Abstract
Background
Paspalum (Poaceae) is an important genus of the tribe Paniceae, which includes several species of economic importance for foraging, turf and ornamental purposes, and has a complex taxonomical classification. Because of the widespread interest in several species of this genus, many accessions have been conserved in germplasm banks and distributed throughout various countries around the world, mainly for the purposes of cultivar development and cytogenetic studies. Correct identification of germplasms and quantification of their variability are necessary for the proper development of conservation and breeding programs. Evaluation of microsatellite markers in different species of Paspalum conserved in a germplasm bank allowed assessment of the genetic differences among them and assisted in their proper botanical classification.
Results
Seventeen new polymorphic microsatellites were developed for Paspalum atratum Swallen and Paspalum notatum Flüggé, twelve of which were transferred to 35 Paspalum species and used to evaluate their variability. Variable degrees of polymorphism were observed within the species. Based on distance-based methods and a Bayesian clustering approach, the accessions were divided into three main species groups, two of which corresponded to the previously described Plicatula and Notata Paspalum groups. In more accurate analyses of P. notatum accessions, the genetic variation that was evaluated used thirty simple sequence repeat (SSR) loci and revealed seven distinct genetic groups and a correspondence of these groups to the three botanical varieties of the species (P. notatum var. notatum, P. notatum var. saurae and P. notatum var. latiflorum).
Conclusions
The molecular genetic approach employed in this study was able to distinguish many of the different taxa examined, except for species that belong to the Plicatula group, which has historically been recognized as a highly complex group. Our molecular genetic approach represents a valuable tool for species identification in the initial assessment of germplasm as well as for characterization, conservation and successful species hybridization.
Similar content being viewed by others
Background
The genus Paspalum L. is an important member of the Paniceae tribe (Poaceae) and includes between 330 and 400 species, most of which are native to the tropical and subtropical regions of the Americas [1–3]. Paspalum species can be found in diverse habitats, such as subtropical rainforests, savannas, marshes and dunes, but they are more frequently found in the natural grasslands of eastern Bolivia, Paraguay, central and southern Brazil, northern Argentina and Uruguay [3]. The main center of origin and diversity of the genus is considered to be located in the South American tropics and subtropics [1], primarily in central Brazil, where numerous species appear to be associated with savannas and rocky terrain. Brazil harbors the greatest number of Paspalum species [4, 5], with approximately 220 species that exist in nearly all herbaceous plant communities within different ecosystems [6].
Because of this degree of complexity, the division of the Paspalum genus into subgenera, sections or informal groups has been proposed by many authors and has been extensively discussed [1–3, 7–16]. Currently, four subgenera are recognized within Paspalum: Paspalum subg. Anachyris Chase, P. subg. Ceresia (Pers.) Rchb., P. subg. Harpostachys (Trin.) S. Denham and P. subg. Paspalum[3], the last of which consists of approximately 265 species divided into 25 informal groups [1, 14]. However, such rankings have been based on morphological similarities, and the evolutionary and genetic relationships between these groups are not always clear. Several species, mainly within Paspalum subg. Paspalum, are of economic importance for foraging, turf and ornamental purposes [17] in different parts of the world. Dallisgrass (Paspalum dilatatum Poir., Dilatata group) and bahiagrass (P. notatum Flüggé, Notata group) are especially important and are widely used for forage, mainly in the southern United States of America (USA) [18, 19]. P. atratum Swallen (Plicatula group) has been the object of growing interest for use as forage in areas that are subjected to periodic flooding in Florida (USA), northeastern Argentina, Brazil, Thailand, the Philippines and Australia [18]. In addition, Paspalum scrobiculatum L. (‘Kodo millet’, Plicatula group) is cultivated in India as a cereal crop [3, 20], and Paspalum vaginatum Sw. (Disticha group) [21] and P. notatum[19] are widely grown as turf grass.
Because of the widespread interest in several species of this genus, many accessions have been conserved in germplasm banks and distributed throughout various countries for cultivar development and cytogenetic studies. However, the use of some accessions is restricted due to difficulty with accurate taxonomical identification. In Brazil, despite constant evaluations of germplasm banks and taxonomic reviews [22], many species of Paspalum, especially those included in the Plicatula Group, remain unidentified. This deficit presents a problem because the correct identification of germplasms and the quantification of their variability are necessary for the development of conservation and breeding programs.
The complexity of the taxonomical classification of Paspalum germplasms results from the complex evolutionary history of the group. Polyploidy occurs at a high frequency within the genus [23, 24] and has played a crucial role in the evolution of Paspalum. Most species have × = 10 as the basic chromosome number, with ploidy levels that range from diploid to hexadecaploid [25]. Diploid species are not rare within the genus [3, 25–32], but nearly 80% of the investigated species are polyploids, among which 50% are tetraploid, and most of these tetraploids are apomictic [24, 33]. Many Paspalum species comprise sexual diploid and apomictic polyploid cytotypes, and several have been shown to have arisen through natural hybridization [34]. However, interspecific hybridization and allopolyploidy are not always morphologically evident in Paspalum species; therefore, additional methods of taxonomic classification are required [35].
Many taxonomical and species characterization problems arise as a consequence of the great morphological variation present in agamic complexes. P. notatum is a good example of this situation, because it forms an agamic complex and presents wide morphological variation [7, 36, 37]. This species is a perennial rhizomatous turf and forage grass and is recognized as a major constituent of the native grasslands of the New World, being found from Central Eastern Mexico to Argentina and throughout the West Indies [1]. This grass is economically important and is widely used for forage production, mainly in the southern USA [19].
Various morphological and cytological forms are recognized within P. notatum[7, 37]. This species includes several genotypes, which differ in both their ploidy levels and their reproductive systems. The diploid cytotype (2n = 2× = 20) is sexual and self-incompatible [38, 39], whereas the tetraploid cytotype (2n = 4× = 40) is a self-compatible pseudogamous aposporous apomict [40]. Apomixis in tetraploid P. notatum can be either obligate or facultative [24]. In botanical terms, the tetraploid cytotypes are usually considered to be the typical form of P. notatum; as such, they form the variety notatum. On the other hand, the diploid cytotypes are classified as belonging to the saurae variety based on their distinct morphological characteristics [36]. In addition to these two widely recognized varieties, Döll [41] proposed the variety latiflorum, which was accepted by some taxonomists [36, 42, 43] until the mid-1980s. However, P. notatum var. latiflorum is not currently recognized.
Molecular makers are of great value in plant studies and have been used for multiple purposes, including estimating the genetic diversity, determining accession relationships, elucidating evolutionary relationships, aiding in the taxonomic classification of many plants [44–49] and aiding in the identification of botanical varieties [50, 51]. In addition, utilizing molecular markers that regularly identify many genetic polymorphisms at low taxonomic levels makes it possible to address the relationship between morphological and genotypic variation.
The molecular markers that are more informative for the discrimination of closely related genotypes include microsatellite sequence markers [52–54]. Microsatellites are tandem repeat sequences of 1 to 6 nucleotides that are widely distributed in the genome [55]. However, these markers are usually species specific, and in the Paspalum genus, few microsatellite markers are available for P. vaginatum[56], P. dilatatum and the related species [57], P. notatum[58] and P. atratum[59]. However, primers designed for source species have been successfully employed to amplify nuclear SSRs in closely related taxa when the DNA regions that flank the microsatellite loci are sufficiently conserved [57, 59–67].
The main objectives of this study are (1) to evaluate the informative potential of SSRs for genetic discrimination in different species of Paspalum using markers developed for P. atratum and P. notatum; (2) to conduct an in-depth study of the extent, distribution and structure of the genetic variation of P. notatum in a South American collection of this species; and (3) to evaluate the genetic patterns and relationships among different morphological types found in this species and correlate them with their geographic distribution.
Methods
Plant material and DNA extraction
A total of 214 accessions of Paspalum (177 accessions sampled from 35 species and 37 unclassified accessions) were included in this study. These accessions have been maintained in vivo at Embrapa Southeast Livestock, located in São Carlos, São Paulo (SP), Brazil and at the Federal University of Rio Grande do Sul in Porto Alegre, Rio Grande do Sul (RS), Brazil (Additional file 1). Fifty-seven P. notatum accessions from germplasm collections were evaluated in a more detailed analysis in this study (Additional file 2). The genomic formula, chromosome number, type of polyploidy and mode of reproduction are presented for all Paspalum species investigated in this study (Additional file 3).
Genomic DNA from each sample was isolated from lyophilized young leaf tissue using the cetyltrimethyl ammonium bromide (CTAB) method proposed by Doyle and Doyle [68] with minor modifications. DNA concentrations were estimated by comparison with known concentrations of λ DNA on 0.8% agarose gels.
Development of new SSR markers for Paspalumand SSR analysis
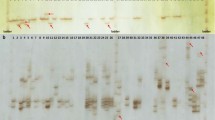
A microsatellite-enriched genomic library was obtained for P. notatum using the method described by Billotte et al. [69]. The steps for obtaining the clones, sequencing and analysis, the determination of the criteria for selecting the microsatellites, the primer design and the amplification were conducted according to previously described methodology [58]. For some loci, a touchdown protocol was used, as previously described [70]. Only the strongest bands were considered because of the possibility that lighter bands could be stutter bands that result from the slippage of Taq polymerase during the PCR amplifications [63, 71].
Cross-species amplification was tested for 214 accessions that correspond to 35 Paspalum species, with a set of 23 SSR primer pairs developed for P. atratum and P. notatum[58, 59] and new SSRs developed in this work. An SSR was considered to be transferable when a band of the expected size was amplified via PCR and an SSR pattern was observed. To avoid false negatives, primers that resulted in null alleles in some of the samples were tested at least twice. The microsatellite loci that were amplified successfully and that showed readable electrophoretic patterns and an absence of nonspecific products were used for the genetic evaluation of the germplasm collection.
