Abstract
Ceramic materials are sometimes used as bearing surfaces of joint implants due to their high wear resistance and biocompatibility. Silicon nitride (Si3N4) is one of the ceramics under investigation for such use, owing to its bacteriostatic properties and high wear resistance. Traditional Si3N4 is sintered using Al2O3 and Y2O3 as sintering aids. To improve the biocompatibility and bioactivity of Si3N4, new sintering aids (SrO, MgO, and SiO2) were used in this work. This substitution may however have substantial effects on the wear properties of the material. Hence, the aim of this study was to evaluate these effects. Multidirectional pin-on-disc wear tests against ultra-high molecular weight polyethylene pins were used to this end, running in fetal bovine serum solution at 37 °C for 2 million cycles. The surface roughness, phase composition, and surface morphology of the surfaces were investigated, together with the pH of the wear test lubricant and compared to a traditional Si3N4 composition. XRD and SEM results showed that the prepared ceramics were composed of α- and β-phase Si3N4. The surface roughness of the Si3N4 ceramic discs was in accordance with the biomedical standard, and the wear test results demonstrated that the new ceramics had a low wear factor and a comparable coefficient of friction to Si3N4 ceramics reported in previous work. However, the dissolution of the materials over time may be a concern for biotribological applications, such as long-term use or in other wear couples.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Silicon nitride (Si3N4) is a technical ceramic that has been widely used in many industrial applications, such as turbine blades, bearings, cutting tools, and spark plugs [1, 2]. In addition to excellent mechanical properties, Si3N4 ceramics have also been reported to be biocompatible and bacteriostatic [3, 4]. Owing to the above advantages of Si3N4, it has been utilized as spinal fusion implants [5]. In addition, Si3N4-based materials have been considered to be used as total hip replacements (THR) and total knee replacements (TKR) both in bulk form as well as coatings, since Si3N4-based materials typically show high wear resistance [3, 5, 6].
Owing to their good wear properties, zirconia (ZrO2) and alumina (Al2O3) have been the main ceramic bulk materials considered for joint bearings, but both of them still have some disadvantages [7]. Yttria-stabilized zirconia (YSZ) exhibits tetragonal to monoclinic phase transformation when exposed to the in vivo environment, sometimes leading to implant failure [8, 9]. Zirconia Toughened Alumina (ZTA) ceramics have shown improvements compared to pure Al2O3 ceramics, e.g., higher fracture strength. However, neither ZrO2-based ceramics nor Al2O3 ceramics have shown bacteriostatic properties as Si3N4 has demonstrated [10,11,12]. Another advantage of Si3N4 over ZrO2 or Al2O3 in terms of use as an implant is that wear debris from Si3N4 can dissolve slowly in aqueous solutions [13], forming biocompatible ions, which potentially reduces any negative effect the wear debris may have on the surrounding tissues.
Sintering additives are needed to assist the densification of Si3N4. Before sintering, Si3N4 raw powder is mixed with a small amount of sintering additives, typically Y2O3 and Al2O3, which aid sintering and densification via formation of a liquid phase [4, 14]. Upon solidification, the additives form an intergranular amorphous phase or slightly crystallized phase at the grain boundaries [15, 16]. An alternative way of achieving dense Si3N4 may be through the use of more biocompatible and bioactive sintering additives, such as SrO, MgO, and SiO2 [4, 17]. These sintering additives have the potential to improve the bioactivity of the Si3N4. For example, it has been found that Sr could trigger osteoblast formation and inhibit osteoclasts, potentially leading to increased bone formation [18]. Researchers have also reported that Sr and Mg could improve osteogenesis and vasculogenesis [17, 19] and that Si may increase bone mineralization [20]. In an earlier study by the authors, a new Si3N4-based bioceramic was developed by adding 10% and 18% of SrO, MgO, and SiO2 as sintering additives [19, 21]. Bioactivity over time was confirmed through exposure to a phosphate buffer solution, resulting in a nanotopographic surface which could be potentially beneficial for osseointegration and cell adhesion [22,23,24,25,26]. Meanwhile, indirect cell studies with MC3T3-E1 (osteoblast) cells showed no toxicity [19, 21].
