Abstract
Hepatitis B virus is a highly infectious blood borne microbial pathogen that causes several hepatic complications like liver cirrhosis and hepatocellular carcinoma. Several methods are available for the detection of HBV, but every method has their own merits and demerits, which restrict their use in clinical laboratories. The aim of this present study is the development of rapid, inexpensive, sensitive, and specific loop-mediated isothermal amplification followed by lateral flow device (LFD) for detection of HBV in blood specimens.
Methods
HBV standard plasma panels and donor plasma specimens were used to evaluate the assay. HBV DNA was extracted by using QiAamp DNA Blood Mini Kit. Amplification was carried out at constant temperature 63 °C for 60 min. LAMP end products were analyzed by using ESE LAMP tube scanner, gel electrophoresis, UV-lamp, and lateral flow device.
Results
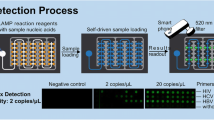
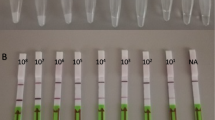
HBV-LAMP-LFD assay revealed sensitivity of 92% (138/150) of HBV positive plasma specimens. Specificity of HBV-LAMP-LFD was calculated 100%.
Conclusion
Our study concludes that HBV-LAMP-LFD is rapid, easy to use, sensitive, and specific point-of-care diagnostic assay for the detection of hepatitis B virus in blood samples. This assay can be used in resource-limited settings as well as in HBV endemic areas.








Similar content being viewed by others
Abbreviations
- LAMP:
-
Loop-mediated isothermal amplification
- LFD:
-
Lateral flow device
- LAMP-LFD:
-
Loop-mediated isothermal amplification-lateral flow device
- HBV:
-
Hepatitis B virus
References
World Health Organization. Hepatitis B (2020) Available at https://www.who.int/newsroom/fact-sheets/detail/hepatitis-b
Sleisenger and Fordtran’s. Gastrointestinal and liver disease. Pathophysiology/diagnosis/management-volume 2. 9th edition. Saunders Elsevier 1600 Jhon F. Kennedy Blvd. Ste 1800. Philadelphia, PA 19103–2899.Chapter78. HepatitisBandD. RobertPerrillo.1287–1312
Zampino R, Boemio A, Sagnelli C et al (2015) Hepatitis B virus burden in developing countries. World J Gastroenterol 21(42):11941–11953
Datta S, Chatterjee S, Veer V, Chakravarty R (2012) Molecular biology of hepatitis B virus for clinicians. J Clin Exp Hepatol 2:53–365. https://doi.org/10.1016/j.jceh.2012.10.003
Sherlock’s Diseases of the liver and Billiary System. 12th Edition (2011) A Jhon Wiley & Sons Ltd. Publication. Chapter18, Hepatitis B. Anna S. F.Lok.Pageno.367–392
Choi R, Oh Y, Park S, Lee SG, Lee EH (2019) Understanding the patient population and test utilization for hepatitis B virus testing. J Clin Lab Anal 33(9):e22987
Mansouri M, Movafagh A, Sayad A et al (2014) Hepatitis B virus infection in patients with blood disorders: a concise review in pediatric study. Iran J Ped Hematol Oncol 4(4):178–87
Bond WW, Favero MS, Petersen NJ et al (1981) Survival of hepatitis B virus after drying and storage for one week. Lancet 1:550–551
Liang TJ (2009) Hepatitis B: the virus and diseases. Hepatology 49(5 suppl):S13-21
Weber B (2005) Recent developments in the diagnosis and monitoring of HBV infection and role of the genetic variability the S gene. Expert Rev Mol Diagn 5:75–91. https://doi.org/10.1586/14737159.5.1.75
Gerlich WH (2013) Medical virology of hepatitis B: how it began and where we are now. Virol J 10:239. https://doi.org/10.1186/1743-422X-10-239
Hoofnagle JH, Seeff LB, Bales ZB, Zimmerman HJ (1978) Type B hepatitis after transfusion with blood containing antibody to hepatitis B core antigen. N Eng J Med 10:1379–1383
Chotiyaputta W, Lok AS (2009) Hepatitis B virus variants. Nat Rev Gastroenterol Hepatol 6:453–462