A more detailed and accurate analysis of the P. notatum accessions was conducted using 15 microsatellite markers developed by Cidade et al. [58, 59] and 15 new SSRs developed in this work.
The total Paspalum germplasm and the P. notatum accessions were genotyped and analyzed in the same manner. No assumptions were made about the genetic nature of the examined alleles because of the high ploidy levels of the majority of the Paspalum accessions. Hence, each SSR allele was treated as dominant in this study due to the high ploidy levels of the samples [72]; these alleles will hereafter be referred to as bands. Each allele was scored as 1 (present) or 0 (absent) and was arranged in a matrix. Non-amplified loci were scored as missing data. Summary statistics, including the total number of bands, the number of bands per locus, and the polymorphism information content (PIC) [72] were determined based on the data matrix that documented the microsatellite genotyping of each locus. To verify the information obtained for these SSR loci for future genetic studies of other Paspalum species, the number of bands and the PIC values were calculated for each marker within each species.
Clustering and population structure analyses
For the analysis of the Paspalum germplasm, genetic similarity (GS) indices were calculated for all the possible pairwise comparisons using Dice’s similarity coefficient [73]. For calculation of the P. notatum GS indices, Jaccard´s similarity index was used [74]. All of the calculations were performed with the software package NTSYS-pc 2.1 [75]. For both analyses, clustering was performed using the Unweighted Pair Group Method with Arithmetic Mean (UPGMA) [76], and the significance of the cophenetic correlation was tested with the Mantel correspondence test [77]. Additionally, a PCO was conducted.
An unrooted dendrogram was constructed for both analyses using the weighted neighbor-joining method (NJ) with DARwin 5.0.157 software [78], and trees were drawn with Figtree v. 1.3.1 software [79]. The bootstrap method was employed to evaluate the reliability of the tree topology. Bootstrap calculations were performed using BOOD 3.0 [80] and DARwin 5.0.157 [78] software based on 1000 replications. The cophenetic coefficient between the matrix of genetic similarity and the dendrogram was computed using NTSYS-pc 2.1 software [75].
The program STRUCTURE, version 2.3.2 [81–83], was used to detect the population structure and to assign individuals to subpopulations. This program employs model-based clustering in which a Bayesian approach identifies groups based on compliance with Hardy–Weinberg equilibrium and linkage equilibrium. In this study, membership in each genotype was tested for the range of genetic groups, from K = 1 to K = 12, with the admixture model and without prior information regarding their origin, using 300,000 replicates for burn-in and 500,000 replicates for Markov Chain Monte Carlo processes for each run. Analyses were conducted with correlated allele frequencies (AFC) for the putative populations. The final subgroups were determined based on the ad hoc measure ΔK [84], which was calculated using an application on the STRUCTURE Harvester website [85].
The binary matrix derived from the SSR data was examined via an analysis of molecular variance (AMOVA) using Arlequin 3.11 software [86]. The partitioning of the genetic variation within Paspalum was calculated, and the significance of each variation was estimated non-parametrically using 1000 permutations. An AMOVA was also performed to evaluate the differentiation between the P. notatum groups obtained from STRUCTURE. Furthermore, the genetic distances among these groups were calculated as the Slatikin distance and pairwise ϕST.
Morphological traits of Paspalum notatum
Data regarding nine morphological traits from [87] and unpublished data from Batista (2008) were used for the morphological analysis of P. notatum. The following vegetative traits were included in this analysis: leaf sheath length (LSL), leaf sheath width (LSW), leaf blade length (LBL), leaf blade width (LBW) and leaf blade hairiness (LBH). In addition, the following four reproductive traits were examined: inflorescence stem length (ISL), average length of inflorescence branches (ALIB), spikelet length (SL) and spikelet width (SW). The plant material was botanically identified according to Parodi [36], Barreto [7] and Canto-Dorow [37].
A data matrix was created that included 46 accessions and nine morphological traits, which comprised a mixed matrix that was composed of one qualitative and eight quantitative traits. The data were standardized by dividing each real value by the standard deviation of the trait. A PCO was performed, and descriptive statistics were calculated using GENES software [88]. Pairwise matrices of Euclidean distance were calculated using NTSYS-pc 2.1 software [75]. Based on the results obtained via principal coordinate analysis (PCA) and PCO, a graph was constructed using STATISTICA software, version 7.0 [89].
To estimate the correlation between the morphological distances and molecular similarities, a Jaccard’s similarity matrix was constructed based on molecular genotyping of the morphologically evaluated accessions. Correlations were calculated using the Mantel test [77] in NTSYS-pc 2.1 software [75].
Geographic origin of Paspalum notatum
The geographic coordinates of the P. notatum accessions were used to construct a matrix of linear distances (in kilometers) between pairs of genotypes using Geographic Distance Matrix Generator software [90]. The correlation between this matrix and the genetic similarity matrix (generated using microsatellites) was tested with NTSYS-pc 2.1 software using the Mantel test. The geographic distribution of the accessions was visualized with the DIVA-GIS program, version 5.2.0.2 [91].
Results
Development of new SSR markers for Paspalumand SSR analysis
Seventeen new SSR polymorphic loci were developed for the investigated Paspalum species (Table 1), among which 15 were isolated from P. notatum and two from P. atratum. A total of 121 bands were scored; the number of bands per locus ranged from 4 to 15, with an average of 7.12 bands per locus (Table 2).
Of the 23 microsatellite primer pairs tested for transferability to other species, 12 (52%) successfully amplified their respective loci in most species (Additional file 4). Exceptions were found for the PA01-C8 locus, which showed weak amplification in Paspalum ovale Nees ex Steud., and the PN03-A6 locus, which showed weak amplification in Paspalum pumilum Ness, Paspalum subciliatum Chase, and P. ovale. All 12 of the SSRs that transferred to other species revealed a high degree of SSR polymorphism within the surveyed species, which allowed an analysis of the organization of genetic diversity within the Paspalum germplasm to be conducted. The mean PIC was 0.65, and an average of 15.58 bands per locus was found (Table 2). The PA02-B1, PA01-B7, PN03-C7, PN03-A6, and PN02-B5B loci were the most informative, with more than 20 bands being amplified for each of these loci (Additional file 4).
Among the P. notatum accessions, the 30 microsatellite primer pairs evaluated amplified a total of 208 bands, with an average of 6.93 fragments per locus and a mean PIC of 0.64 being obtained (Table 2). The combined electrophoretic profile of all of the loci revealed 44 distinct genotypes, which suggests that there were duplicates or clones in the analyzed material.
Clustering analyses in the Paspalumgenus
All of the analyses performed supported the formation of three major groups in the evaluated Paspalum germplasm (Figures 1, 2 and 3), which corresponded to the Paspalum botanical groups Notata and Plicatula and a group formed by all the other species of Paspalum. Because of the differences between the matrices obtained when analyzing different microsatellite loci, we will address only the major groups and the most robustly supported groups derived from distinct methods of analysis.
Principal coordinate analysis based on Dice’s similarity matrix for the 214 accessions from the Paspalum species. Principal coordinate analysis (goodness-of-fit 0.86 (P < 0.001)) based on Dice’s similarity matrix for the 214 accessions from the Paspalum species from the analysis of 187 SSR bands. The three ordination factors together explain 40.21% of the variation in the data matrix. “A” is a bi-dimensional scatter plot, and “B” is a tri-dimensional scatter plot.
The PCO showed a goodness-of-fit value of 0.86 (P < 0.001) and indicated the existence of three main groups (Figure 1), which correspond to the Plicatula group (except for some dispersed species) and the Notata group (P. notatum, P. pumilum, P. subciliatum, Paspalum cromyorhizon Trin. ex Döll and Paspalum ionanthum Chase, with the last two being grouped more distantly from the other species in this group), and to a third group that is composed of other species from different botanical groups of Paspalum (Additional file 1). The three-dimensional PCO scatter plot (Figure 1B) showed the same trend of a tripartite division; however, the species of the Plicatula group were more dispersed. The three principal vectors, PC1 (26.77), PC2 (9.52) and PC3 (3.92), accounted for 40.21% of the variation. The NJ trees based on Dice´s similarity coefficient support the results of the PCO analysis, identifying three main groups – the Notata Group, the Plicatula Group and a third group formed by species from other botanical groups (Figure 2).
The Bayesian analysis of the population structure using a model-based approach provided support for a genetic division of the Paspalum germplasm into three distinct groups (Figure 3), with K = 3 (Additional file 5). Out of the twelve runs for K = 3, the run with the highest likelihood value was selected to assign posterior membership coefficients (Q) to each accession, which supported the results obtained from the other methods of analysis, described previously (Figure 3).
The inter-species pair-wise genetic dissimilarity among the 214 accessions that belong to different Paspalum species varied from 0.00 among some P. notatum accessions to 0.80 among Paspalum conjugatum P.J.Bergius and Paspalum rhodopedum L.B.Sm. & Wassh., with an overall average of 0.43 being obtained.
Population structure in Paspalum notatum
The Bayesian analysis performed with STRUCTURE software revealed two main values of K (Additional file 6). Figure 4 shows sub-figures for two K values up to the number of groups detected in UPGMA because both levels of subdivisions were instructive. K = 2 separates individuals from S. Tome (Argentina) from the accessions from other localities, and the K = 7 groups show correspondence to the botanical varieties of P. notatum. The accessions from groups A, B, C and D belong to the variety notatum, while the accessions from group E and some of the accessions from groups F and G belong to the variety latiflorum. The saurae variety accessions belong to the same genetic group (G). The number of accessions that correspond to each botanical variety and to each of the seven STRUCTURE groups with more than a 50% membership probability is shown in Additional file 7. The M54 accession from the Guaíba region, RS, Brazil, showed a mixed group membership, which corresponded to both groups D (0.49) and G (0.487); however, this accession exhibits morphological traits that are more similar to group G. Group F corresponds to the accessions from S. Tome (Argentina), which separated when K = 2.