It is well known that wear properties play a significant role when Si3N4-based bioceramics are to be used as implants for total hip replacements (THR) and total knee replacements (TKR). However, the wear properties of the newly-developed Si3N4 bioceramics have not been evaluated. Hence, the aim of this work was to evaluate the wear performance of this new Si3N4 bioceramic. The Si3N4 of this study was densified using a spark plasma sintering (SPS) instrument with SrO, MgO, and SiO2 as sintering additives. Surface roughness and morphology were characterized by optical profilometry and scanning electron microscopy (SEM), respectively. The phase composition was analyzed by X-ray diffraction (XRD) measurements. A multidirectional wear test was used for wear resistance tests.
Materials and Methods
Spark Plasma Sintering
The starting raw materials, including Si3N4 powder (β phase, with an average particle size of approximately 20 μm), SrO, MgO, and SiO2 (as sintering aids), were mixed for 3 h in a polyethylene jar using Si3N4 balls and ethanol (> 99%) as a mixing medium. Two compositions were manufactured, i.e., 2%SiO2–4%MgO–4%SrO–90%Si3N4 (wt%) and 2%SiO2–8%MgO–8%SrO–82%Si3N4 (wt%). These materials are hereinafter denoted as SN10 and SN18, respectively. According to our previous studies, Si3N4 sintered with 10-wt% and 18-wt% glass phase showed significant differences in terms of densification behavior, mechanical properties, phase composition, and ion release. Hence, in this study, we choose to move forward with both 10-wt% and 18-wt% glass phase to explore their effects on the wear behavior of the Si3N4. Si3N4 discs were sintered using spark plasma sintering (SPS-825, Fuji Electronic Industrial Co., Ltd, Japan). A traditional Si3N4 ceramic with a composition of 4%Al2O3–6%Y2O3–90%Si3N4 (wt%) was also manufactured as a control and was denoted as SNC. The detailed sintering process has been described elsewhere [19, 21], but it is reproduced herein for clarity. The SN10, SN18, and SNC samples were sintered with slightly different sintering parameters, as follows: a heating rate of 100 °C/min from room temperature to the sintering temperature, an applied pressure of 60 MPa for the three groups, and a dwelling time of 3 min at the sintering temperature, then a sintering temperature of 1680 °C for the SN10 group and SN18 group, and a sintering temperature of 1700 °C for SNC group. All samples were sintered in a N2 atmosphere. All the sintered samples were prepared as discs (Ø20mm x 2 mm). The sintered samples were wet ground and polished on an automatic polishing machine to achieve smooth surfaces. The average relative density of the SN10 group and the SN18 group was approximately 92.2% and 91.4% [19], respectively, while the relative density of the control group was 93.7%.
Multidirectional Wear Tests (MWT)
The wear behavior of the prepared Si3N4 against ultra-high molecular weight polyethylene (UHMWPE) was assessed using cylindrical UHMWPE pins with a nominal length of 19.1 mm and a diameter of 9.5 mm. The pins were made of UHMWPE GUR1020 (Peter Brehm GmbH, Weisendorf, Germany). Multidirectional wear tests (MWTs) were performed in 0.2-μm filtered fetal bovine serum solution (25%) at 37 °C. Prior to the test, the pins were pre-soaked in serum and cleaned according to the standard [27]. According to ASTM F732-17 [27], wear testing was carried out with a nominal load of 213 N, resulting in an estimated contact pressure of 3 MPa. A frequency of 2 Hz and a sliding velocity 40 mm/s were used for 2,000,000 cycles (2.0 MC), using a 5-mm square path for a sliding distance of 20 mm/cycle. To evaluate the wear of the UHMWPE pins, a gravimetric method was used [28]. The volumetric wear was calculated to obtain the wear factor (k), which is the volumetric wear divided by the applied load and multiplied by the sliding distance [28].
Material Characterizations
Phase identification was carried out by XRD (Diffractometer D5000, Siemens, Germany). XRD patterns were recorded in the range of 5°–90° with a scan step of 0.2 s/step and a step size of 0.012° using a Ni-filtered Cu Kα X-ray resource (40 kV and 40 mA). The average surface roughness was measured before and after wear testing using a Nexview™ NX2 3D Optical Surface Profiler, at 10X and a field of view (FOV) of 2.0, corresponding to an area of 500 × 500 μm2. Five measurements were performed on each sample to obtain Sa (arithmetical mean height). Morphological characterization was carried out by scanning electron microscopy (SEM, Zeiss Merlin with AZtec EDS/EBSD, Oberkochen, Germany) before and after wear tests using secondary electrons (SE) at 20 kV. The studied samples were sputtered with Ag/Pd coating for 40 s to avoid charging effects. Before observation, the samples were etched with 5% HF solution for 60 min for grain exposure. After every 0.5 MC, the pH of the lubricant solution was measured at room temperature (around 21.5 °C) using a pH meter (METTLER TOLEDO, Ohio, USA).
