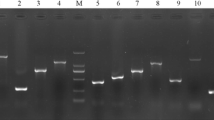
Abstract
Helicobacter pylori is the etiological agent of chronic gastritis, peptic ulcer, and gastric cancer. The duodenal ulcer-promoting gene dupA, which is located in the plasticity region of the H. pylori genome, is homologous to the virB gene which encodes a type IV secretion protein in Agrobacterium tumefaciens. Studies have shown associations between H. pylori dupA-positive strains and gastroduodenal diseases. However, whether dupA acts as a risk factor or protective factor in these diseases remains unclear. Therefore, in this study, we aimed to verify the presence of the dupA gene in infectious H. pylori strains in the Brazilian mid-west and to investigate its association with the clinical outcomes of patients with dyspepsia. Additionally, the phylogenetic origin of the strains was determined. Gastric biopsies from 117 patients with dyspepsia were analyzed using histological and molecular techniques. The hpx gene (16S rRNA) was used to screen for H. pylori infection, and positive samples were then subjected to dupA gene detection and sequencing. The estimated prevalence of H. pylori infection was 64.1%, with the dupA gene being detected in a high proportion of infectious strains (70.7%). Furthermore, a risk analysis revealed that for women, a dupA-positive H. pylori infection increased the chance of developing gastritis by twofold. The partial dupA sequences from isolated infectious strains in this work are similar to those of strains isolated in westerns countries. This study provides useful insights for understanding the role of the H. pylori dupA gene in disease development.


Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
Code availability
Not applicable.
References
Burucoa C, Axon A (2017) Epidemiology of Helicobacter pylori infection. Helicobacter 22:e12403. https://doi.org/10.1111/hel.12403
Hooi JKY, Lai WY, Ng WK, Suen MMY, Underwood FE, Tanyingoh D, Malfertheiner P, Graham DY, Wong VWS, Wu JCY, Chan FKL, Sung JJY, Kaplan GG, Ng SC (2017) Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology 153:420–429. https://doi.org/10.1053/j.gastro.2017.04.022
Guisset M, Coton T, Rey P, Debonne JM (1997) Helicobacter pylori infection in developing countries. Med Trop (Mars) 57:77–82. https://doi.org/10.4103/1319-3767.54743
Zamani M, Ebrahimtabar F, Zamani V, Miller WH, Alizadeh-Navaei R, Shokri-Shirvani J, Derakhshan MH (2018) Systematic review with meta-analysis: the worldwide prevalence of Helicobacter pylori infection. Aliment Pharmacol Ther 47:868–876. https://doi.org/10.1111/apt.14561
Baj J, Forma A, Sitarz M, Portincasa P, Garruti G, Krasowska D, Maciejewski R (2020) Helicobacter pylori virulance factors-mechanisms of bacterial pathogenicity in the gastric microenvironment. Cells 10:27. https://doi.org/10.3390/cells10010027
Buzás GM (2018) Benign and malignant gastroduodenal diseases associated with Helicobacter pylori: a narrative review and personal remarks in 2018. Minerva Gastroenterol Dietol 64:280–296. https://doi.org/10.23736/S1121-421X.18.02481-9
IARC Working Group (1994) IARC monographs on the evaluation of carcinogenic risks to humans. In: Schistosomes, liver flukes and Helicobacter pylori. IARC Monogr. Eval. Carcinog. Risks Hum, Lyon, pp 1–241
Zhang RG, Duan GC, Fan QT, Chen SY (2016) Role of Helicobacter pylori infection in pathogenesis of gastric carcinoma. World J Gastrointest Pathophysiol 7:97–107. https://doi.org/10.4291/wjgp.v7.i1.97
Backert S, Tegtmeyer, Selback M (2010) The versatility of Helicobacter pylori CagA effector protein functions: the master key hypothesis. 15:163–176. https://doi.org/10.1111/j.1523-5378.2010.00759.x
Pandya HB, Agravat HH, Patel JS (2017) Prevalence of specific Helicobacter pylori cagA, vacA, iceA, ureC genotypes and its clinical relevance in the patients with acid-peptic diseases. J Clin Diagn Res 11:DC23–DC26. https://doi.org/10.7860/JCDR/2017/27812.10457
Paredes-Osses E, Sáez K, Sanhueza E, Hebel S, González C, Briceño C, García Cancino A (2017) Association between cagA, vacAi, and dupA genes of Helicobacter pylori and gastroduodenal pathologies in Chilean patients. Folia Microbiol (Praha) 62:437–444. https://doi.org/10.1007/s12223-017-0514-y