Similarity and distance-based methods (PCA, NJ, UPGMA) (Figures 5, 6, 7) revealed accession relationships that were consistent with the STRUCTURE-based membership assignment for most of the accessions (using K = 7, Figure 4), as indicated by the colors in the UPGMA dendrogram (Figure 6). On the other hand, the groups formed in PCA (Figure 7) indicate a correlation between the botanical varieties observed in P. notatum. The coefficients of similarity for all of the P. notatum accessions ranged from 0.17 to 1.0, with an average value of 0.42 being obtained. The cophenetic correlation coefficient in the UPGMA dendrogram was 0.92, which suggests that there were low levels of distortion between the matrix and the phenograms.
Principal component analysis based on Jaccard’s similarity for P . notatum . PCA based on Jaccard’s similarity, which resulted from 57 accessions of Paspalum notatum. The branch colors correspond to the colors of the STRUCTURE clusters from Figure 4, K=7.
Dendrogram based on Jaccard´s similarity coefficient for 57 Paspalum notatum accessions. An UPGMA dendrogram based on Jaccard´s similarity coefficient for 57 Paspalum notatum accessions. The branch colors correspond to the colors in the STRUCTURE clusters from Figure 4, K=7.
Principal component analysis based on nine morphological traits for P . notatum . A principal component analysis was based on nine morphological traits in 46 Paspalum notatum accessions. The branch colors correspond to the colors in the STRUCTURE clusters from Figure 4, K=7.
The AMOVA showed highly significant (P < 0.001) differentiation among groups A, B, C, D, E, F and G, which indicates that approximately 47% of the observed variation can be explained by the subdivision of the accessions into these seven groups (Table 3).
In the NJ (not shown) and UPGMA trees (Figure 6), greater genetic divergence was observed among the saurae variety accessions in comparison to other varieties, and saurae behaved as an outgroup in the dendrogram. This variety is distinguished from the others mainly by its ploidy level (diploid) and reproductive system (sexual self-incompatible), while P. notatum var. notatum and P. notatum var. latiflorum are either obligate or facultative tetraploid apomictics.
Exclusive bands were found between the different varieties (Table 4); however, these bands were not present in all of the accessions of a given variety but were instead differentially distributed between the corresponding gene groups.
Genetic diversity and relationships among the seven groups of Paspalum notatumgroups
Descriptive statistics for the P. notatum groups detected via STRUCTURE analysis (Table 5) revealed that groups E and G exhibited the highest genetic diversity. The overall ϕST among the seven groups was 0.47 (95% confidence interval, p < 0.001), with the ϕST value for each cluster ranging from 0.46 to 0.52. Pairwise comparisons on the basis of the ϕST values can be interpreted as standardized population distances between any two groups. The pairwise ϕST values obtained in this work ranged from 0.181 between groups E and G to 0.803 between groups B and F (Table 6). The genetic distance data agreed with the ϕST estimates. The smallest genetic distance was observed between groups E and G (0.222), whereas groups F and B presented the greatest genetic distance (4.075).
Morphological and molecular results from P. notatum
The PCA results indicated three main groups (Figure 7), which correspond to the three botanical varieties accepted by Parodi [36]. The features that contributed most to the morphological divergence between the groups were ALIB and LBL in principal component 1 (PC1) and SL and SW in principal component 2 (PC2), which together accounted for more than 55% of the detected variation. The group shown in dark green in the upper graph was botanically identified as var. latiflorum due to the size of its spikelets and the length of its inflorescence branches. However, the LBL and LBW were smaller than in other accessions that belong to var. latiflorum, which likely accounts for this difference. The results of the descriptive analysis (Additional files 8 and 9) and correspondence between informal Paspalum botanical groups and the STRUCTURE analysis groups (Additional file 10) are presented.
The correlation between the morphological traits and the SSR markers indicated a weak correlation between the morphological and molecular data (r = 0.42; p < 0.001).
Geographic distance and genetic dissimilarity in Paspalum notatum
The correlation between the geographic distance and the genetic similarity matrix was estimated for all the accessions and revealed no significant association between genetic and geographic variation based on r = 0.0017 for the Mantel test (Additional file 11).
Discussion
Microsatellite markers
The P. notatum and P. atratum marker sets revealed polymorphisms in many of the other Paspalum species analyzed, which suggests that they are useful in genetic analyses of the Paspalum genus.
Some SSRs were highly polymorphic among the evaluated loci, and according to Huang et al. [92], the correlation between the number of bands revealed per locus and the PIC value is equivalent to Nei’s gene diversity [93]. Testolin et al. [94] reported that highly polymorphic markers are promising for use in DNA fingerprinting to identify plant varieties or cultivars. We recommend that a subset of these loci be used in future fingerprinting studies, with a preference for loci that are easy to score and that exhibit high heterozygosity, as indicated by high PIC values.
The genetic structure of the three genetic groups was confirmed using an AMOVA, where 46.83% of the variation was explained by the division into the three groups (Table 7). This finding suggests that the different species could contain unique alleles or genotypes and that these data can discriminate between taxa, especially the more distant taxa. Similar results were obtained by Jungmann et al. [47], who found that 44% of the variation in apomictic Brachiaria humidicola (Rendle) Schweick accessions could be explained by the subdivision of the germplasm into five groups.
The proportion of detected polymorphisms decreased as the genetic distance increased. This finding corroborates other results previously obtained in both plant [61, 66] and animal studies [95]. We observed a reduced number of bands at some loci in species that were more distantly genetically related to the species for which the primers were designed (P. notatum and P. atratum). We suggest that this reduction might have been caused by the presence of null alleles or mutations that could have arisen in the primer-binding sites for the SSRs. Homoplasy has been frequently detected among cross-species-amplified SSR markers [96], and it tends to complicate the interpretation of SSR variations. Non-amplifying or null alleles are not commonly found in plant species, but some examples of these phenomena have been observed [97]. The occurrence of null alleles could cause heterozygotes to be confused with homozygotes, which are usually inferred from significant heterozygote deficits. Undetected null alleles in populations can have dramatic effects on the interpretation of genotype frequency distributions and could lead to mistaken interpretations about the level of inbreeding in a population [98]. Therefore, care should be taken when using the studied loci to analyze species that are distant from the Notata and Plicatula groups, as they could lead to incorrect estimates. However, all of the examined loci were generally useful as an initial approach for the evaluation of Paspalum germplasm, especially for species that were more genetically distant from P. atratum and P. notatum.
We observed the presence of exclusive bands between different varieties of P. notatum, which suggests that the use of these microsatellite markers can benefit taxonomic classification and breeding programs. These bands were not present in all the representatives of the same variety but were shared between accessions from the same gene pool. More detailed morphological and molecular characterizations of these groups should be performed to determine whether these characteristics are related to specific cultivars or biotypes, which we could not detect in our work.
Cluster analysis in the Paspalumgenus
Our data support the usefulness of SSRs as a tool for the identification of genetic diversity and global similarity among members of a species. Therefore, we adopted a phenetic approach, which evaluates the genetic similarities between individuals.
All of the analyses identified three main groups (Figures 1, 2 and 3). This trend toward the formation of three main groups could have resulted from the low representative numbers of some species and groups as well as the nature of the markers used. These markers tended to be more conserved in the more genetically distant species of taxa for which the primer sets were originally developed (P. notatum and P. atratum, from the Notata and the Plicatula groups, respectively).
In this study, the species of the Notata group that form the subgenus Paspalum are represented by five taxa (P. pumilum, P. notatum, P. subciliatum, P. cromyorhizon and P. ionanthum). The former two species were clearly distinguishable at the molecular level, whereas P. pumilum, P. notatum, P. subciliatum were grouped together. These results are in accordance with phylogenetic studies based on internal transcribed sequences (ITS) and chloroplast DNA (cpDNA), which support a close genetic relationship between P. pumilum, P. notatum and P. subciliatum[99–101] (these species form a monophyletic clade). Moreover, whether P. cromyorhizon and P. ionanthum should be included in the subgenus Paspalum remains uncertain, but evidence provided by cpDNA sequences [99] supports a close relationship between these two species. P. pumilum, which is a sexual autogamous diploid species, showed less genetic divergence from P. notatum when compared with other species from the group. P. subciliatum exhibited a relatively intermediate degree of genetic divergence from the other species in the Notata group, whereas P. cromyorhizon and P. ionanthum were classified as being somewhat more distant from the other group members (Figures 1, 2 and 3).
In this study, the Plicatula group showed high genetic variability within its species, but there was no clear distinction between different species (Figures 1, 2 and 3). The lack of a clear delineation between the different taxa of the Plicatula group is most likely caused by the genetic closeness of these species as well as the interspecific hybridization that has occurred throughout their evolution, which favors allele sharing. The complexity of the Plicatula was described in a taxonomic review of this group [22]. Many species of the Plicatula group are morphologically polymorphic, including P. atratum, Paspalum plicatulum Michx., Paspalum compressifolium Swallen, and Paspalum limbatum Henrard. Oliveira [22] suggested that the current circumscription adopted for the separation of some species of the Plicatula group could include different taxa and that the pattern of evolution in the group is reticulated, with no well-defined morphological boundaries occurring between taxa. Although the species of this group have not been clearly delineated, the species of the Plicatula group were consistently grouped together in our analyses, with few exceptions. Polyploidy and hybridization are strategies that have played a role in the evolution of the species of the Plicatula group, as in most species of Paspalum, and should be accounted for when defining a species in this group. The species comprising the Plicatula group were considered to be those with populations that were formed by different co-specific cytotypes with at least a basic polyploid series of 2n = 20 or 40 [22].