Statistical Analysis
IBM SPSS Statistics v 22 was used for all statistical analyses. A repeated measures ANOVA was used to determine statistical differences in roughness, coefficient of friction, and wear factor as a function of material and time point. A critical level of α = 0.05 was used to determine significance.
Results and Discussion
Phase Identification
The phase composition of the materials is shown in Fig. 1. It was observed that all three groups were composed of α-Si3N4 and β-Si3N4. For the samples sintered with SrO, MgO, and SiO2, no other crystalline phases containing Sr, Mg, or Si were detected, indicating that these additives (SrO, MgO, and SiO2) were amorphous after sintering. Even for the SN18 sample where the content of sintering additives was 18 wt%, the additives (SrO, MgO, and SiO2) did not crystallize, neither did they react with each other to form other compound. For the reference sample (SNC), no crystalline phases containing Al or Y were detected either (Fig. 1). Hence, these additives (Al2O3 and SiO2) were also amorphous after sintering. The sintering additives did not alter the crystallization behavior of the Si3N4.
Coefficient of Friction
The coefficient of friction of the studied Si3N4 is presented in Fig. 2, from which it can be seen that in all tests it ranged between 0.06 and 0.10 (Fig. 2), which is comparable to previous studies on Si3N4 bulk material and coatings [29, 30]. Previous work has found that a tribofilm forms on the surface of Si3N4 in aqueous environments during wear tests [31,32,33]. This tribofilm is a thin layer consisting of SiO2 and Si(OH)2, and this layer can improve the wear resistance of Si3N4 by acting as a self-lubricating layer and reducing the coefficient of friction [5, 34]. No statistically significant difference could be found between the three Si3N4 at any of the time points, nor was there any significant effect of time detected. In other words, both the SN10 and the SN18 samples showed friction coefficients as low as that of the SNC sample. Having a low coefficient of friction is one of the most important prerequisites for biomaterials to be used as total hip replacements (THR) and total knee replacements (TKR) [16]. Hence, the new Si3N4 sintered by SrO, MgO, and SiO2 fulfilled this prerequisite.
Wear Factor
The wear factors of the UHMWPE pins sliding against the studied Si3N4 are displayed in Fig. 3. There was no statistically significant difference between the wear of the UHMWPE pins against the different material groups. While the wear factors measured in these tests were higher for the first 1.5-M cycles when compared to previously reported values, they were lower for the last 500-k cycles—by one order of magnitude. For instance, the wear factor of UHMWPE pins sliding against a CoCrMo alloy (Ra = 15 nm) was reported to be 2.2 × 10–6 mm3/N.m [35], and UHMWPE pins sliding against Charnley implants (Ra = 10 nm) after clinical use was 2.1 × 10–6 mm3/N·m [36]. While the contact pressures were lower in those studies, another reason for the lower wear factors could be due to differences in surface roughness of the mating materials. Indeed, on the pins’ surfaces (Figs. 7–9), the formation of scratches and plastic deformation (highlighted by arrows) was observed. This was likely the result of abrasion and adhesion of the polyurethane with microscopic asperities on the ceramics’ surfaces [37]. On the other hand, the repetitive movement during the wear cycles led to a preferred orientation of the UHMWPE molecules in the main sliding direction, resulting in strain hardening [38,39,40], which may explain the lower wear factor toward the end of the test. A multidirectional wear path is commonly chosen to better mimic clinical wear [41] because path shear and tensile forces occur in several directions [42]. Despite the multidirectional movement, the same pattern was followed over time, and the increased hardness of the polymer may be a reason for the lower wear rate in the last part of the test. Another contributing reason may be the formation of a tribofilm, as previously discussed.
