Abstract
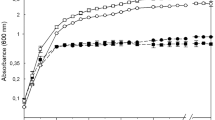
Blumeria graminis f. sp. tritici (Bgt) is an obligate parasite that only infects living tissues of wheat (Triticum aestivum L.). Long term preservation of Bgt is useful for genetic studies but it is challenging due to difficulty of artificial cultivation. In this study a simple protocol was developed in which desiccating the conidia using crystals (3–6 mm in diameter) of silica gel allowed viable storage for 12 months. The conidia were mixed with silica gel, dried for 5 h at 23 °C, and then stored at −80 °C. The preserved Bgt isolates still maintained their viability after 12 months and successfully infected detached leaves of wheat seedlings. Analysis of 20 Bgt isolates revealed that the cryopreservation process had no effect on two key phenotypic variants: virulence against 12 near-isogenic wheat cultivars and their sensitivity to the fungicide triadimefon. DNA sequencing analysis confirmed that the nucleotide sequences of the 1.4 α-demethylase inhibitor (cyp51), chitin synthase 1 (chs1), and β-tubulin (tub2) genes had not changed and still contained the corresponding single nucleotide mutations. These results indicate that the silica gel-based desiccation protocol is suitable for the long term preservation of large numbers of Bgt isolates.


Similar content being viewed by others
References
Bardin M, Suliman ME, Sage- Palloix AM, Mohamed YF, Nicot PC (2007) Inoculum production and long-term conservation methods for cucurbits and tomato powdery mildews. Mycol Res 111:740–747
Brown JKM, Wolfe MS (1990) Structure and evolution of a population of Erysiphe graminis f. sp. hordei. Plant Pathol 39:376–390
Challen MP, Elliott TJ (1986) Polypropylene straw ampoules for the storage of microorganisms in liquid nitrogen. J Microbiol Methods 5:11–22
Gong SJ, Yang LJ, Liu H, Xiang LB, Yu DZ (2011) Simple and rapid method for DNA genome micro-extraction from wheat powdery mildew (Blumeria graminis f. sp. tritici). J Microbiol 31:24–27
Hermansen JE (1972) Successful low temperature storage of conidia of Erysiphe graminis produced under dry conditions. Friesia 10:86–88
Homolka L, Lisa L, Eichlerova I, Valaskova V, Baldrian P (2010) Effect of long-term preservation of basidiomycetes on perlite in liquid nitrogen on their growth, morphological, enzymatic and genetic characteristics. Fungal Biol 114:929–935
Hopwood DA, Ferguson HM (1969) A rapid method for lyophilizing Streptomyces cultures. J Appl Bacteriol 32:434–436
Hughes HP, Macer RCF (1964) The preservation of Puccinia striiformis and other obligate cereal pathogens by vacuum-drying. Trans Br Mycol Soc 47:477–484
Koitabashi M, Yoshida S, Tsushima S (2011) Labor-saving preservation of powdery mildew of strawberry by sterilized seedling culture. Jpn Agric Res Q 45:405–409
Kutzner HJ (1972) Storage of streptomyces in soft agar and by other methods. Experientia 28:1395–1396
Nicot PC, Bardin M, Dic AJ (2002) Basic methods for epidemiological studies of powdery mildew: culture and preservation of isolates, production and delivery of inoculums, and disease assessment. In: Bélanger RR, Bushnell WR, Dik AJ, Carver TLW (eds) The powdery mildews: a comprehensive treatise. APS Press, St. Paul, pp 83–99
Niewoehner AS, Leath S (1998) Virulence of Blumeria graminis f. sp. tritici on winter wheat in the eastern United States. Plant Dis 82:64–68
Pérez-Garcia A, Mingorance E, Rivera ME, Del Pino D, Romero D, Tores JA, De Vicente A (2006) Long-term preservation of Podosphaera fusca using silica gel. J Phytopathol 154:190–192
Perkins DD (1962) Preservation of Neurospora stock cultures with anhydrous silica gel. Can J Microbiol 8:591–594
Ryan MJ, Smith D (2004) Fungal genetic resource centers and the genomic challenge. Mycol Res 108:1351–1362
Satou M (2000) Studies of physiological specialization of downy mildew of crucifers caused by Peronospora parasitica. J Gen Plant Pathol 66:283
Singh SK, Upadhyay RC, Kamal S, Tiwari M (2004) Mushroom cryopreservation and its effect on survival, yield and genetic stability. CryoLetters 25:23–32
Smith D (1998) The use of cryopreservation in the ex situ conservation of fungi. CryoLetters 19:79–90
Stummel BE, Zanker T, Scott ES (1999) Cryopreservation of air-dried conidia of Uncinula necator. Australas Plant Pathol 28:82–84
Trollope DR (1975) The preservation of bacteria and fungi on anhydrous silica gel: an assessment of survival over four years. J Appl Bacteriol 38:115–120
Wang L, Chen P, Zhou YL, Duan XY, Cao XR (2011) Sensitivity of Blumeria graminis f. sp. tritici isolates to triadimefon and azoxystrobin in 2009 in China. Acta Phytopathol Sin 41:654–658
Wei GR, Wang XJ (2011) Purification and storage of Puccinia striiformis f. sp. tritici. J Henan Agric Sci 40:90–92
Yang XJ, Yang LJ, Wang SN, Yu DZ, Ni HW (2007) Synergistic interaction of physcion and chrysophanol on plant powdery mildew. Pest Manag Sci 63:511–515
Yang XJ, Yang LJ, Wang SN, Yu DZ, Ni HW (2008) Effect of physcion, a natural anthraquinone derivative, on the infection process of Blumeria graminis on wheat. Can J Plant Pathol 30:391–396
Acknowledgments
We thank James K. M. Brown and Margaret Corbitt of the John Innes Centre for providing information about silica gels. This research was supported by Modern Agricultural Industry Technology System (CARS 03-04B), the Special Program for Agro-scientific Research in the Public Interest (20130316), the National Basic Research Program of China (2013CB127700), the “948” project from the Department of Agriculture (2012Z60), and the Foundation for Youth of Hubei Academy of Agricultural Sciences (2012NKYJJ08).
Author information
Authors and Affiliations
Corresponding author
Additional information
Section Editor: Alan Wood
Rights and permissions
About this article
Cite this article
Gong, S., Yang, L., Xiang, L. et al. An approach for long-term preservation of Blumeria graminis f. sp. tritici . Trop. plant pathol. 40, 127–133 (2015). https://doi.org/10.1007/s40858-015-0014-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40858-015-0014-z