Few conclusions could be made regarding the third main group, because it was composed of species from the other Paspalum botanical groups that were not included in the first two groups because of the small number of representative species. Previous molecular phylogenetic studies [99] have suggested a greater genetic distance between the third main group compared to the Plicatula and Notata groups, which explains the lower frequency of microsatellite polymorphisms observed for species from the third group. However, there was a clear distinction between the species of this group. Thus, the markers employed in this study have proven to be effective in distinguishing the different Paspalum species and can assist in the identification of distinct species when combined with other molecular techniques.
The accessions from subgenus Anachyris were grouped and presented a close genetic similarity to the species from the Paniculata group (Paspalum juergensii Hack. and Paspalum paniculatum L.) (Figures 2 and 3). Our results corroborate the monophyly of the subgenus Anachyris[99] and the existence of close genetic relationships between Anachyris and Paspalum falcatum Nees ex Steud. (Falcata), Paspalum paucifolium Swallen (Eriantha), Paspalum humboldtianum Flüggé (Subgenus Ceresia), Paspalum quarinii Morrone and Zuloaga (Quadrifaria), P. juergensii and P. paniculatum (Paniculata).
The Virgata and Dilatata groups represent species complexes that have been extensively studied and have been shown to be entirely composed of allopolyploid species based on cytogenetic evidence [27, 102–112]. Paspalum conspersum Schrad., P. dilatatum and Paspalum pauciciliatum (Parodi) Herter exhibit genomic formulae of IIJ2J2, IIJJX and IIJJ, respectively (Additional file 3). The donor species for genome I is Paspalum intermedium Munro ex Morong (Quadrifaria group), and the donor species for genome J is P. juergensii (Paniculata group). Although the Dilatata group has the genomic formulae IIJJ (Additional file 3), the two species from this group included in this study were not genetically close to the Quadrifaria and Paniculata groups, which include the donor species for genomes I and J, respectively.
The classification of Paspalum into two informal groups is controversial. P. conspersum (Virgata) and Paspalum exaltatum J. Presl (Quadrifaria) clustered together in the UPGMA dendrogram (Figure 6) and were shown to be genetically related to Paspalum regnellii Mez (Virgata). The genetic proximity of these species corroborates previous phylogenetic studies [99, 113]. The Virgata group should be considered monophyletic, and a number of additional species, such as P. exaltatum, should be included in it [113]. Moreover, their data supported the idea that P. regnellii should be excluded from the Virgata group and included in the Macrophylla group.
Paspalum maritimum Trin. (Caespitosa) is considered to be allopolyploid, with a predominant bivalent chromosome pairing [114], and this species clustered close to P. conjugatum and Paspalum corcovadense Raddi. P. conjugatum and P. corcovadense were grouped on the basis of the dendrogram and were shown to be genetically distant from the other species studied, corroborating previous phylogenetic studies [99].
Diversity and population structure in Paspalum notatum
Population structure analysis using STRUCTURE software corroborates the most supported groups found in the UPGMA analysis. Group F is the most consistent group throughout all the analyses, and it corresponds to all the accessions from S. Tome (Argentina) (Figures 4, 5 and 6). The K = 7 analysis confirmed the differences among the genotypes that are dispersed throughout southern South America (Additional file 11). The methodology used to analyze the molecular data in this work has been successfully employed for understanding how genetic diversity is organized in other economically important crops [47, 115–117].
Populations of predominantly apomictic grasses are predicted to exhibit low levels of within-population genetic variation due to founder effects and a lack of recombination in offspring, whereas high levels of differentiation among populations are expected because of the limited gene flow via pollen and the divergence of populations over time caused by selection, drift and the accumulation of new mutations [118]. Our work focused on evaluating the relationships between accessions, which could represent distinct populations. Consequently, we expected to detect a high level of differentiation between individuals due to reproductive and geographic isolation. However, a high level of differentiation between individuals was not observed for all the accessions, such as the accessions from gene pools C and D, which are widely distributed but are genetically close. Furthermore, some accessions showed a large amount of genetic variation, especially those derived from Rio Grande do Sul (Brazil) and Argentina. The studied species is widely distributed in the Americas, and Argentina is considered to be its center of origin [119]. The most widely distributed accessions of P. notatum most likely originated from a few apomictic individuals who were better adapted to different environmental conditions, as reported for Pennisetum setaceum, an apomictic grass that has an invasive capacity and can colonize large areas with only a single clone [120].
Moreover, P. notatum comprises sexual diploid and facultative apomictic tetraploid individuals, and diversity could be especially high in the apomictic species complexes that are derived from multiple, independent hybridization events. Gene flow in this species occurs between tetraploid and diploid cytotypes when they are in sympatry [44]; thus, diploid commercial cultivars can introduce variability into an apomictic complex. Furthermore, facultative apomictic tetraploid species can retain residual sexuality such that their occasional hybridization and genetic recombination introduces variability into natural populations. The better-adapted genotypes are then fixed via apomixis, which maintains the maternal genotype and heterozygosity through successive generations [121].
Morphological and molecular results in P. notatum
The great morphological variability of P. notatum in Brazil has been previously described [7, 37], and the morphological and genetic data reported here confirm these findings. Our genetic analyses show a weak correlation with phenotypic data, possibly as a consequence of the small number of morphological characters that we were able to use in this work, combined with the phenotypic plasticity of the morphological characteristics, which were mainly vegetative. Our results show that there is a tendency toward clustering the P. notatum accessions following the botanical variety classifications; however, our data could not confirm the informal intraspecific categories proposed by other authors because the morphological features employed to make these distinctions are continuous and often overlapping or are otherwise incapable of reflecting the total observed variation.
Using spikelet size as a diagnostic characteristic, Döll [41] described the variety latiflorum, which features spikelets that are orbicular-elliptical in shape and larger than the spikelets of the notatum variety. Paspalum notatum var. latiflorum Döll was previously accepted and cited by some authors [36, 42, 43], but this classification is not currently regarded as valid. Parodi [36] studied the variety latiflorum and proposed a new variety, P. notatum var. saurae, which is accepted and used to this day. To differentiate these varieties, Parodi [36] employed spikelet size and the length and number of inflorescence branches, which are diagnostic characteristics that exhibit little or no phenotypic plasticity. The morphological and molecular evidence obtained in this study supports the division of the species into three varieties because there is a strong tendency for accessions of the same variety to remain grouped into similar gene groups. Out of the seven distinct gene pools identified using Bayesian STRUCTURE software analysis, four represent the botanical variety notatum, and three pools correspond to the latiflorum variety.
Sexual diploid strains grow only in a limited area in Argentina, which is considered to be the center of origin for P. notatum[44, 45]. Some tetraploids that share the diploid gene pool might have maintained the genetic patterns of the Argentine diploid populations or might have arisen via independent self-polyploidization events. The diploid accessions (P. notatum var. saurae) were grouped into a single gene pool, which suggests that they share a common origin. These accessions could have derived from the cultivar Pensacola, which originated in Argentina [44, 119], which is in agreement with the existence of gene flow between tetraploid and diploid cytotypes of P. notatum when they are sympatric [44]. However, a group of tetraploid accessions from western Rio Grande do Sul (Brazil) and Uruguay that were botanically identified as P. notatum var. latiflorum share the same gene pool with P. notatum var. saurae.
According to Quarín [24], autotetraploid species of Paspalum might have appeared in a two-step process. Crossbreeding sexual diploids sometimes develop aposporous embryo sacs. Occasionally, an unreduced oosphere is fertilized by the reduced spermatic nuclei of a diploid, forming a triploid (2n, 2× + n = 3×). The triploids then produce offspring that, when fertilized by diploid pollen, give rise to tetraploid (2n, 3× + n = 4×) genotypes.
Geographic distance and genetic dissimilarity in Paspalum notatum
Our data suggest that there is no correlation between genetic dissimilarity and geographic distance among the P. notatum accessions. This arrangement might occur due to an apomixis and polyploidy-induced buffer that restrains the genetic status of the species and promotes the fixation of genetic fitness over a wide range of environments [45]. Moreover, the incongruence between the genetic and geographic data might also be a consequence of anthropogenic dispersal, because P. notatum is a turf and forage grass that is widely sown commercially and can be accidentally dispersed by cattle or other grazing animals. Although no correlation between genetic and geographical data was found in our analysis, a tendency for accessions from the same region to share the same gene pool was evident (Additional file 11).
Conclusions
In summary, our molecular genetic approach using cross-species amplification was proven to be useful for distinguishing the different taxa of Paspalum that were examined, with the exception of those that belong to the complex Plicatula group. The use of a molecular genetics approach that employs microsatellite markers in an initial assessment of germplasms was shown to be useful in species identification and in evaluating the possibility of successful species hybridization.
The methodological approach applied in this study allowed us to understand the genetic organization of different P. notatum genotypes. Comparing the genetic organization with the morphological characteristics provided evidence that supports the existence of three botanical varieties (P. notatum var. notatum, P. notatum var. saurae and P. notatum var. latiflorum[36]), which are represented by different gene groups, with few exceptions. However, the wide genetic variation within each variety deserves more detailed study. We suggest using the methodology that we have proposed in this work to aid in species classification, especially for plants that exhibit wide morphological variation and are difficult to identify. In addition, the microsatellites developed and used throughout this work can be employed for taxonomic classification, breeding programs and DNA fingerprinting for P. notatum cultivar identification.