Surface Roughness
The surface roughness of the studied Si3N4’s before and after wear tests are presented in Fig. 4. As can be seen, both the SN10 and the SN18 samples showed surface roughness values lower than 50 nm before the wear tests, which was in accordance with the standard [43]. However, that of the SNC sample was slightly (around 5%) higher than the recommended value. It can also be observed that the surface roughness of the SNC sample was, in general, the highest during the wear test, an effect that was statistically significant for all time points, except vs SN10 at 0–0.5MC and at 1.5–2.0MC. In fact, the surface roughness of the SN10 sample increased progressively during the test, reaching around 50 nm at the end of the test. Whereas, the SN18 sample maintained a surface roughness value in agreement with the standard [43] during the whole test, which was significantly lower than the SNC sample at each time point. The increase in surface roughness of the SN10 sample indicated that some dissolution of the surface could have taken place during the test. However, the actual dissolution rate of the new Si3N4 needs further investigation. It is a complex matter depending on many factors, such as the type of liquid medium, temperature, and pH. A comprehensive study on the dissolution rate of the materials was not carried out herein because it was out of the scope of the present study. The focus herein was on an initial evaluation of the wear behavior of the two new Si3N4 ceramics.
Surface roughness (Ra) of UHMWPE pins before and after wear tests is shown in Fig. 5. The pins had similar surface roughness (1.5 μm) before the wear test. No statistically significant difference in roughness was found between the pins ran against the control material or against SN10, except at the 1–1.5MC time point. Also, there was no significant difference between the pins against SNC and SN18 except at 0 cycles and at 1–1.5MC, where the pin ran against SN18 had a significantly lower roughness. Therefore, while a generally lower roughness may have been expected for the pins running against the new materials, which had a lower roughness than the control throughout the tests, this effect was not evident. One limiting factor here may be the relatively low number of samples tested. Another reason may be the high scatter of the data, possibly due to an abrasive process in the beginning of the tests—with the highest scatter—and the polymer strain-hardening effect and/or tribofilm resulting in a lower scatter in the remainder of the tests. This is also in accordance with the lower wear factors toward the later parts of the test. Indeed, the effect of the tribofilm may have canceled any effect of the difference in roughness of the ceramic materials.
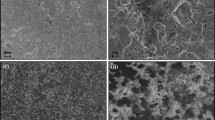
The surface morphology evolution of the pins can be seen in Figs. 6, 7, and 8. The pins showed a smooth surface before the wear tests (Figs. 6a, 7a, and 8a). For the pins slid against the SNC sample, plastic deformation occurred on the surface of the pin after 0.5 MC (Fig. 6). On the other hand, for the pins which slid against the SN10 sample, a few machining marks were visible after 0.5 MC, and plastic deformation occurred after 1.0 MC. After 2.0 MC, deformities can be observed on the pin surface (Fig. 7). As for pins slid against the SN18 sample, plastic deformation only occurred after 1.0 MC and after 2.0 MC (Fig. 8). Hence, the type and relative amount of the glass phase in the Si3N4 showed effects on the surface morphology of the pins after wearing, suggesting it may also have had an effect on the tribofilm.
Surface evolution the UHMWPE pins which slid against the SN10 sample. a Before wear test; b after 0.5 MC with few machining marks; c after 1.0 MC with plastic deformation; and d after 2.0 MC showing deformities on the pin surface. Plastic deformation/wear occurring during the wear cycles are highlighted by arrows
Surface Morphology
The surface morphologies of the studied Si3N4 before and after wear tests were observed with SEM and the results are displayed in Fig. 9. The as-sintered Si3N4 showed relatively smooth surfaces after polishing, except for a small number of shallow voids (Fig. 9a–c). The shallow voids could be pores formed because of incomplete densification or surface defects caused by the grinding and polishing. To more clearly visualize the Si3N4 grains, the surfaces were etched with HF acid and the corresponding SEM images are shown in Fig. 9g–I, from which long rod-like β-Si3N4 grains were observed [44]. During the wear tests, slight dissolution occurred on the surface of the ceramic discs (Fig. 9d–f), which was in accordance with earlier studies [19, 21]. The glass phase slowly dissolved exposing the Si3N4 grains, resulting in a rougher surface, which could therefore increase the wear of the counter surface. However, the pin wear rates decreased over time, which could be due to polishing and hardening effects of the polymer surface, as mentioned earlier. No particles could be found on the surface of the materials, nor any visible debris of the ceramics. This could be indicative of good wear resistance, since the presence of particles or debris on the interfaces of the ceramics would not only result in the formation of more particles but also aggravate friction and wear due to a plowing effect [45].
