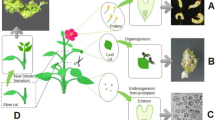
Abstract
Plants have a remarkably regenerative capability to replace the damaged organs or form the new organs and individuals both in vivo and in vitro, which is fundamental for their developmental plasticity and the agricultural practices. The regenerative capacities of plants are highly dependent on the totipotency or pluripotency of somatic cells, whose fates are directed by phytohormones, wounding, and other stimuli. Recent studies have revealed that the two types of cellular reprogramming are involved in the acquisition of cell pluripotency during plant in vitro and in vivo regeneration programs. This review focuses on the recent advances of the cellular origin, molecular characteristic, and genetic and epigenetic regulations of cell pluripotency acquisition in plants, highlighting the molecular frameworks of cellular reprogramming activated by diverse stimuli and their possible potentials in regeneration-based plant biotechnologies.

Similar content being viewed by others
References
Ahmad P, Ashraf M, Younis M, Hu X, Kumar A, Akram NA, Al-Qurainy F (2012) Role of transgenic plants in agriculture and biopharming. Biotechnol Adv 30:524–540. https://doi.org/10.1016/j.biotechadv.2011.09.006
Aichinger E, Villar CB, Di Mambro R, Sabatini S, Kohler C (2011) The CHD3 chromatin remodeler PICKLE and polycomb group proteins antagonistically regulate meristem activity in the Arabidopsis root. Plant Cell 23:1047–1060. https://doi.org/10.1105/tpc.111.083352
Aida M et al (2004) The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell 119:109–120
Atta R et al (2009) Pluripotency of Arabidopsis xylem pericycle underlies shoot regeneration from root and hypocotyl explants grown in vitro. Plant J 57:626–644. https://doi.org/10.1111/j.1365-313X.2008.03715.x
Bagchi R et al (2018) The Arabidopsis ALF4 protein is a regulator of SCF E3 ligases. EMBO J 37:255–268. https://doi.org/10.15252/embj.201797159
Banno H, Ikeda Y, Niu QW, Chua NH (2001) Overexpression of Arabidopsis ESR1 induces initiation of shoot regeneration. Plant Cell 13:2609–2618. https://doi.org/10.1105/tpc.010234
Birnbaum KD, Sanchez Alvarado A (2008) Slicing across kingdoms: regeneration in plants and animals. Cell 132:697–710
Che P, Lall S, Howell SH (2007) Developmental steps in acquiring competence for shoot development in Arabidopsis tissue culture. Planta 226:1183–1194
Ckurshumova W, Smirnova T, Marcos D, Zayed Y, Berleth T (2014) Irrepressible MONOPTEROS/ARF5 promotes de novo shoot formation. New Phytol 204:556–566. https://doi.org/10.1111/nph.13014
Daniel B et al (2015) Oxidation of monolignols by members of the berberine bridge enzyme family suggests a role in plant cell wall metabolism. J Biol Chem 290:18770–18781. https://doi.org/10.1074/jbc.M115.659631
Dastidar MG et al (2019) ARF5/MONOPTEROS directly regulates miR390 expression in the Arabidopsis thaliana primary root meristem. Plant Direct 3:e00116. https://doi.org/10.1002/pld3.116
DiDonato RJ et al (2004) Arabidopsis ALF4 encodes a nuclear-localized protein required for lateral root formation. Plant J 37:340–353. https://doi.org/10.1046/j.1365-313X.2003.01964.x
Ding Z, Friml J (2010) Auxin regulates distal stem cell differentiation in Arabidopsis roots. Proc Natl Acad Sci USA 107:12046–12051. https://doi.org/10.1073/pnas.1000672107
Fan M, Xu C, Xu K, Hu Y (2012) Lateral organ boundaries domain transcription factors direct callus formation in Arabidopsis regeneration. Cell Res 22:1169–1180. https://doi.org/10.1038/cr.2012.63
Fukaki H, Tameda S, Masuda H, Tasaka M (2002) Lateral root formation is blocked by a gain-of-function mutation in the SOLITARY-ROOT/IAA14 gene of Arabidopsis. Plant J 29:153–168. https://doi.org/10.1046/j.0960-7412.2001.01201.x
Furuta K et al (2011) The CKH2/PKL chromatin remodeling factor negatively regulates cytokinin responses in Arabidopsis calli. Plant Cell Physiol 52:618–628. https://doi.org/10.1093/pcp/pcr022
Galinha C, Hofhuis H, Luijten M, Willemsen V, Blilou I, Heidstra R, Scheres B (2007) PLETHORA proteins as dose-dependent master regulators of Arabidopsis root development. Nature 449:1053–1057. https://doi.org/10.1038/nature06206
Gordon SP, Heisler MG, Reddy GV, Ohno C, Das P, Meyerowitz EM (2007) Pattern formation during de novo assembly of the Arabidopsis shoot meristem. Development 134:3539–3548