Abbreviations
- ALIB:
-
Average length of inflorescence branches
- AMOVA:
-
Analysis of molecular variance
- CTAB:
-
Cetyltrimethylammonium bromide
- GS:
-
Genetic similarity
- ISL:
-
Inflorescence stem length
- LBH:
-
Leaf blade hairiness
- LBL:
-
Leaf blade length
- LBW:
-
Leaf blade width
- LSL:
-
Leaf sheath length
- LSW:
-
Leaf sheath width
- PCA:
-
Principal component analysis
- PCO:
-
Principal coordinate analysis
- PCR:
-
Polymerase chain reaction
- PIC:
-
Polymorphism information content
- RS:
-
Rio Grande do Sul state
- SL:
-
Spikelet length
- SP:
-
São Paulo state
- SSR:
-
Simple sequence repeat
- SW:
-
Spikelet width
- UPGMA:
-
Unweighted Pair Group Method with Arithmetic Mean
- USA:
-
United States of America
References
Chase A: The North American species of Paspalum. Contributions from the United States National Herbarium. 1929, 28: 1-310.
Clayton WD, Renvoize SA: Genera graminum: grasses of the world. 1986, London: Her Majesty’s Stationery Office, 1-389.
Zuloaga FO, Morrone O: Revisión de las especies de Paspalum para América del Sur Austral (Argentina, Bolivia, sur del Brasil, Chile, Paraguay y Uruguay). Monogr Syst Bot Mo Bot Gard. 2005, 102: 1-297.
Judziewicz EJ: Flora of the Guianas, 8. Edited by: RA A, Görts-van R. 1990, Königstein: Koltz Scientific Books, 1-727. Family 187. Poaceae (Gramineae), Series A: Phanerogams,
Rua GH: Estudos filogenéticos em Paniceae: os casos de Paspalum e Digitaria. Anais do 57o Congresso Nacional de Botânica Os Avanços da Botânica no início do século XXI morfologia fisiologia taxonomia ecologia e genética. Edited by: Mariath JEA, Santos Porto Alegre RP. 2006, Brasi: Sociedade Botânica do, 170-173.
Valls JFM: Recursos genéticos de espécies de Paspalum no Brasil. Encontro Internacional sobre melhoramento genético de Paspalum. 1987, Brasil: Nova Odessa, 3-13.
Barreto IL: O gênero Paspalum (Gramineae) no Rio Grande do Sul. PhD thesis. 1974, Rio Grande do Su: Universidade Federal
Canto-Dorow TS: Revisão taxonômica das espécies de Paspalum grupo Notata (Poaceae-Paniceae) do Rio Grande do Sul, Brasil. Iheringia. 1996, 47: 3-44.
Morrone O, Carbonó E: Revision del grupo Racemosa del genero Paspalum (Poaceae: Panicoideae: Paniceae). Ann Mo Bot Gard. 1995, 82: 82-116. 10.2307/2399982.
Morrone O, Vega A, Zuloaga FO: Revisión de las especies del género Paspalum (Poaceae: Panicoideae: Paniceae), grupo Dissecta (s. str.). Candollea. 1996, 51: 103-138.
Morrone O, Denham SS, Aliscioni SS, Zuloaga FO: Revisión de las especies de Paspalum (Panicoideae: Paniceae), subgénero Anachyris. Candollea. 2000, 55: 105-135.
Oliveira RC, Valls JFM: Taxonomia de Paspalum L., grupo Linearia (Gramineae-Paniceae) do Brasil. Rev Bras Bot. 2002, 25: 371-389. 10.1590/S0100-84042002012000001.
Denham SS, Zuloaga FO, Morrone O: Systematic revision and phylogeny of Paspalum subgenus Ceresia (Poaceae: Panicoideae: Paniceae). Ann Mo Bot Gard. 2002, 89: 337-399. 10.2307/3298599.
Denham SS: Revisión sistemática del subgénero Harpostachys de Paspalum (Poaceae: Panicoideae: Paniceae). Ann Mo Bot Gard. 2005, 92: 463-532.
Rua GH, Aliscioni SS: A morphology-based cladistic analysis of Paspalum sect. Pectinata (Poaceae). Syst Bot. 2002, 27: 489-501.
Zuloaga FO, Pensiero J, Morrone O: Systematics of Paspalum group Notata (Poaceae- panicoideae- Paniaceae). Syst Bot Monogr. 2004, 71: 1-75.
Burson BL, Bennett HW: Chromosome numbers, microsporogenesis, and mode of reproduction of seven Paspalum species. Crop Sci. 1971, 11: 292-294. 10.2135/cropsci1971.0011183X001100020038x.
Evers GW, Burson BL: Dallisgrass and other Paspalum species. Warm-Season (C 4) Grasses, Agronomy Monographs 45. Edited by: Moser L, Burson BL, Sollenberger LE M. 2004, 681-713. ASA, CSSA, and SSSA
Gates RN, Quarín CL, Gates RN, Quarín CL, Pedreira CGS: Bahiagrass. Warm-Season (C 4) Grasses, Agronomy Monographs 45. Edited by: Moser L, Burson BL, Sollenberger LE M. 2004, 651-680. ASA, CSSA, and SSSA
De Wet JMJ, Prasada Rao KE, Mengesha MH, Brink DE: Diversity in Kodo Millet. Paspalum scrobiculatum. Economic Botany. 1983, 37: 159-163. 10.1007/BF02858779.
Duncan RR, Carrow RN: Seashore Paspalum: the environmental turfgrass. 2000, Hoboken, NJ: J. Wiley & Sons, 281-
Oliveira RC: O gênero Paspalum L., grupo Plicatula (Poaceae: Paniceae), no Brasil. PhD thesis. 2004, Universidade Estadual de Campinas
Pagliarini MS, Carraro LR, Freitas PM, Adamowski EV, Batista LAR, Valls JFM: Cytogenetic characterization of Brazilian Paspalum accessions. Hereditas. 2001, 135: 27-34.
Quarín CL: The nature of apomixis and its origin in Panicoid grasses. Apomixis Newsl. 1992, 5: 8-15.
Burton GW: A cytological study of some species in the genus Paspalum. J Agric Res. 1940, 60: 193-197.
Norrmann GA, Quarín CL, Burson BL: Cytogenetics and reproductive behavior of different chromosome races in six Paspalum species. J Hered. 1989, 80: 24-28.
Honfi AI, Quarin CL, Valls JFM: Karyological studies in South American grasses. Darwiniana. 1990, 30: 87-94.
Norrmann GA, Quarin CL, Killeen TJ: Chromosome numbers in Bolivian grasses (Gramineae). Ann Mo Bot Gard. 1994, 81: 768-10.2307/2399921.
Norrmann GA, Bovo OA, Quarín CL: Post-zygotic seed abortion in sexual diploid × apomictic tetraploid intraspecific Paspalum crosses. Aust J Bot. 1994, 42: 449-10.1071/BT9940449.
Quarín CL, Pozzobon MT, Valls JFM: Cytology and reproductive behavior of diploid, tetraploid and hexaploid germplasm accessions of a wild forage grass: Paspalum compressifolium. Euphytica. 1996, 90: 345-349. 10.1007/BF00027486.
Pozzobon MT, Valls JFM: Chromosome number in Brazilian germplasm accessions of Paspalum hydrophilum, P. modestum and P. palustre (Gramineae; Paniceae). Genet Mol Biol. 2003, 26: 365-36. 10.1590/S1415-47572003000300023.
Pozzobon MT, Machado ACC, Vaio M, Valls JFM, Peñaloza APS, Santos S, Côrtes AL, Rua GH: Cytogenetic analyses in Paspalum L. reveal new diploid species and accessions. Cienc Rural. 2008, 38: 1292-1299. 10.1590/S0103-84782008000500014.
Quarin CL, Espinoza F, Martinez EJ, Pessino SC, Bovo OA: A rise of ploidy level induces the expression of apomixis in Paspalum notatum. Sex Plant Reprod. 2001, 13: 243-249. 10.1007/s004970100070.
Quarín CL, Norrmann GA: Interspecific hybrids between five Paspalum species. Bot Gaz. 1990, 151: 366-369. 10.1086/337837.
Vaio M, Speranza P, Valls JFM, Guerra M, Mazzella C: Localization of the 5S and 45S rDNA sites and cpDNA sequence analysis in species of the Quadrifaria group of Paspalum (Poaceae, Paniceae). Ann Bot. 2005, 96: 191-200. 10.1093/aob/mci168.
Parodi LR: Gramíneas Argentinas nuevas o críticas. I. La variacíon en Paspalum notatum Flügge. Rev Argent Agronomía. 1948, 15: 53-57.
Canto-Dorow TS: Revisão taxonômica das espécies sul-riograndenses de Paspalum L. (grupo Notata) Poaceae - Paniceae, com ênfase na análise da variação intra-específica de Paspalum notatum Flügge. 1993, Universidade Federal do Rio Grande do Sul: Msc dissertation
Burton GW: Bahiagrass types. J Am Soc Agron. 1946, 38: 273-281. 10.2134/agronj1946.00021962003800030008x.
Burton GW: The method of reproduction in common bahiagrass. Paspalum notatum. J Am Soc Agron. 1948, 40: 443-452. 10.2134/agronj1948.00021962004000050008x.
Forbes I, Burton GW: Cytology of diploids, natural and induced tetraploids and intraspecies hybrids of bahiagrass, Paspalum notatum Fluegge. Crop Sci. 1961, 1: 402-406. 10.2135/cropsci1961.0011183X000100060006x.
Döll JC: Gramineae II. Flora brasiliensis. Edited by: Martius CFP, Eichler AW, Monachii F. 1877, Fleischer, 276-296.
Rosengurtt B, Arrelliga BR, Izaguirre P: Gramíneas uruguayas. 1970, Universidad de la República: Montevideo
Lombardo A: Flora montevidensis. 1984, Montivideo: Intendencia Municipal de Montevideo
Daurelio LD, Espinoza F, Quarin CL, Pessino SC: Genetic diversity in sexual diploid and apomictic tetraploid populations of Paspalum notatum situated in sympatry or allopatry. Plant Syst Evol. 2004, 244: 189-199. 10.1007/s00606-003-0070-6.