pH Measurements
Figure 10 shows the pH of the wear test solutions. The pH value of the fetal bovine serum solution was approximately 7.7. The wear test solutions showed a pH of around 8.5. This means that basic ions, probably ammonium ions (NH4+) and their modifications, were released from the Si3N4 surfaces to the wear test solutions during the test. Si3N4 has an oxidized surface layer of a silicon oxynitride (Si–O–N). Friction occurring at the surface of Si3N4 might have resulted in the formation and release of ammonium ions. It has been reported that this might induce bacteriolysis [46], which renders Si3N4 bacteriostatic properties. Both the SN10 sample and the SN18 sample had a statistically significant higher pH value than that of the SNC sample at all time points except at 1.5–2.0MC. It has previously been shown that not only does the pH interfere with the dissolution of the material, but it also affects the coefficient of friction and wear [47]. When the system has a higher pH and a higher surface charge, the tribofilm seems to be thinner, resulting in a higher electrostatic repulsive force between the debris generated during wear and the tested surface [48]. Further investigation into the effect of pH on the current system would be of interest, especially considering that inflammation may give rise to a local pH decrease [49, 50]. In vivo, the synovial fluid pH is around 7.3—7.4 for normal conditions [49, 50], whereas pH values ranging between 6.6 and 7.4 have been found in fluids of inflamed joints [51].
Conclusions
In this work, the wear properties of two new Si3N4 bioceramics were evaluated. The new Si3N4 bioceramics were sintered using SrO, MgO, and SiO2 as sintering additives, rather than traditional aids, i.e., Al2O3 and Y2O3. Multidirectional wear tests showed that the new Si3N4 bioceramics had promising wear resistance, including low coefficients of friction and a low wear factor of UHMWPE pins at longer cycle times. The formation of a lubricating tribolayer appeared important to the wear factor of the mating UHMWPE surface. There was no statistically significant difference in pin wear rates between the new Si3N4 compositions and the reference Si3N4 sintered using Al2O3 and Y2O3 as additives. Although a slight dissolution occurred on the surface of the Si3N4 during the test, no particles or debris were found at the friction interface. It can be concluded the new Si3N4 bioceramics showed promise in terms of wear performance but further tests simulating more realistic in vivo conditions are necessary.
References
F.L. Riley, Silicon nitride and related materials. J. Am. Ceram. Soc. 83, 245–265 (2004). https://doi.org/10.1111/j.1151-2916.2000.tb01182.x
Z. Krstic, V.D. Krstic, Silicon nitride: the engineering material of the future. J. Mater. Sci. 47, 535–552 (2012). https://doi.org/10.1007/s10853-011-5942-5
M. Mazzocchi, D. Gardini, P.L. Traverso, M.G. Faga, A. Bellosi, On the possibility of silicon nitride as a ceramic for structural orthopaedic implants. Part II: Chemical stability and wear resistance in body environment. J. Mater. Sci. Mater. Med. 19, 2889–2901 (2008). https://doi.org/10.1007/s10856-008-3437-y
M. Pettersson, Z. Pakdaman, H. Engqvist, Y. Liu, Z. Shen, E. Östhols, Spark plasma sintered β-phase silicon nitride with Sr and Ca as a sintering aid for load bearing medical applications. J. Eur. Ceram. Soc. 32, 2705–2709 (2012). https://doi.org/10.1016/j.jeurceramsoc.2011.12.027
R.M. Bock, B.J. McEntire, B.S. Bal, M.N. Rahaman, M. Boffelli, G. Pezzotti, Surface modulation of silicon nitride ceramics for orthopaedic applications. Acta Biomater. 26, 318–330 (2015). https://doi.org/10.1016/j.actbio.2015.08.014
