridium difficile. J Antimicrob Chemother. 2003, 51: 599-603. 10.1093/jac/dkg112.
Solomon K, Fanning S, McDermott S, Murray S, Scott L, Martin A, Skally M, Burns K, Kuijper E, Fitzpatrick F, et al: PCR ribotype prevalence and molecular basis of macrolide-lincosamide-streptogramin B (MLSB) and fluoroquinolone resistance in Irish clinical Clostridium difficile isolates. J Antimicrob Chemother. 2011, 66: 1976-1982. 10.1093/jac/dkr275.
Pre-publication history
The pre-publication history for this paper can be accessed here:http://www.biomedcentral.com/1471-2334/12/109/prepub
Acknowledgments
This work was supported by a grant from the National Research Foundation of Korea (KRF-2011-0014685). Moxifloxacin E-test strips (AB-BIOdisc, Solna, Sweden) were kindly provided by BioMerieux SA, Korea.
Presented in part at the 51st Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); Sept. 17–20, 2011, Chicago (slide K1716)
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
J Kim contributed to protocol preparation, data collection and analysis, as well as manuscript preparation. H Pai provided the funding for this study and made a major contribution to the interpretation of data and appraisal of the manuscript. M Seo participated in the microbiologic studies, and J O Kang contributed to the microbiologic studies, discussion of results and revision of the manuscript. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Kim, J., Pai, H., Seo, Mr. et al. Clinical and microbiologic characteristics of tcdA-negative variant clostridium difficile infections. BMC Infect Dis 12, 109 (2012). https://doi.org/10.1186/1471-2334-12-109
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1471-2334-12-109