Espinoza F, Daurelio LD, Pessino SC, Valle EM, Quarin CL: Genetic characterization of Paspalum notatum accessions by AFLP markers. Plant Syst Evol. 2006, 258: 147-159. 10.1007/s00606-005-0401-x.
Speranza PR: Evolutionary patterns in the Dilatata group (Paspalum, Poaceae). Plant Syst Evol. 2009, 282: 43-56. 10.1007/s00606-009-0205-5.
Jungmann L, Vigna BBZ, Boldrini KR, Sousa ACB, Do Valle CB, Resende RMS, Pagliarini MS, Zucchi MI, DeSouza AP: Genetic diversity and population structure analysis of the tropical pasture grass Brachiaria humidicola based on microsatellites, cytogenetics, morphological traits, and geographical origin. Genome. 2010, 53: 698-709. 10.1139/G10-055.
Oliveira RP, Borba EL, Longhi-Wagner HM, Pereira ACS, Lambert SM: Genetic and morphological variability in the Raddia brasiliensis complex (Poaceae: Bambusoideae). Plant Syst Evol. 2008, 274: 25-35. 10.1007/s00606-008-0034-y.
Cidade FW, Dall’Agnol M, Bered F, Souza-Chies TT: Genetic diversity of the complex Paspalum notatum Flügge (Paniceae: Panicoideae). Genet Resour Crop Evol. 2008, 55: 235-246. 10.1007/s10722-007-9231-8.
Pazos-Navarro M, Dabauza M, Correal E, Hanson K, Teakle N, Real D, Nelson MN: Next generation DNA sequencing technology delivers valuable genetic markers for the genomic orphan legume species Bituminaria bituminosa. BMC Genet. 2011, 12: 104-
Santos-Garcia MO, Karia CT, Resende RMS, Chiari L, Vieira MLC, Zucchi MI, Souza AP: Identification of Stylosanthes guianensis varieties using molecular genetic analysis. AoB PLANTS. 2012, pls001-10.1093/aobpla/pls001.
Esselman EJ, Jianqiang L, Crawford DJ, Windus JL, Wolfe AD: Clonal diversity in the rare Calamagrostis porteri ssp. insperata (Poaceae): comparative results for allozymes and random amplified polymorphic DNA (RAPD) and inter simple sequence repeat (ISSR) markers. Mol Ecol. 1999, 8: 443-451. 10.1046/j.1365-294X.1999.00585.x.
Assefa K, Merker A, Tefera H: Inter simple sequence repeat (ISSR) analysis of genetic diversity in tef [Eragrostis tef(Zucc.) Trotter]. Hereditas. 2003, 139: 174-83.
Arnaud-Haond S, Alberto F, Teixeira S, Procaccini G, Serrão EA, Duarte CM: Assessing genetic diversity in clonal organisms: low diversity or low resolution? Combining power and cost efficiency in selecting markers. J Hered. 2005, 96: 434-40. 10.1093/jhered/esi043.
Morgante M, Olivieri AM: PCR-amplified microsatellites as markers in plant genetics. Plant J. 1993, 3: 175-82. 10.1111/j.1365-313X.1993.tb00020.x.
Liu Z, Jarret R, Kresovich S, Duncan RR: Characterization and analysis of simple sequence repeat (SSR) loci in seashore paspalum (Paspalum vaginatum Swartz). Theor Appl Genet. 1995, 91 (1): 47-52.
Speranza P, Malosetti M: Nuclear and cytoplasmic microsatellite markers for the species of the Dilatata group of Paspalum (Poaceae). Plant Genet Resour. 2007, 5: 14-26. 10.1017/S1479262107192145.
Cidade FW, Souza-Chies TT, Batista LAR, Dall’agnol M, Zucchi MI, Jungmann L, Souza AP: Isolation and characterization of microsatellite loci in Paspalum notatum Flüggé (Poaceae). Conserv Genet. 2009, 10: 1977-1980. 10.1007/s10592-009-9872-6.
Cidade FW, Souza-Chies TT, Souza FHD, Batista LAR, Dall’Agnol M, Valls JFM, Zucchi MI, Souza AP: Microsatellite loci for Paspalum atratum (Poaceae) and cross-amplification in other species. Am J Bot. 2010, 97: e107-e110. 10.3732/ajb.1000207.
Byrne M, Marquez-Garcia MI, Uren T, Smith DS, Moran GF: Conservation and genetic diversity of microsatellite loci in the genus Eucalyptus. Aust J Bot. 1996, 44: 331-341. 10.1071/BT9960331.
Steinkellner H, Lexer C, Turetschek E, Glossl J: Conservation of (GA)n microsatellite loci between Quercus species. Mol Ecol. 1997, 6: 1189-1194. 10.1046/j.1365-294X.1997.00288.x.
Peakall R, Gilmore S, Keys W, Morgante M, Rafalski A: Cross-species amplification of soybean (Glycine max) simple sequence repeats (SSRs) within the genus and other legume genera: implications for the transferability of SSRs in plants. Mol Biol Evol. 1998, 15: 1275-87. 10.1093/oxfordjournals.molbev.a025856.
Roa AC, Chavarriaga-Aguirre P, Duque MC, Maya MM, Bonierbale MW, Iglesias C, Tohme J: Cross-species amplification of cassava (Manihot esculenta)(Euphorbiaceae) microsatellites: allelic polymorphism and degree of relationship. Am J Bot. 2000, 87: 1647-1655. 10.2307/2656741.
Dirlewanger E, Cosson P, Tavaud M, Aranzana J, Poizat C, Zanetto A, Arús P, Laigret F: Development of microsatellite markers in peach [Prunus persica (L.) Batsch] and their use in genetic diversity analysis in peach and sweet cherry (Prunus avium L.). Theor Appl Genet. 2002, 105: 127-138. 10.1007/s00122-002-0867-7.
Hempel K, Peakall R: Cross-species amplification from crop soybean Glycine max provides informative microsatellite markers for the study of inbreeding wild relatives. Genome. 2003, 46: 382-93. 10.1139/g03-013.
González-Martínez SC, Robledo-Arnuncio JJ, Collada C, Díaz A, Williams CG, Alía R, Cervera MT: Cross-amplification and sequence variation of microsatellite loci in Eurasian hard pines. Theor Appl Genet. 2004, 109: 103-11. 10.1007/s00122-004-1596-x.
Poncet V, Hamon P, Minier J, Carasco C, Hamon S, Noirot M: SSR cross-amplification and variation within coffee trees (Coffea spp.). Genome. 2004, 47: 1071-81. 10.1139/g04-064.
Doyle JJ, Doyle JL: A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull. 1987, 19: 11-15.
Billotte N, Lagoda PJL, Risterucci AM, Baurens FC: Microsatellite-enriched libraries: applied methodology for the development of SSR markers in tropical crops. Fruits. 1999, 54: 277-288.
Don RH, Cox PT, Wainwright BJ, Baker K, Mattick JS: “Touchdown” PCR to circumvent spurious priming during gene amplification. Nucleic Acids Res. 1991, 19 (14): 4008-10.1093/nar/19.14.4008.
Wu KS, Tanksley SD: Abundance, polymorphism and genetic mapping of microsatellites in rice. Mol Gen Genet. 1993, 241–241: 225-235.
Cordeiro GM, Pan Y, Henry RJ: Sugarcane microsatellites for the assessment of genetic diversity in sugarcane germplasm. Plant Sci. 2003, 165: 181-189. 10.1016/S0168-9452(03)00157-2.
Dice LR: Measures of the amount of ecologic association between species. Ecology. 1945, 26: 297-302. 10.2307/1932409.
Jaccard P: Nouvelles recherches sur la distribution florale. Bull Soc Vaudoise Sci Nat. 1908, 44: 223-270.
Rohlf FJ: NTSYS-pc: numerical taxonomy and multivariate analysis system, version 2.1. 2000, New York: Exeter Software
Sneath PHA, Sokal RR: Numerical taxonomy. The principles and practice of numerical classification. 1973, San Francisco: Freeman, 573-
Mantel N: The detection of disease clustering and a generalized regression approach. Cancer Res. 1967, 27: 209-220.
Perrier X, Jacquemoud-Collet JP: DARwin software. Computer program and documentation distributed by the author. 2006, http://darwin.cirad.fr/darwin/Home.php,
Rambaut A: FigTree. 2009, ver. 1.3.1
Coelho ASG: BOOD-Avaliação de dendrogramas baseados em estimativas de distâncias/similaridades genéticas através do procedimento de bootstrap, Versão 3.0. 2001, UFG: Laboratório Genética Vegetal, DBG/ICB
Pritchard JK, Stephens M, Donnelly P: Inference of population structure using multilocus genotype data. Genetics. 2000, 155: 945-59.
Falush D, Stephens M, Pritchard JK: Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics. 2003, 164: 1567-87.
Falush D, Stephens M, Pritchard JK: Inference of population structure using multilocus genotype data: dominant markers and null alleles. Mol Ecol Notes. 2007, 7: 574-578. 10.1111/j.1471-8286.2007.01758.x.
Evanno G, Regnaut S, Goudet J: Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol. 2005, 14: 2611-20. 10.1111/j.1365-294X.2005.02553.x.
Earl DA, VonHoldt BM: STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour. 2011, online first
Excoffier L, Laval G, Schneider S: Arlequin (version 3.0): An integrated software package for population genetics data analysis. Evol. Bioinformatics. 2005, 1: 47-50.
