Abstract
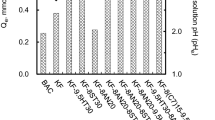
In this work, the effect of adding manganese oxide towards polyacrylonitrile-based activated carbon nanofibers (ACNFs) was evaluated. The properties of PAN-based ACNFs/MnO2 were analyzed by using scanning electron microscopy (SEM), Brunauer-Emmett-Teller (BET), and Fourier transform infrared (FTIR). The sorption study of the electrospun ACNFs/MnO2 in comparison to neat ACNFs and commercial granular AC towards lead (Pb) was also conducted. SEM micrograph analysis displays more compact nanofibers with dispersion of beads that were observed in the ACNF/MnO2 with the diameter of 437.2 nm while aligned nanofibers with the diameter of 575.5 nm were observed in the neat ACNFs. The FTIR analysis showed the peak of Mn–O, which indicates the presence of MnO2 in the ACNFs/MnO2. In comparison to the neat ACNFs, surface area of the prepared ACNFs/MnO2 is lower. It was found out that the removal of Pb(II) using ACNFs and ACNFs/MnO2 is higher than commercial granular activated carbon with a removal rate of 100% at initial concentration of 3.5 ppm. The promising results of ACNFs/MnO2 contributed by their satisfactory specific surface area and vast presence of surface functional groups.



Similar content being viewed by others
References
N. Abdullah, R.J. Gohari, N. Yusof, A.F. Ismail, J. Juhana, W.J. Lau, T. Matsuura, Polysulfone/hydrous ferric oxide ultrafiltration mixed matrix membrane: Preparation, characterization and its adsorptive removal of lead (II) from aqueous solution. Chem. Eng. J. 289, 28–37 (2016)
G. Flora, D. Gupta, A. Tiwari, Toxicity of lead: A review with recent updates. Interdiscip. Toxicol. 5, 47–58 (2012). https://doi.org/10.2478/v10102-012-0009-2
N. Abdullah, N. Yusof, J. Jaafar, A.F. Ismail, H. Hasbullah, F.E. Othman, et al., Preparation of polyacrylonitrile (PAN)/manganese oxide based activated carbon nanofiber (ACNFs) for adsorption of cadmium (II) from aqueous solution. IOP Conf Ser Environ Sci 36, 1–6 (2016)
R. Jamshidi Gohari, W.J. Lau, T. Matsuura, E. Halakoo, A.F. Ismail, Adsorptive removal of Pb(II) from aqueous solution by novel PES/HMO ultrafiltration mixed matrix membrane. Sep. Purif. Technol. 120, 59–68 (2013). https://doi.org/10.1016/j.seppur.2013.09.024
M. Barakat, New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 4, 361–377 (2011). https://doi.org/10.1016/j.arabjc.2010.07.019
M.A.P. Cechinel, S.M.A.G. Ulson De Souza, A.A. Ulson De Souza, Study of lead (II) adsorption onto activated carbon originating from cow bone. J. Clean. Prod. 65, 342–349 (2014). https://doi.org/10.1016/j.jclepro.2013.08.020
A. Al Mamun, Y.M. Ahmed, M.F.R. AlKhatib, A.T. Jameel, M. AlSaadi, Lead sorption by carbon nanofibers grown on powdered activated carbon — Kinetics and equilibrium. Nano 10, 1550017 (2015). https://doi.org/10.1142/S1793292015500174
S. Ramakrishna, K. Fujihara, W.E. Teo, T. Yong, Z. Ma, R. Ramaseshan, Electrospun nanofibers: Solving global issues. Mater. Today 9, 40–50 (2006). https://doi.org/10.1016/S1369-7021(06)71389-X
N. Yusof, A.F. Ismail, Polyacrylonitrile/acrylamide-based carbon fibers prepared using a solvent-free coagulation process: Fiber properties and its structure evolution during stabilization and carbonization. Polym. Eng. Sci. 52, 360–366 (2011)
S. Dadvar, H. Tavanai, M. Morshed, Journal of analytical and applied pyrolysis effect of embedding MgO and Al 2 O 3 nanoparticles in the precursor on the pore characteristics of PAN based activated carbon nanofibers. J. Anal. Appl. Pyrolysis 98, 98–105 (2012). https://doi.org/10.1016/j.jaap.2012.08.001
G. Oh, Y. Ju, H. Jung, W. Lee, Preparation of the novel manganese-embedded PAN-based activated carbon nanofibers by electrospinning and their toluene adsorption. J. Anal. Appl. Pyrolysis 81, 211–217 (2008). https://doi.org/10.1016/j.jaap.2007.11.006
N. Yusof, A.F. Ismail, D. Rana, T. Matsuura, Carbon fibre as a composite materials precursor- a review. Mater. Lett. 8, 16–18 (2012)
P. Kampalanonwat, P. Supaphol, The study of competitive adsorption of heavy metal ions from aqueous solution by aminated polyacrylonitrile nanofiber mats. Energy Procedia 56, 142–151 (2014). https://doi.org/10.1016/j.egypro.2014.07.142
G. Choi, Y. Ju, H. Jung, C. Kim, K. Yang, W. Lee, Preparation of manganese/PAN-based carbon nanofiber web by co-electrospinning. Appl Chem 9, 49–52 (2005)
S.G. Wang, W.X. Gong, X.W. Liu, Y.W. Yao, B.Y. Gao, Q.Y. Yue, Removal of lead(II) from aqueous solution by adsorption onto manganese oxide-coated carbon nanotubes. Sep. Purif. Technol. 58, 17–23 (2007). https://doi.org/10.1016/j.seppur.2007.07.006
L. Ji, Z. Lin, A.J. Medford, X. Zhang, Porous carbon nanofibers from electrospun polyacrylonitrile / SiO 2 composites as an energy storage material. Carbon N Y 47, 3346–3354 (2009). https://doi.org/10.1016/j.carbon.2009.08.002
F.E. Othman, N. Yusof, A. Raffi, H. Hasbullah, F. Aziz, S.W. Wan, et al., Preparation and characterizations of different loading of zinc oxide on activated carbon nanofiber. Malaysian J Anal Sci 21, 365–371 (2017)
N. Watanabe, J. Yeh, G. Lee, J. Miyawaki, I. Mochida, S. Yoon, The manganese oxide phase study on the decomposition and mechanism for formaldehyde and longevity. American Carbon Society, Biennial Conference on Carbon, Oak Ridge National, USA, 2010
Acknowledgements
The main author (Norfadhilatuladha Binti Abdullah) as well as all co-authors would like to extend their appreciation to the Ministry of Education of Malaysia and Universiti Teknologi Malaysia under Fundamental Research Grant Scheme (RJ130000.7846.4F929), GUP Grant Scheme (Q.J130000.2546.16H29), HICOE grant scheme (R.J090301.7846.4J179), and PRGS-ICC UTM (RJ130000.7746.4J329). The main author would also like to thank Research Management Centre (RMC) and Zamalah Scholarship of Universiti Teknologi Malaysia on their full technical and management support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Abdullah, N., Yusof, N., Ismail, A.F. et al. Effects of manganese(VI) oxide on polyacrylonitrile-based activated carbon nanofibers (ACNFs) and its preliminary study for adsorption of lead(II) ions. emergent mater. 1, 89–94 (2018). https://doi.org/10.1007/s42247-018-0008-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42247-018-0008-y