B. Sonny, M. Raham, The Rationale for Silicon Nitride Bearings in Orthopaedic Applications, in Advances in ceramics—electric and magnetic ceramics, bioceramics, ceramics and environment. ed. by C. Sikalidis (InTech, London, 2011), pp.421–432
C.C. Berndt, Concise encyclopedia of advanced ceramic materials. Mater. Sci. Eng. A 160, 283–284 (1993). https://doi.org/10.1016/0921-5093(93)90458-q
J. Chevalier, What future for zirconia as a biomaterial? Biomaterials 27, 535–543 (2006). https://doi.org/10.1016/j.biomaterials.2005.07.034
J. Chevalier, A. Liens, H. Reveron, F. Zhang, P. Reynaud, T. Douillard, L. Preiss, V. Sergo, V. Lughi, M. Swain, N. Courtois, Forty years after the promise of «ceramic steel?»: Zirconia-based composites with a metal-like mechanical behavior. J. Am. Ceram. Soc. 103, 1482–1513 (2020). https://doi.org/10.1111/jace.16903
D.J. Gorth, S. Puckett, B. Ercan, T.J. Webster, M. Rahaman, B. Sonny Bal, Decreased bacteria activity on Si3N4 surfaces compared with PEEK or titanium. Int. J. Nanomed. 7, 4829–4840 (2012). https://doi.org/10.2147/IJN.S35190
C.C. Guedes e Silva, O.Z. Higa, J.C. Bressiani, Cytotoxic evaluation of silicon nitride-based ceramics. Mater. Sci. Eng. C 24, 643–646 (2004). https://doi.org/10.1016/j.msec.2004.08.007
B. McEntire, R. Bock, M. Rahaman, B.S. Bal, T. Webster, G. Pezzotti, Anti-infective and osteointegration characteristics of silicon nitirde spinal fusion implants. Orthop. Proc. 98B, 32–32 (2016). https://doi.org/10.1302/1358-992X.98BSUPP_3.ISTA2014-032
J. Olofsson, T.M. Grehk, T. Berlind, C. Persson, S. Jacobson, H. Engqvist, Evaluation of silicon nitride as a wear resistant and resorbable alternative for total hip joint replacement. Biomatter. 2, 94–102 (2012). https://doi.org/10.4161/biom.20710
E.Y. Sun, P.F. Becher, K.P. Plucknett, C.H. Hsueh, K.B. Alexander, S.B. Waters, K. Hirao, M.E. Brito, Microstructural design of silicon nitride with improved fracture toughness: II, Effects of yttria and alumina additives. J. Am. Ceram. Soc. 81, 2831–2840 (1998). https://doi.org/10.1111/j.1151-2916.1998.tb02703.x
Z. Luo, H. Liang, C. Qin, J. Zhang, T. Liu, A. Lu, Sintering behavior, microstructures and mechanical properties of porous CaO–Al2O3–SiO2–Si3N4 glass-ceramics. J. Alloys Compd. 773, 71–77 (2019). https://doi.org/10.1016/j.jallcom.2018.09.231
M. Rahaman, W. Xiao, Silicon nitride bioceramics in healthcare. Int. J. Appl. Ceram. Technol. 15, 861–872 (2018). https://doi.org/10.1111/ijac.12836
S. Tarafder, W.S. Dernell, A. Bandyopadhyay, S. Bose, SrO- and MgO-doped microwave sintered 3D printed tricalcium phosphate scaffolds: Mechanical properties and in vivo osteogenesis in a rabbit model, J. Biomed. Mater. Res. Part B Appl. Biomater. 103, 679–690 (2015). https://doi.org/10.1002/jbm.b.33239
H. Zreiqat, Y. Ramaswamy, C. Wu, A. Paschalidis, Z.F. Lu, B. James, O. Birke, M. McDonald, D. Little, C.R. Dunstan, The incorporation of strontium and zinc into a calcium-silicon ceramic for bone tissue engineering. Biomaterials 31, 3175–3184 (2010). https://doi.org/10.1016/j.biomaterials.2010.01.024
L. Fu, H. Engqvist, W. Xia, Spark plasma sintering of biodegradable Si3N4 bioceramic with Sr, Mg and Si as sintering additives for spinal fusion. J. Eur. Ceram. Soc. 38, 2110–2119 (2018). https://doi.org/10.1016/j.jeurceramsoc.2017.10.003
D.M. Reffitt, N. Ogston, R. Jugdaohsingh, H.F.J. Cheung, B.A.J. Evans, R.P.H. Thompson, J.J. Powell, G.N. Hampson, Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro. Bone 32, 127–135 (2003). https://doi.org/10.1016/S8756-3282(02)00950-X