Guo F et al (2018) Callus initiation from root explants employs different strategies in rice and Arabidopsis. Plant Cell Physiol 59:1782–1789. https://doi.org/10.1093/pcp/pcy095
Heyman J et al (2016) The heterodimeric transcription factor complex ERF115-PAT1 grants regeneration competence. Nat Plants 2:16165. https://doi.org/10.1038/nplants.2016.165
Himanen K et al (2004) Transcript profiling of early lateral root initiation. Proc Natl Acad Sci USA 101:5146–5151. https://doi.org/10.1073/pnas.0308702101
Holec S, Berger F (2012) Polycomb group complexes mediate developmental transitions in plants. Plant Physiol 158:35–43. https://doi.org/10.1104/pp.111.186445
Hu X, Xu L (2016) Transcription factors WOX11/12 directly activate WOX5/7 to promote root primordia initiation and organogenesis. Plant Physiol 172:2363–2373. https://doi.org/10.1104/pp.16.01067
Ikeuchi M, Sugimoto K, Iwase A (2013) Plant callus: mechanisms of induction and repression. Plant Cell 25:3159–3173. https://doi.org/10.1105/tpc.113.116053
Ikeuchi M et al (2015) PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis. Nat Plants 1:15089. https://doi.org/10.1038/nplants.2015.89
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451. https://doi.org/10.1242/dev.134668
Ikeuchi M et al (2017) Wounding triggers callus formation via dynamic hormonal and transcriptional changes. Plant Physiol 175:1158–1174. https://doi.org/10.1104/pp.17.01035
Ikeuchi M, Favero DS, Sakamoto Y, Iwase A, Coleman D, Rymen B, Sugimoto K (2019) Molecular mechanisms of plant regeneration. Annu Rev Plant Biol 70:377–406. https://doi.org/10.1146/annurev-arplant-050718-100434
Iwase A et al (2011) The AP2/ERF transcription factor WIND1 controls cell dedifferentiation in Arabidopsis. Curr Biol 21:508–514. https://doi.org/10.1016/j.cub.2011.02.020
Iwase A et al (2017) WIND1 promotes shoot regeneration through transcriptional activation of ENHANCER OF SHOOT REGERATION 1 in Arabidopsis. Plant Cell 29:54–69. https://doi.org/10.1105/tpc.16.00623
Kareem A et al (2015) PLETHORA genes control regeneration by a two-step mechanism. Curr Biol 25:1017–1030. https://doi.org/10.1016/j.cub.2015.02.022
Kim JY, Yang W, Forner J, Lohmann JU, Noh B, Noh YS (2018) Epigenetic reprogramming by histone acetyltransferase HAG1/AtGCN5 is required for pluripotency acquisition in Arabidopsis. EMBO J 37:e98726. https://doi.org/10.15252/embj.201798726
Kirch T, Simon R, Grunewald M, Werr W (2003) The DORNROSCHEN/ENHANCER OF SHOOT REGENERATION1 gene of Arabidopsis acts in the control of meristem ccll fate and lateral organ development. Plant Cell 15:694–705. https://doi.org/10.1105/tpc.009480
Laplaze L et al (2005) GAL4-GFP enhancer trap lines for genetic manipulation of lateral root development in Arabidopsis thaliana. J Exp Bot 56:2433–2442. https://doi.org/10.1093/jxb/eri236
Lee K, Seo PJ (2018) Dynamic epigenetic changes during plant regeneration. Trends Plant Sci 23:235–247. https://doi.org/10.1016/j.tplants.2017.11.009
Lee HW, Kim MJ, Kim NY, Lee SH, Kim J (2013) LBD18 acts as a transcriptional activator that directly binds to the EXPANSIN14 promoter in promoting lateral root emergence of Arabidopsis. Plant J 73:212–224. https://doi.org/10.1111/tpj.12013
Lee K, Park OS, Seo PJ (2017) Arabidopsis ATXR2 deposits H3K36me3 at the promoters of LBD genes to facilitate cellular dedifferentiation. Sci Signal 10:eaan0316
Lee K, Park OS, Seo PJ (2018) JMJ30-mediated demethylation of H3K9me3 drives tissue identity changes to promote callus formation in Arabidopsis. Plant J 95:961–975. https://doi.org/10.1111/tpj.14002
Li Y et al (2015) The histone methyltransferase SDG8 mediates the epigenetic modification of light and carbon responsive genes in plants. Genome Biol 16:79. https://doi.org/10.1186/s13059-015-0640-2
Liu J, Sheng L, Xu Y, Li J, Yang Z, Huang H, Xu L (2014) WOX11 and 12 are involved in the first-step cell fate transition during de novo root organogenesis in Arabidopsis. Plant Cell 26:1081–1093. https://doi.org/10.1105/tpc.114.122887
Liu Z et al (2016) Repression of callus initiation by the miRNA-directed interaction of auxin-cytokinin in Arabidopsis thaliana. Plant J 87:391–402. https://doi.org/10.1111/tpj.13211
Liu J, Hu X, Qin P, Prasad K, Hu Y, Xu L (2018) The WOX11-LBD16 pathway promotes pluripotency acquisition in callus cells during de novo shoot regeneration in tissue culture. Plant Cell Physiol 59:734–743. https://doi.org/10.1093/pcp/pcy010
