Abstract
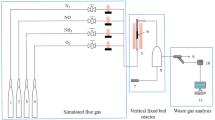
In this study, coconut shell-activated carbon was modified by the impregnation method. The best-modified reagent was 1% KOH. Under this condition, acetone’s equilibrium adsorption capacity by modified activated carbon (1% KOH-AC) reached 0.96 mg g−1. The effects of inlet time, inlet flow rate, inlet concentration, and adsorption temperature on the removal efficiency were discussed. With the increase of intake time, the removal efficiency of benzene by coconut shell-activated carbon kept 100% at first and then decreased. The higher the inlet flow rate and concentration, the worse the benzene removal. The lower the operating temperature was, the better the benzene removal was. The purification and adsorption mechanisms were explored by BET and FTIR. The results showed that the Bangham kinetic model fitted the equilibrium adsorption capacity of benzene best.
Graphical abstract











Similar content being viewed by others
References
Wang H, Nie L, Li J et al (2013) Characterization and assessment of volatile organic compounds (VOCs) emissions from typical industries. Chinese Sci Bull 58:724–730. https://doi.org/10.1007/s11434-012-5345-2
Ruan X, Liu H, Chang C-Y, Fan X (2014) Preparation of organobentonite by a novel semidry-method and its adsorption of 2,4–dichlorophenol from aqueous solution. Int Biodeter Biodegr 95:212–218. https://doi.org/10.1016/j.ibiod.2014.06.007
Wang B, Wu T, Angaiah S et al (2018) Development of nanocomposite adsorbents for heavy metal removal from wastewater. ES Mater Manuf 2:35–44. https://doi.org/10.30919/esmm5f175
Chen J, Wang X, Huang Y et al (2018) Adsorption removal of pollutant dyes in wastewater by nitrogen-doped porous carbons derived from natural leaves. Eng Sci 5:30–38. https://doi.org/10.30919/es8d666
Karimnezhad L, Haghighi M, Fatehifar E (2014) Adsorption of benzene and toluene from waste gas using activated carbon activated by ZnCl2. Front Environ Sci Eng 8:835–844
Qezelsefloo E, Khalili S, Jahanshahi J et al (2020) Adsorptive removal of CO2 on Nitrogen-doped porous carbon derived from polyaniline: effect of chemical activation. Mater Chem Phys 239:122034. https://doi.org/10.1016/j.matchemphys.2019.122304
Ma Y, Zhuang Z, Ma M et al (2019) Solid polyaniline dendrites consisting of high aspect ratio branches self-assembled using sodium lauryl sulfonate as soft templates: Synthesis and electrochemical performance. Polym 182:121808. https://doi.org/10.1016/j.polymer.2019.121808
Ma Y, Ma M, Yin X et al (2018) Tuning polyaniline nanostructures via end group substitutions and their morphology dependent electrochemical performances. Polym 156:128–135. https://doi.org/10.1016/j.polymer.2018.09.051
Hernández-Ortiz M, Lozano-López JD, Durón SM et al (2019) Quantitative measurement of functional groups on nanocarbon allotropes surface by Boehm titration. J Micro Nano-manufacturing 7:011002. https://doi.org/10.1115/1.4043419
Kim YS, Park CR (2016) Materials science and engineering of carbon. Undefined 273–286. https://doi.org/10.1016/b978-0-12-805256-3.00013-1
Shan R, Yan L, Yang K et al (2015) Adsorption of Cd(II) by Mg–Al–CO3- and magnetic Fe3O4/Mg–Al–CO3-layered double hydroxides: kinetic, isothermal, thermodynamic and mechanistic studies. J Hazard Mater 299:42–49. https://doi.org/10.1016/j.jhazmat.2015.06.003
Shi X, Kang L, Hong J et al (2021) Strong selectivity and high capacity in the adsorption of As (V) from wastewater by glycine-modified Fe/Cu-layered double hydroxides. J Alloy Compd 865:158956. https://doi.org/10.1016/j.jallcom.2021.158956
Shen Y, Zhao X, Zhang X et al (2014) Removal of Cu2+ from the aqueous solution by tartrate-intercalated layered double hydroxide. Desalin Water Treat 57:2064–2072. https://doi.org/10.1080/19443994.2014.981866
Inyinbor AA, Adekola FA, Olatunji GA (2016) Kinetics, isotherms and thermodynamic modeling of liquid phase adsorption of Rhodamine B dye onto Raphia hookerie fruit epicarp. Water Resour Ind 15:14–27. https://doi.org/10.1016/j.wri.2016.06.001
Acknowledgements
We thank the Yunnan Innovative Research Team of Environmental pollution, Food Safety, and Human Health for granting the technical guidance towards this research work.
Funding
Partial support of the present study by the Open fund of Key Laboratory of Ecological Environment Evolution and Pollution Control in Mountainous Rural Areas of Yunnan Province (Project No. 2020YB006), Key Laboratory of State Forestry and Grassland Administration on Highly-Efficient Utilization of Forestry Biomass Resources in Southwest China, Southwest Forestry University (Project No. 2020YB006), Scientific research fund of Yunnan Education Department (Project No. YB202023), and Scientific research fund project of Yunnan Education Department (Project No. 2021J0163).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Deng, Z., Deng, Q., Wang, L. et al. Modifying coconut shell activated carbon for improved purification of benzene from volatile organic waste gas. Adv Compos Hybrid Mater 4, 751–760 (2021). https://doi.org/10.1007/s42114-021-00273-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42114-021-00273-6