Cidade FW: Análise da variabilidade genética de Paspalum notatum Flügge (Poaceae, Panicoideae) com o uso de marcadores moleculares, morfológicos e citometria de fluxo. 1974, Universidade Federal do Rio Grande do Sul: Msc dissertation
Cruz CD: Programa Genes - versão Windows: aplicativo computacional em genética e estatística. [http://www.ufv.br/dbg/genes/genes.htm]
STATISTICA software, version 7.0. [http://www.statsoft.com/support/download]
Ersts PJ: Geographic Distance Matrix Generator, version 1.2.3. [http://biodiversityinformatics.amnh.org/open_source/gdmg]
Hijmans R, Guarino L: Computer tools for spatial analysis of plant genetic resources data: 1. DIVA-GIS. Plant Genet Resour Newsl. 2001, 127: 15-19.
Huang Q, Börner A, Röder S, Ganal W: Assessing genetic diversity of wheat (Triticum aestivum L.) germplasm using microsatellite markers. Theor Appl Genet. 2002, 105: 699-707. 10.1007/s00122-002-0959-4.
Nei M: Analysis of gene diversity in subdivided populations. Proc Nat Acad Sci USA. 1973, 70: 3321-3. 10.1073/pnas.70.12.3321.
Testolin R, Marrazzo T, Cipriani G, Quarta R, Verde I, Dettori MT, Pancaldi M, Sansavini S: Microsatellite DNA in peach (Prunus persica L. Batsch) and its use in fingerprinting and testing the genetic origin of cultivars. Genome. 2000, 43: 512-20.
Primmer CR, Møller AP, Ellegren H: A wide-range survey of cross-species microsatellite amplification in birds. Mol Ecol. 1996, 5: 365-378.
Park YJ, Lee JK, Kim NS: Simple sequence repeat polymorphisms (SSRPs) for evaluation of molecular diversity and germplasm classification of minor crops. Molecules. 2009, 14: 4546-69. 10.3390/molecules14114546.
Sousa SN, Finkeldey R, Gailing O: Experimental verification of microsatellite null alleles in Norway Spruce (Picea abies [L.] Karst.): implications for population genetic studies. Plant Mol Biol. 2005, 23: 113-119. 10.1007/BF02772701.
Pemberton JM, Slate J, Bancroft DR, Barrett JA: Nonamplifying alleles at microsatellite loci: a caution for parentage and population studies. Mol Ecol. 1995, 4: 249-252. 10.1111/j.1365-294X.1995.tb00214.x.
Rua GH, Speranza PR, Vaio M, Arakaki M: A phylogenetic analysis of the genus Paspalum (Poaceae) based on cpDNA and morphology. Plant Syst Evol. 2010, 288: 227-243. 10.1007/s00606-010-0327-9.
Souza-Chies TT, Essi L, Rua GH, Valls JFM, Miz RB: A preliminary approach to the phylogeny of the genus Paspalum (Poaceae). Genetica. 2006, 126: 15-32. 10.1007/s10709-005-1428-1.
Essi L, Souza-Chies TT: Phylogeny of Linearia and Notata groups of Paspalum L. (Poaceae, Panicoideae, Paniceae) and related species. Genet Resour Crop Evol. 2006, 54: 779-791.
Burson BL, Lee H, Bennett HW: Genome relations between tetraploid Paspalum dilatatum and four diploid Paspalum species. Crop Sci. 1973, 13: 739-743. 10.2135/cropsci1973.0011183X001300060045x.
Burson BL: Genome relations between Paspalum conspersum and two diploid Paspalum species. Can J Genet Cytol. 1978, 20: 365-372.
Burson BL: Phylogenetic investigations of Paspalum dilatatum and related species. Proceedings of the 14 th International Grassland Congress. Edited by: Smith JA, Hays VW B. 1983, Westview Press
Burson BL: Cytogenetics of Paspalum urvillei X P. intermedium and P. dilatatum X P. paniculatum hybrids. Crop Sci. 1979, 19: 534-538. 10.2135/cropsci1979.0011183X001900040025x.
Burson BL: Homology of chromosomes of the X genomes in common and Uruguayan dallisgrass, Paspalum dilatatum. Genome. 1991, 34: 950-953. 10.1139/g91-147.
Burson BL: Genome relationships between tetraploid and hexaploid biotypes of dallisgrass, Paspalum dilatatum. Bot Gaz. 1991, 152a: 219-223.
Burson BL: Genome relationship and reproductive behavior of intraspecific Paspalum dilatatum hybrids: Yellow-Anthered x Uruguaiana. Int J Plant Sci. 1995, 156: 326-331. 10.1086/297254.
Burson BL, Bennett HW: Cytogenetics of Paspalum conspersum and its genomic relationship with yellow-anthered P. dilatatum and P. malacophyllum. Can J Genet Cytol. 1976, 18: 701-708.
Burson BL, Quarín CL: Cytology of Paspalum virgatum and its relationship with P. intermedium and P. jurgensii. Can J Genet Cytol. 1982, 24: 219-226.
Caponio I, Quarín CL: Intra-and interspecific hybridization between dallisgrass and vaseygrass. Crop Sci. 1990, 30: 362-364. 10.2135/cropsci1990.0011183X003000020023x.
Quarín CL, Caponio I: Cytogenetics and reproduction of Paspalum dasypleurum and its hybrids with P. urvillei and P. dilatatum ssp. flavescens. Int J Plant Sci. 1995, 156: 232-235. 10.1086/297245.
Giussani LM, Zuloaga FO, Quarín CL, Cota-Sánchez JH, Ubayasena K, Morrone O: Phylogenetic relationships in the genus Paspalum (Poaceae: Panicoideae: Paniceae): an assessment of the Quadrifaria and Virgata informal groups. Syst Bot. 2009, 34: 32-43. 10.1600/036364409787602258.
Adamowski EV, Pagliarini MS, Batista LAR: Chromosome number and microsporogenesis in Paspalum maritimum (Caespitosa group; gramineae). Braz Arch Biol Technol. 2000, 43: 301-305. 10.1590/S1516-89132000000300009.
Liu K, Goodman M, Muse S, Smith JS, Buckler E, Doebley J: Genetic structure and diversity among maize inbred lines as inferred from DNA microsatellites. Genetics. 2003, 165: 2117-28.
Barkley NA, Roose ML, Krueger RR, Federici CT: Assessing genetic diversity and population structure in a citrus germplasm collection utilizing simple sequence repeat markers (SSRs). Theor Appl Genet. 2006, 112: 1519-31. 10.1007/s00122-006-0255-9.
Kwak M, Gepts P: Structure of genetic diversity in the two major gene pools of common bean (Phaseolus vulgaris L., Fabaceae). Theor Appl Genet. 2009, 118: 979-92. 10.1007/s00122-008-0955-4.
Carino DA, Daehler CC: Genetic variation in an apomictic grass, Heteropogon contortus, in the Hawaiian Islands. Mol Ecol. 1999, 8: 2127-2132. 10.1046/j.1365-294x.1999.00786.x.
Burton GW: A search for the origin of Pensacola Bahia grass. Econ Bot. 1967, 21: 379-382. 10.1007/BF02863165.
Poulin J, Weller SG, Sakai AK: Genetic diversity does not affect the invasiveness of fountain grass (Pennisetum setaceum) in Arizona, California and Hawaii. Divers Distrib. 2005, 11: 241-247. 10.1111/j.1366-9516.2005.00136.x.
Kellogg EA: Variation and species limits in agamospermous grasses. Syst Bot. 1990, 15: 112-123. 10.2307/2419021.
Pagliarini MS, Takayama SY, Freitas PM, Carraro LR, Adamowski EV, Silva N, Batista LAR: Failure of cytokinesis and 2n gamete formation in Brazilian accessions of Paspalum. Euphytica. 1999, 108: 129-135. 10.1023/A:1003660327223.
Adamowski EV, Pagliarini MS, Bonato ABM, Batista LAR, Valls JFM: Chromosome numbers and meiotic behavior of some Paspalum accessions. Genet Mol Biol. 2005, 28: 773-780. 10.1590/S1415-47572005000500020.
Takayama SY, Freitas PM, Pagliarini MS, Batista LAR: Chromosome number in germplasm accessions of Paspalum (Plicatula group) from different regions in Brazil. Euphytica. 1998, 99: 89-94. 10.1023/A:1018336200053.
Pozzobon MT, Valls JFM: Cytogeography and variation of stomatal size of Paspalum glaucescens (Gramineae; Paniceae) in Southern Brazil. Euphytica. 2000, 116: 251-256. 10.1023/A:1004005230300.
Dahmer N, Schifino-Wittmann MT, Dall’Agnol M, Castro B: Cytogenetic data for Paspalum notatum Flüggé accessions. Sci Agric. 2008, 65: 381-388. 10.1590/S0103-90162008000400009.
Moraes-Fernandes MIB, Barreto IL, Salzano FM, Sacchet AMOF: Cytological and evolutionary relationships in Brazilian forms of Paspalum (Gramineae). Caryologia. 1974, 27 (4): 455-464.
Quarín CL: Híbridos interespecíficos de Paspalum notatum x P. modestum. Bonplandia. 1983, V: 235-242.
Quarín CL: Recuentos cromosómicos en gramíneas de Argentina Subtropical. Hickenia. 1977, 1: 73-78.
Mehra PN, Chaudhary JD: Male meiosis in some grasses of the tribe Paniceae from North Eastern India. I. Genus Paspalum. Cytologia. 1981, 46: 265-278.
Soster M: Caracterização morfológica e citogenética de acessos de Paspalum coletados no sul do Brasil. 2009, Universidade Federal de Santa Catarina: PhD thesis
Bashaw EC, Forbes JR: chromosome numbers and microsporogenesis in Dallisgrass Paspalum dilatatum Poir1. Agron J. 1958, 50: 441-445. 10.2134/agronj1958.00021962005000080006x.
