Abstract
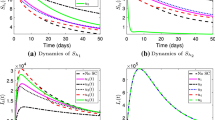
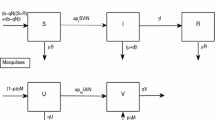
Rift Valley fever (RVF) is an emerging viral infectious disease capable of infecting livestock and human populations. Owing to possible delay in RVF diagnosis, as well as its potential adverse effects on human lives and huge economic impacts on animal trade, this paper seeks to develop a new mathematical model for RVF transmission dynamics with treatment delay and environmental transmission route. The formulated model is a ten-dimensional system of ordinary differential equations that takes into account the time-dependent vaccination of livestock and sanitation of environment as the control variables. Well-posedness of the model is established through positivity of state variables in an epidemiologically feasible region. Effective reproduction number of the model is determined, and it is observed that increase in values of constant vaccination parameters reduces the disease spread. While increase in treatment delay is shown to increase the spread of RVF in livestock and human populations. The analysis of the model is based largely on optimal control theory application. Lipschitz continuity approach is used to prove the existence of solution to the optimal control problem. Pontryagin’s maximum principle is employed to characterize both optimal vaccination and sanitation controls and the resulting optimality system is quantitatively solved. In addition, efficiency analysis is performed to determine the most efficient of the vaccination, environmental sanitation and both optimal controls required to curb the spread of RVF. The study shows that combination of both controls is the most efficient strategy that can be implemented to reduce the spread of RVF in the population significantly.








Similar content being viewed by others
References
Aldila D (2022) Dynamical analysis on a malaria model with relapse preventive treatment and saturated fumigation. Comput Math Methods Med 2022:1135452. https://doi.org/10.1155/2022/1135452
Alrabaiah H, Safi MA, DarAssi MH, Al-Hdaibat B, Ullah S, Khan MA, Shah SA (2020) Optimal control analysis of hepatitis B virus with treatment and vaccination. Results Phys 19:103599. https://doi.org/10.1016/j.rinp.2020.103599
Asamoah JKK, Okyere E, Abidemi A, Moore SE, Sun G-Q, Jin Z, Acheampong E, Gordon JF (2022) Optimal control and comprehensive cost-effectiveness analysis for COVID-19. Results Phys 33:105177. https://doi.org/10.1016/j.rinp.2022.105177
Bandekar SR, Ghosh M (2022) Mathematical modeling of COVID-19 in India and its states with optimal control. Model Earth Syst Environ 8:2019–2034. https://doi.org/10.1007/s40808-021-01202-8
Bhadauria AS, Devi S, Gupta N (2021) Modelling and analysis of a SEIQR model on COVID-19 pandemic with delay. Model Earth Syst Environ. https://doi.org/10.1007/s40808-021-01279-1
Berge T, Lubuma JM-S, Moremedi GM, Morris N, Kondera-Shava R (2016) A simple mathematical model for Ebola in Africa. J Biol Dyn 11(1):42–74. https://doi.org/10.1080/17513758.2016.1229817
Bron GM, Strimbu K, Cecilia H, Lerch A, Moore SM, Tran Q, Perkins TA, ten Bosch QA (2021) Over 100 years of Rift Valley fever: a patchwork of data on pathogen spread and spillover. Pathogens 10:708. https://doi.org/10.3390/pathogens10060708
Cecilia H, Metras R, Fall AG, Lo MM, Lancelot R, Ezanno P (2020) It’s risky to wander in September: modelling the epidemic potential of Rift Valley fever in a Sahelian setting. Epidemics 33:100409. https://doi.org/10.1016/j.epidem.2020.100409
Centers for Disease Control and Prevention. The story of the Rift Valley fever virus vaccine. U.S. Department of Health and Human Services. 2018. https://www.cdc.gov/onehealth/in-action/rvf-vaccine.html, Accessed 28 May 2022