Sayehmiri F, Kiani F, Sayehmiri K, Soroush S, Asadollahi K, Alikhani MY, Delpisheh A, Emaneini M, Bogdanović L, Varzi AM, Zarrilli R, Taherikalani M (2015) Prevalence of cagA and vacA among Helicobacter pylori-infected patients in Iran: a systematic review and meta-analysis. J Infect Dev Ctries 9:686–696. https://doi.org/10.3855/jidc.5970
Lu H, Hsu PI, Graham DY, Yamaoka Y (2005) Duodenal ulcer promoting gene of Helicobacter pylori. Gastroenterology 128:833–848. https://doi.org/10.1053/j.gastro.2005.01.009
Alam J, Sarkar A, Karmakar BC, Ganguly M, Paul S, Mukhopadhyay AK (2020) Novel virulence factor dupA of Helicobacter pylori as an important risk determinant for disease manifestation: an overview. World J Gastroenterol 26:4739–4752. https://doi.org/10.3748/wjg.v26.i32.4739
Matsunari O, Shiota S, Suzuki R, Watada M, Kinjo N, Murakami K, Fujioka T, Kinjo F, Yamaoka Y (2012) Association between Helicobacter pylori virulence factors and gastroduodenal diseases in Okinawa, Japan. J Clin Microbiol 50:876–883. https://doi.org/10.1128/JCM.05562-11
Camilo V, Sugiyama T, Touati E (2017) Pathogenesis of Helicobacter pylori infection. Helicobacter 22:1–6. https://doi.org/10.1111/hel.12405
Vinagre IDF, de Queiroz AL, da Silva Júnior MR, Vinagre RMDF, Martins LC (2015) Infecção pelo Helicobacter pylori em pacientes com diferentes doenças gastrointestinais do Norte do Brasil. Arq Gastroenterol 52:266–271. https://doi.org/10.1590/S0004-28032015000400004
Alam J, Maiti S, Ghosh P, De R, Chowdhury A, Das S, Macaden R, Devarbhavi H, Ramamurthy T, Mukhopadhyay AK (2012) Significant association of the dupA gene of Helicobacter pylori with duodenal ulcer development in a South-east Indian population. J Med Microbiol 61:1295–1302. https://doi.org/10.1099/jmm.0.038398-0
Talebi BAA, Taghvaei T, Wolfram L, Kusters JG (2012) Infection with Helicobacter pylori strains lacking dupA is associated with an increased risk of gastric ulcer and gastric cancer development. J Med Microbiol 61:23–30. https://doi.org/10.1099/jmm.0.027052-0
Kusters JG, Van Vliet AHM, Kuipers EJ (2006) Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev 19:449–490. https://doi.org/10.1128/CMR.00054-05
Chiurillo MA, Moran Y, Cañas M, Valderrama E, Granda N, Sayegh M, Ramírez JL (2013) Genotyping of Helicobacter pylori virulence-associated genes shows high diversity of strains infecting patients in western Venezuela. Int J Infect Dis 17:e750–e756. https://doi.org/10.1016/j.ijid.2013.03.004
Liu J, He C, Chen M, Wang Z, Xing C, Yuan Y (2013) Association of presence/absence and on/off patterns of Helicobacter pylori oipA gene with peptic ulcer disease and gastric cancer risks: a meta-analysis. BMC Infect Dis 13:555. https://doi.org/10.1186/1471-2334-13-555
Yong X, Tang B, Li BS, Xie R, Hu CJ, Luo G, Qin Y, Dong H, Yang SM (2015) Helicobacter pylori virulence factor CagA promotes tumorigenesis of gastric cancer via multiple signaling pathways. Cell Commun Signal 13:30. https://doi.org/10.1186/1471-2334-13-555
McClain M, Beckett AC, Cover TL (2017) Helicobacter pylori vacuolating toxin and gastric cancer. Toxins (Basel) 9:316. https://doi.org/10.3390/toxins9100316
Wang MY, Chen C, Gao XZ, Li J, Yue J, Ling F, Wang XC, Shao SH (2013) Distribution of Helicobacter pylori virulence markers in patients with gastroduodenal diseases in a region at high risk of gastric cancer. Microb Pathog 59–60:13–18. https://doi.org/10.1016/j.micpath.2013.04.001
Liu J, Liu H, Zhang T, Ren X, Nadolny C, Dong X, Huang L, Yuan K, Tian W, Jia Y (2014) Serum Helicobacter pylori NapA antibody as a potential biomarker for gastric cancer. Sci Rep 4:4143. https://doi.org/10.1038/srep04143
Souod N, Sarshar M, Dabiri H, Momtaz H, Kargar M, Mohammadzadeh A, Abdi S (2015) The study of the oipA and dupA genes in Helicobacter pylori strains and their relationship with different gastroduodenal diseases. Gastroenterol Hepatol Bed Bench 8:S47–S53
Sallas ML, Melchiades JL, Zabaglia LM, Moreno JRP, Orcini WA, Chen ES, Smith MAC, Payão SLM, Rasmussen LT (2017) Prevalence of Helicobacter pylori vacA, cagA, dupA and oipA genotypes in patients with gastric disease. Adv Microbiol 7:1–9. https://doi.org/10.4236/aim.2017.71001