Bashaw EC, Hovin AW, Hold EC: Apomixis, its evolutionary significance and ulitization in plant breeding. Proceedings of the XI International Grassland Congress. Edited by: Norman MJT. 1970, Queensland, Australia: St. Lucia, 245-242.
Burson BL: Cytogenetic relationships between Paspalum jurgensii and P. intermedium, P. vaginatum, and P. setaceum var. ciliatifolium. Crop Sci. 1981, 21: 515-519. 10.2135/cropsci1981.0011183X002100040009x.
Hojsgaard D, Honfi AI, Rua G, Daviña J: Chromosome numbers and ploidy levels of Paspalum species from subtropical South America (Poaceae). Genet Resour Crop Evol. 2009, 56: 533-545. 10.1007/s10722-008-9384-0.
Pozzobon M: Chromosome counts in Brazilian species of Paspalum L. (Gramineae). Acta Bot Bras. 2000, 14: 151-162.
Gould FW: Chromosome numbers in southwestern grasses. Am J Bot. 1958, 45: 757-767. 10.2307/2439737.
Gould FW: Chromosome numbers of Texas grasses. Can J Bot. 1968, 46: 1315-1325. 10.1139/b68-175.
Hojsgaard D, Schegg E, Valls JFM, Martínez E, Quarín CL: Sexuality, apomixis, ploidy levels, and genomic relationships among four Paspalum species of the subgenus Anachyris (Poaceae). Flora. 2008, 203: 535-547. 10.1016/j.flora.2007.09.005.
Burson BL, Hussey MA: Cytology of Paspalum malacophyllum and its relationship to P. juergensii and P. dilatatum. Int J Plant Sci. 1998, 159: 153-159. 10.1086/297533.
Quarín CL, Hanna WW: Chromosome behavior, embryo sac development, and fertility of Paspalum modestum, P. boscianum, and P. conspersum. J Hered. 1980, 71: 419-422.
Quarín CL, Burson BL: Cytology of sexual and apomictic Paspalum species. Cytologia. 1991, 56: 223-228. 10.1508/cytologia.56.223.
Martínez EJ, Quarín CL: Citoembriologia y comportamiento reproductivo de un citotipo diploide de Paspalum hydrophilum y sus hibridos con P. palustre (Poaceae, Paniceae). Darwiniana. 1999, 37: 223-228.
Burson BL, Bennett HW: Meiotic and reproductive behavior of some introduced Paspalum species. J Miss Acad Sci. 1971, 17: 5-8.
Pozzobon MT, Valls JFM: Caracterização citogenética em acessos de germoplasma de espécies brasileiras de Paspalum (Gramineae). Encontro Internacional sobre melhoramento genético de Paspalum. 1987, Nova Odessa, 73-78.
Burton GW: The method of reproduction in common Bahia grass, Paspalum notatum. Journal of the American Society of Agronomy. 1948, 40: 443-452. 10.2134/agronj1948.00021962004000050008x.
Burson BL: Genome relations among four diploid Paspalum species. Bot Gaz. 1981, 142: 592-596. 10.1086/337261.
Burton GW: Breeding Pensacola bahiagrass, Paspalum notatum: I. Method of reproduction. Agron J. 1955, 47: 311-314.
Quarín CL, Burson BL: Cytogenetic relations among Paspalum notatum var. saurae, P. pumilum, P. indecorum, and P. vaginatum. Bot Gaz. 1983, 144: 433-438. 10.1086/337394.
Quarín CL, Hanna WW, Fernández A: Genetic studies in diploid and tetraploid Paspalum species. J Hered. 1982, 72: 254-256.
Quarín CL, Burson BL, Burton GW: Cytology of intra- and interspecific hybrids between two cytotypes of Paspalum notatum and P. cromyorrhizon. Bot Gaz. 1984, 145: 420-426. 10.1086/337474.
Quarin CL, Norrmann GA: Cytology and reproductive behavior of Paspalum equitans, P. ionanthum, and their hybrids with diploid and tetraploid cytotypes of P. cromyorrhizon. Bot Gaz. 1987, 148: 386-391. 10.1086/337667.
Burson BL, Bennett HW: Cytology and reproduction of three Paspalum species. J Hered. 1970, 61: 129-132.
Sede S, Escobar A, Morrone O, Zuloaga FO: Chromosome studies in American Paniceae (Poaceae, Panicoideae) 1. Ann Mo Bot Gard. 2010, 97: 128-138. 10.3417/2007118.
Quarín CL, Valls JFM, Urbani MH: Cytological and reproductive behaviour of Paspalum atratum, a promising forage grass for the tropics. Trop Grassl. 1997, 31: 114-116.
Rodrigues LG, Santos S, Oliveira RC, Peñaloza APS: Contagens cromossômicas em espécies brasileiras de Paspalum (Gramineae). Anais do III Simpósio de Recursos Genéticos para a América Latina e Caribe. 2001, Londrina, 395-397.
Burton GW: A cytological study of some species in the tribe Paniceae. Am J Bot. 1942, 29: 355-360. 10.2307/2437217.
Acknowledgments
We thank the Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) for financial support (Project No. 05/51010-0) and a graduate fellowship (2006/61242-8) awarded to F. W. Cidade. We also thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for financial support and a research fellowship to A. P. Souza.
Author information
Authors and Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
FWC performed experimental and statistical analyses and drafted the manuscript. MIZ participated in the statistical analysis. FHDS, JFMV and MD participated in the germplasm selection and the design and implementation of the study. APS and TTSC conceived the study and participated in its design and coordination. APS and BBZV helped draft the manuscript. All authors read and approved the final manuscript.
Electronic supplementary material
12863_2012_1118_MOESM1_ESM.xlsx
Additional file 1: Accessions from the Paspalum collection used in the whole germplasm analysis. Accessions from the Paspalum collection used in the whole germplasm analysis. The ID, BRA code, species, botanical groups, origin, chromosome numbers and the reference for the chromosome number determination are shown [23, 31, 32, 114, 122–126]. (XLSX 25 KB)
12863_2012_1118_MOESM2_ESM.docx
Additional file 2: Paspalum notatum accessions evaluated in this study. Paspalum notatum accessions evaluated in this study. The CODE used in the figures, ID, collector identification, BRA CODE, site of origin, chromosome number, ploidy level, geographic coordinates and availability of morphological data are shown. (DOCX 25 KB)
12863_2012_1118_MOESM3_ESM.xlsx
Additional file 3: Chromosome number, polyploidy type, reproduction mode and genomes found in the Paspalum species. Chromosome number, type of polyploidy, reproduction mode and genomes found in the Paspalum species based on the literature [17, 27, 30–32, 102–104, 106–108, 110, 113, 114],[122, 124, 125, 127–157]. (XLSX 14 KB)
12863_2012_1118_MOESM4_ESM.xlsx
Additional file 4: Size range, number of bands and PIC from the 12 SSR loci transferred to the Paspalum taxa. Size range, number of bands and PIC from the 12 SSR loci transferred to the Paspalum taxa. (XLSX 46 KB)
12863_2012_1118_MOESM5_ESM.pdf
Additional file 5: Magnitude of Δ K from STRUCTURE analysis of the germplasm. Magnitude of ΔK from STRUCTURE analysis of K (arithmetic mean ± s.d. over 10 replicates) calculated following the ΔK method proposed by Evanno et al. [84] for Paspalum microsatellite data. The modal values of these distributions indicate that the true K, or the uppermost level of the STRUCTURE analysis, is seven genetic groups. (PDF 13 KB)
12863_2012_1118_MOESM6_ESM.xlsx
Additional file 6: Magnitude of Δ K from the STRUCTURE analysis of the Paspalum notatum accessions. Magnitude of ΔK from the STRUCTURE analysis of K (mean ± SD over 8 replicates), calculated following the ΔK method proposed by Evanno et al. [84] for Paspalum notatum microsatellite data. (XLSX 17 KB)
12863_2012_1118_MOESM7_ESM.docx
Additional file 7: Mean probabilities for each population from the STRUCTURE analysis for K = 7 for Paspalum notatum. Mean probabilities for each population from the STRUCTURE analysis for K = 7 for Paspalum notatum. (DOCX 12 KB)
12863_2012_1118_MOESM8_ESM.xlsx
Additional file 8: Statistics describing nine morphological traits from 46 Paspalum notatum accessions. Statistics describing nine morphological traits from 46 Paspalum notatum accessions. (XLSX 10 KB)
12863_2012_1118_MOESM9_ESM.xlsx
Additional file 9: Principal components of nine morphological traits from 46 Paspalum notatum accessions. Principal components of nine morphological traits from 46 Paspalum notatum accessions. (XLSX 9 KB)
12863_2012_1118_MOESM10_ESM.xlsx
Additional file 10: Botanical identification of Paspalum notatum accessions according to different authors and the corresponding STRUCTURE group. Botanical identification of Paspalum notatum accessions according to different authors and the corresponding STRUCTURE group. (XLSX 13 KB)
12863_2012_1118_MOESM11_ESM.tiff
Additional file 11: Map of the geographic distribution of 57 of the Paspalum notatum accessions analyzed. Geographic distribution of the 57 Paspalum notatum accessions that were analyzed genotypically and phenotypically. Point colors correspond to the colors in the STRUCTURE clusters from Figure 5. A sole accession from Florida, USA, has not been represented in the map. (TIFF 342 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open Access This article is published under license to BioMed Central Ltd. This is an Open Access article is distributed under the terms of the Creative Commons Attribution License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Cidade, F.W., Vigna, B.B., de Souza, F.H. et al. Genetic variation in polyploid forage grass: Assessing the molecular genetic variability in the Paspalumgenus. BMC Genet 14, 50 (2013). https://doi.org/10.1186/1471-2156-14-50
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/1471-2156-14-50