L. Fu, Y. Xiong, G. Carlsson, M. Palmer, S. Örn, W. Zhu, X. Weng, H. Engqvist, W. Xia, Biodegradable Si3N4 bioceramic sintered with Sr, Mg and Si for spinal fusion: Surface characterization and biological evaluation. Appl. Mater. Today. 12, 260–275 (2018). https://doi.org/10.1016/j.apmt.2018.06.002
M.S. Lord, M. Foss, F. Besenbacher, Influence of nanoscale surface topography on protein adsorption and cellular response. Nano Today 5, 66–78 (2010). https://doi.org/10.1016/j.nantod.2010.01.001
T.J. Webster, J.U. Ejiofor, Increased osteoblast adhesion on nanophase metals: Ti, Ti6Al4V, and CoCrMo. Biomaterials 25, 4731–4739 (2004). https://doi.org/10.1016/j.biomaterials.2003.12.002
T.J. Webster, R.W. Siegel, R. Bizios, Osteoblast adhesion on nanophase ceramics. Biomaterials 20, 1221–1227 (1999). https://doi.org/10.1016/S0142-9612(99)00020-4
T.J. Webster, C. Ergun, R.H. Doremus, R.W. Siegel, R. Bizios, Enhanced osteoclast-like cell functions on nanophase ceramics. Biomaterials 22, 1327–1333 (2001). https://doi.org/10.1016/S0142-9612(00)00285-4
T.J. Webster, C. Ergun, R.H. Doremus, R.W. Siegel, R. Bizios, Enhanced osteoclast like-cell functions on nanophase ceramics. Ann. Biomed. Eng. 28, 1803–1810 (2000)
ASTM Standard F732, Standard test method for wear testing of polymeric materials used in total joint prostheses (ASTM International, West Conshohocken, 2017)
ASTM, Astm Standard, F2025–06, Standard practice for gravimetric measurement of polymeric components for wear assessment (ASTM International, West Conshohocken, 2018)
D.D. La Grange, N. Goebbels, A. Santana, R. Heuberger, T. Imwinkelried, L. Eschbach, A. Karimi, Effect of niobium onto the tribological behavior of cathodic arc deposited Nb–Ti–N coatings. Wear 368–369, 60–69 (2016). https://doi.org/10.1016/j.wear.2016.09.003
A. Escudeiro, M.A. Wimmer, T. Polcar, A. Cavaleiro, Tribological behavior of uncoated and DLC-coated CoCr and Ti-alloys in contact with UHMWPE and PEEK counterbodies. Tribol. Int. 89, 97–104 (2015). https://doi.org/10.1016/j.triboint.2015.02.002
R.C. Dante, C.K. Kajdas, A review and a fundamental theory of silicon nitride tribochemistry. Wear (2012). https://doi.org/10.1016/j.wear.2012.03.001
J. Xu, K. Kato, Formation of tribochemical layer of ceramics sliding in water and its role for low friction. Wear 245, 61–75 (2000). https://doi.org/10.1016/S0043-1648(00)00466-X
I.W. Park, D.S. Kang, J.J. Moore, S.C. Kwon, J.J. Rha, K.H. Kim, Microstructures, mechanical properties, and tribological behaviors of Cr–Al–N, Cr–Si–N, and Cr–Al–Si–N coatings by a hybrid coating system. Surf. Coatings Technol. 201, 5223–5227 (2007). https://doi.org/10.1016/j.surfcoat.2006.07.118
M. Das, K. Bhimani, V.K. Balla, In vitro tribological and biocompatibility evaluation of sintered silicon nitride. Mater. Lett. 212, 130–133 (2018). https://doi.org/10.1016/j.matlet.2017.10.061
L.A. Korduba, A. Wang, The effect of cross-shear on the wear of virgin and highly-crosslinked polyethylene. Wear 271, 1220–1223 (2011). https://doi.org/10.1016/j.wear.2011.01.039
R.M. Hall, A. Unsworth, Wear in retrieved Charnley acetabular sockets. Proc. Inst Mech. Eng. Part H J. Eng. Med. 210, 197–207 (1996). https://doi.org/10.1243/PIME_PROC_1996_210_413_02
J.R. Cooper, D. Dowson, J. Fisher, Macroscopic and microscopic wear mechanisms in ultra-high molecular weight polyethylene. Wear 162–164, 378–384 (1993). https://doi.org/10.1016/0043-1648(93)90521-M
A. Wang, A. Essner, V.K. Polineni, C. Stark, J.H. Dumbleton, Lubrication and wear of ultra-high molecular weight polyethylene in total joint replacements. Tribol. Int. 31, 17–33 (1998). https://doi.org/10.1016/S0301-679X(98)00005-X
A. Wang, V.K. Polineni, A. Essner, M. Sokol, D.C. Sun, C. Stark, J.H. Dumbleton, The significance of nonlinear motion in the wear screening of orthopaedic implant materials. J. Test. Eval. 25, 239–245 (1997). https://doi.org/10.1520/jte11485j