Marhava P, Hoermayer L, Yoshida S, Marhavy P, Benkova E, Friml J (2019) Re-activation of stem cell pathways for pattern restoration in plant wound healing. Cell 177(4):957–969.e13. https://doi.org/10.1016/j.cell.2019.04.015
Melnyk CW, Meyerowitz EM (2015) Plant grafting. Curr Biol 25:R183–188. https://doi.org/10.1016/j.cub.2015.01.029
Miller G et al (2009) The plant NADPH oxidase RBOHD mediates rapid systemic signaling in response to diverse stimuli. Sci Signal 2:ra45. https://doi.org/10.1126/scisignal.2000448
Mousavi SA, Chauvin A, Pascaud F, Kellenberger S, Farmer EE (2013) GLUTAMATE RECEPTOR-LIKE genes mediate leaf-to-leaf wound signalling. Nature 500:422–426. https://doi.org/10.1038/nature12478
Okushima Y, Fukaki H, Onoda M, Theologis A, Tasaka M (2007) ARF7 and ARF19 regulate lateral root formation via direct activation of LBD/ASL genes in Arabidopsis. Plant Cell 19:118–130
Pulianmackal AJ, Kareem AV, Durgaprasad K, Trivedi ZB, Prasad K (2014) Competence and regulatory interactions during regeneration in plants. Front Plant Sci 5:142. https://doi.org/10.3389/fpls.2014.00142
Radhakrishnan D, Kareem A, Durgaprasad K, Sreeraj E, Sugimoto K, Prasad K (2018) Shoot regeneration: a journey from acquisition of competence to completion. Curr Opin Plant Biol 41:23–31. https://doi.org/10.1016/j.pbi.2017.08.001
Roudier F et al (2011) Integrative epigenomic mapping defines four main chromatin states in Arabidopsis. EMBO J 30:1928–1938. https://doi.org/10.1038/emboj.2011.103
Rymen B et al (2019) Histone acetylation orchestrates wound-induced transcriptional activation and cellular reprogramming in Arabidopsis. Commun Biol 2:404. https://doi.org/10.1038/s42003-019-0646-5
Shang B, Xu C, Zhang X, Cao H, Xin W, Hu Y (2016) Very-long-chain fatty acids restrict regeneration capacity by confining pericycle competence for callus formation in Arabidopsis. Proc Natl Acad Sci USA 113:5101–5106. https://doi.org/10.1073/pnas.1522466113
Skoog F, Miller CO (1957) Chemical regulation of growth and organ formation in plant tissues cultured in vitro. Symp Soc Exp Biol 54:118–130
Stobbe H, Schmitt U, Eckstein D, Dujesiefken D (2002) Developmental stages and fine structure of surface callus formed after debarking of living lime trees (Tilia sp.). Ann Bot 89:773–782. https://doi.org/10.1093/aob/mcf137
Sugimoto K, Jiao Y, Meyerowitz EM (2010) Arabidopsis regeneration from multiple tissues occurs via a root development pathway. Dev Cell 18:463–471. https://doi.org/10.1016/j.devcel.2010.02.004
Sugimoto K, Gordon SP, Meyerowitz EM (2011) Regeneration in plants and animals: dedifferentiation, transdifferentiation, or just differentiation? Trends Cell Biol 21:212–218. https://doi.org/10.1016/j.tcb.2010.12.004
Vanneste S et al (2005) Cell cycle progression in the pericycle is not sufficient for SOLITARY ROOT/IAA14-mediated lateral root initiation in Arabidopsis thaliana. Plant Cell 17:3035–3050
Viacheslavova AO, Berdichevets IN, Tiurin AA, Shimshilashvili KhR, Mustafaev O, Goldenkova-Pavlova IV (2012) Expression of heterologous genes in plant systems: new possibilities. Genetika 48:1245–1259
Xu K, Liu J, Fan M, Xin W, Hu Y, Xu C (2012) A genome-wide transcriptome profiling reveals the early molecular events during callus initiation in Arabidopsis multiple organs. Genomics 100:116–124. https://doi.org/10.1016/j.ygeno.2012.05.013
Xu C et al (2018) Control of auxin-induced callus formation by bZIP59-LBD complex in Arabidopsis regeneration. Nat Plants 4:108–115. https://doi.org/10.1038/s41477-017-0095-4
Zhang G et al (2019) Jasmonate-mediated wound signaling promotes plant regeneration. Nat Plants 5:491–497. https://doi.org/10.1038/s41477-019-0408-x
Zhou W et al (2019) A jasmonate signaling network activates root stem cells and promotes regeneration. Cell 177(4):942–956.e14. https://doi.org/10.1016/j.cell.2019.03.006
Acknowledgements
Some mentioned works in this review are partly funded by the National Natural Science Foundation of China (Grant no. 31830055 and Grant no. 31771632) and the Strategic Priority Research Program of Chinese Academy of Sciences (Grant no. XDB27030102).
Author information
Authors and Affiliations
Contributions
YXH and CYX wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Xu, C., Hu, Y. The molecular regulation of cell pluripotency in plants. aBIOTECH 1, 169–177 (2020). https://doi.org/10.1007/s42994-020-00028-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42994-020-00028-9