Datta S, Chatterjee S, Veer V (2014) Recent advances in molecular diagnostics of hepatitis B virus. World J Gastroenterol 20(40):14615–14625
Caliendo AM, Valsamakis A, Bremer JW et al (2011) Multi laboratory evaluation of real-time PCR tests for hepatitis virus DNA quantification. J Clin Microbiol 49:2854–2858
Kao JH (2008) Diagnosis of hepatitis B virus infection through serological and virological markers. Expert Rev Gastroenterol Hepatol 2:553–562
Wang F, MaC ZengX et al (2012) Chemiluminescence molecular detection of sequence-specific HBV-DNA using magnetic nanoparticles. J Bi-omed Nanotechnol 8:786–790
Hepatitis B. Fact sheet, Reviewd (2017) www.who.int/mediacentre/contacts/en/
Jain R, Aggarwal P, Gupta GN et al (2012) Need for nucleic acid testing in countries with high prevalence of transfusion-transmitted infections. ISRN Hematol 2012:718671
Blumberg BS (1997) Hepatitis B virus, the vaccine, and the control of primary cancer of the liver. Proc Natl Acad Sci USA 94:7121–7125
Nyan D-C, Ulitzky LE, Cehan N, Williamson P, Winkelman V, Rios M, Taylor DR (2014) Rapid detection of hepatitis B virus in blood plasma by a specific and sensitive loop mediated isothermal amplification assay. CID 59(1):16–23
Chen C-M, Ouyang S, Lin L-Y, Wu L-J, Xie T-A, Chen J-J, Li Z-X, Zhu G-D, Ji T-X, Pan Z-Y, Xia Y, Guo X-G (2020) Diagnostic accuracy of LAMP assay for HBV infection. J Clin Lab Anal 34:e23281
Chen H-W, Belinskaya T, Zhang Z, Ching W-M (2019) Simple detection of hepatitis B virus in using loop-mediated isothermal amplification method. Mil Med 184(7–8):e275
Quoc NB, Phuong NDN, Chau NNB, Linh DTP (2019) Closed tube loop-mediated isothermal amplification assay for rapid detection of hepatitis B virus in human blood. Heliyon 4(3):e00561
Nyan DC, Ulitzky LE, Cehan N et al (2014) Rapid detection of hepatitis B virus in blood plasma by a specific and sensitive loop-mediated isothermal amplification assay. ClinInfect Dis 2014(59):16–23
R Surabattula, MP Vejandla, PC Mallepaddi, K Faulstich, R Polavarapu (2013). Simple, rapid, inexpensive platform for the diagnosis of malaria by loop mediated isothermal amplification (LAMP). Exp Parasitol; 333–340
Global Alert and Response (GAR). HepatitisB. Available from: URL: http://www.who.Int/csr/disease/hepatitis/whocdscsrlyo20022/en/index3.html
Acknowledgements
The authors are thankful to the Genomix Biotech for providing control DNA samples for conducting the research work.
Funding
The complete work was funded and supported by institutional research grants committee.
Author information
Authors and Affiliations
Contributions
SNM, RP, RDP, and PCM carried out the experimental work and contributed in sample collection. SNM and PM conceptualized the complete work. SNM, RP, and PM contributed in writing and in developing the manuscript. PCM and Yogyata Marothi helped in the design and critically reviewed the manuscript. All the authors read and approved the final copy of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The research work was approved by institutional ethical committee of M. N. Raju Medical College and Hospital, Telengana, India conducted on 11-08-2016, approval number ECR/834/Inst/TG/2016.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Mauricio Nogueira
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maity, S.N., Poonati, R., Punati, R.D. et al. Development of sensitive and specific loop-mediated isothermal amplification combined with lateral flow device for the rapid detection of hepatitis B virus infection. Braz J Microbiol 53, 615–623 (2022). https://doi.org/10.1007/s42770-022-00690-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-022-00690-9