A. Wang, C. Stark, J.H. Dumbleton, Mechanistic and morphological origins of ultra-high molecular weight polyethylene wear debris in total joint replacement prostheses. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 210, 141–155 (1996). https://doi.org/10.1243/PIME_PROC_1996_210_407_02
V. Saikko, J. Kostamo, A new method and device for advanced wear simulation of orthopaedic biomaterials. J. Biomech. 44, 810–814 (2011). https://doi.org/10.1016/j.jbiomech.2010.12.024
A. Wang, D.C. Sun, S.S. Yau, B. Edwards, M. Sokol, A. Essner, V.K. Polineni, C. Stark, J.H. Dumbleton, Orientation softening in the deformation and wear of ultra-high molecular weight polyethylene. Wear 203–204, 230–241 (1997). https://doi.org/10.1016/S0043-1648(96)07362-0
ASTM Standard F2033–12, Specification for total hip joint prosthesis and hip endoprosthesis bearing surfaces made of metallic, ceramic, and polymeric materials (ASTM International, West Conshohocken, 2012)
Y. Duan, N. Liu, J. Zhang, H. Zhang, X. Li, Cost effective preparation of Si3N4 ceramics with improved thermal conductivity and mechanical properties. J. Eur. Ceram. Soc. 40, 298–304 (2020). https://doi.org/10.1016/j.jeurceramsoc.2019.10.003
L. Fu, B. Wang, J. Song, C. Xu, G. Xu, Q. Sun, J. Huang, H. Engqvist, W. Xia, Understanding microstructure-mechanical properties relationship in ZrO2–SiO2 nanocrystalline glass-ceramics: The effect of ZrO2 content. Mater. Sci. Eng. A. (2022). https://doi.org/10.1016/j.msea.2022.142904
G. Pezzotti, R.M. Bock, B.J. McEntire, E. Jones, M. Boffelli, W. Zhu, G. Baggio, F. Boschetto, L. Puppulin, T. Adachi, T. Yamamoto, N. Kanamura, Y. Marunaka, B.S. Bal, Silicon nitride bioceramics induce chemically driven lysis in Porphyromonas gingivalis. Langmuir 32, 3024–3035 (2016). https://doi.org/10.1021/acs.langmuir.6b00393
R.P. De Oliveira, E. Dos Santos, T. Cousseau, A. Sinatora, Effect of pH on wear and friction of silicon nitride sliding against alumina in water. Tribol. Int. 90, 356–361 (2015). https://doi.org/10.1016/j.triboint.2015.04.003
M. Kalin, S. Novak, J. Vižintin, Surface charge as a new concept for boundary lubrication of ceramics with water. J. Phys. D. Appl. Phys. 39, 3138–3149 (2006). https://doi.org/10.1088/0022-3727/39/15/S03
N.A. Cummings, G.L. Nordby, Measurement of synovial fluid pH in normal and arthritic knees. Arthritis Rheum. 9, 47–56 (1966). https://doi.org/10.1002/art.1780090106
E.H. Jebens, M.E. Monk-Jones, On the viscosity and pH of synovial fluid and the pH of blood. J. Bone Joint Surg. Br. 41B, 388–400 (1959). https://doi.org/10.1302/0301-620x.41b2.388
P.S. Treuhaft, D.J. McCarty, Synovial fluid pH, lactate, oxygen and carbon dioxide partial pressure in various joint diseases. Arthritis Rheum. 14, 475–484 (1971). https://doi.org/10.1002/art.1780140407
Acknowledgements
Dr Alejandro Lopez and Ms Camilla Berg are gratefully acknowledged for assistance with the XRD analysis.
Funding
Open access funding provided by Uppsala University. This research was funded by the European Union, Grant Number FP7-NMP-2012-310477 (Life Long Joints project); EBW+Project Erasmus Mundus Program, Action 2—STRAND 1, Lot 9 (Latin America), Brazil, Grant number 2014-0982; and the Chinese Scholarship Council (CSC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Correa Filho, L., Fu, L., Engqvist, H. et al. Wear Performance of a Novel Silicon Nitride Ceramic for Biomedical Applications. Biomedical Materials & Devices 1, 990–999 (2023). https://doi.org/10.1007/s44174-022-00061-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44174-022-00061-w