Emura F, Sharma P, Arantes V, Cerisoli C, Parra-Blanco A, Sumiyama K, Araya R, Sobrino S, Chiu P, Matsuda K, González R, Fujishiro M, Tajiri H (2019) Principles and practice to facilitate complete photodocumentation of the upper gastrointestinal tract: World Endoscopy Organization position statement. Dig Endosc 32:168–179. https://doi.org/10.1111/den.13530
Coelho LGV, Marinho JR, Genta R, Ribeiro LT, Passoss MDCF, Zaterka S et al (2018) IVth Brazilian consensus conference on Helicobacter pylori infection. Arq Gastroenterol 55:97–121. https://doi.org/10.1590/s0004-2803.201800000-20
Stolte M, Meining A (2001) The updated Sydney system: classification and grading of gastritis as the basis of diagnosis and treatment. Can J Gastroenterol 15:591–598. https://doi.org/10.1155/2001/367832
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Abadi AT, Perez-Perez G (2016) Role of dupA in virulence of Helicobacter pylori. World J Gastroenterol 22:10118–10123. https://doi.org/10.3748/wjg.v22.i46.10118
Gomes LI, Rocha GA, Rocha AM, Soares TF, Oliveira CA, Bittencourt PF, Queiroz DM (2008) Lack of association between Helicobacter pylori infection with dupA-positive strains and gastroduodenal diseases in Brazilian patients. Int J Med Microbiol 298:223–230. https://doi.org/10.1016/j.ijmm.2007.05.006
Pacheco AR, Proença-Módena JL, Sales AI, Fukuhara Y, da Silveira WD, Pimenta-Módena JL, de Oliveira RB, Brocchi M (2008) Involvement of the Helicobacter pylori plasticity region and cag pathogenicity island genes in the development of gastroduodenal diseases. Eur J Clin Microbiol Infect Dis 11:1053–1059. https://doi.org/10.1007/s10096-008-0549-8
Argent RH, Burette A, Miendje Deyi VY, Atherton JC (2007) The presence of dupA in Helicobacter pylori is not significantly associated with duodenal ulceration in Belgium, South Africa, China, or North America. Clin Infect Dis 45:1204–1206. https://doi.org/10.1086/522177
Schmidt HM, Andres S, Kaakoush NO, Engstrand L, Eriksson L, Goh KL, Fock KM, Hilmi I, Dhamodaran S, Forman D, Mitchell H (2009) The prevalence of the duodenal ulcer promoting gene (dupA) in Helicobacter pylori isolates varies by ethnic group and is not universally associated with disease development: a case-control study. Gut pathogens 1:1–5. https://doi.org/10.1186/1757-4749-1-5
Yeh YC, Cheng HC, Chang WL, Yang HB, Sheu BS (2010) Matrix metalloproteinase-3 promoter polymorphisms but not dupA-H. pylori correlate to duodenal ulcers in H. pylori-infected females. BMC Microbiol 10:218. https://doi.org/10.1186/1471-2180-10-218
Suzuki R, Shiota S, Yamaoka Y (2012) Molecular epidemiology, population genetics, and pathogenic role of Helicobacter pylori. Infect Genet Evol 12:203–213. https://doi.org/10.1016/j.meegid.2011.12.002
Israel DA, Salama N, Krishna U, Rieger UM, Atherton JC, Falkow S, Peek RM Jr (2001) Helicobacter pylori genetic diversity within the gastric niche of a single human host. Proc Natl Acad Sci USA 98:14625–14630. https://doi.org/10.1073/pnas.251551698
Author information
Authors and Affiliations
Contributions
LLLS and AKSO performed data collection, data analysis, and interpretation and wrote the scientific work; ARG and JDGV helped in scientific writing and data collection; AMTCS, AJVB, and LTR data analysis; LLC and MSB conceived the study, participated in its design and coordination, and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The study was conducted according to the ethical standards of the Research Ethics Committee of the University Hospital of the Federal University of Goias (HC/UFG), under the opinion number 2.519.032, according to Resolution CNS/196/96.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tânia A. Tardelli Gomes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Lima Silva, L.L., Oliveira, A.K.S., Gama, A.R. et al. Helicobacter pylori virulence dupA gene: risk factor or protective factor?. Braz J Microbiol 52, 1921–1927 (2021). https://doi.org/10.1007/s42770-021-00553-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-021-00553-9