Centers for Disease Control and Prevention. Rift Valley Fever. U.S. Department of Health and Human Services. 2020. https://www.cdc.gov/vhf/rvf/index.html. Accessed 28 May 2022
Chado UD, Abdullahi MB, Akinwande NI, Abdulrahman S, Awojoyogbe OB, Hakimi D (2016) Rift Valley fever optimal control: insecticide spray, bed net and vaccination. LAJANS 1(1):29–43
Cheneke KR, Rao KP, Edessa GK (2021) Bifurcation and stability analysis of HIV transmission model with optimal control. J Math 2021:7471290. https://doi.org/10.1155/2021/7471290
Chitnis N, Hyman JM, Manore CA (2013) Modelling vertical transmission in vector-borne diseases with applications to Rift Valley fever. J Biol Dyn 7(1):11–40. http://dx.doi.org/10.1080/17513758.2012.73342
Daubney R, Hudson JR, Garnham PC (1931) Enzootic hepatitis of Rift Valley fever, an undescribed virus disease of sheep, cattle and man from East Africa. J Pathol Bacteriol 34:545–579
Diagne MI, Rwezaura H, Tchoumi SY, Tchuenche JM (2021) A mathematical model of COVID-19 with vaccination and treatment. Comput Math Method Med 2021:1250129. https://doi.org/10.1155/2021/1250129
Fischer et al (2013) The transmission potential of Rift Valley fever virus among livestock in the Netherlands: a modelling study. Vet Res 44:58
Gaff HD, Hartley DM, Leahy NP (2007) An epidemiological model of Rift Valley fever. Electron J Differ Equ 15:1–12
Goswami NK, Shanmukha B, Hategekimana F (2022) A theoretical study of mathematical model for the spread of Zika virus disease. Nonlinear Stud 29(2):477–495
Guo J, Gao S, Yan S, Liao Z (2022) Bifurcation and optimal control analysis of delayed models for huanglongbing. Int J Biomath. https://doi.org/10.1142/S1793524522500498
Keno TD, Makinde OD, Obsu LL (2021) Optimal control and cost-effectiveness analysis of SIRS malaria disease model with temperature variability factor. J Math Fundam Sci 53(1):134–163. https://doi.org/10.5614/j.math.fund.sci.2021.53.1.10
Lenhart S, Workman JT (2007) Optimal control applied to biological models. Chapman & Hall, London
Lo lacono G, Cunningham AA, Bett B, Grace D, Redding DW, Wood JL (2018) Environmental limits of Rift Valley fever revealed using ecoepidemiological mechanistic models. PNAS 115(31):7448–7456
Lugoye J, Wairimu J, Alphonce CB, Ronoh M (2016) Modeling Rift Valley fever with treatment and trapping control strategies. Appl Math 7:556–568. https://doi.org/10.4236/am.2016.76051
Mondal J, Samu P, Chatterjee AN (2021) Effect of SOF/VEL antiviral therapy for HCV treatment: a control theoretic approach. Lett Biomath 8(1):191–213
Mpeshe SC, Haario H, Tchuenche JM (2011) A Mathematical model of Rift Valley fever with human host. Acta Biotheor 59:231–250. https://doi.org/10.1007/s10441-011-9132-2
Mpeshe SC, Luboobi LS, Nkansah-Gyekye Y (2014) Modeling the impact of climate change on the dynamics of Rift Valley fever. Comput Math Methods Med 2014:627586. https://doi.org/10.1155/2014/627586
Mpeshe SC (2021) Fractional-order derivative model of Rift Valley fever in urban peridomestic cycle. Discrete Dyn Nat Soc 2021:2941961. https://doi.org/10.1155/2021/2941961
Ndii MZ, Beay LK, Anggriani N, Nukul KNN, Djahi BS (2022) Estimating the time reproduction number in Kupang city Indonesia, 2016–2020, and assessing the effects of vaccination and different Wolbalchia strains on dengue transmission dynamics. Mathematics 10(12):2075. https://doi.org/10.3390/math10122075
Olaniyi S, Mukamuri M, Okosun KO, Adepoju OA (2022) Mathematical analysis of a social hierarchy-structured model for malaria transmission dynamics. Results Phys 34:104991. https://doi.org/10.1016/j.rinp.2021.104991
Peter OJ, Kumar S, Kumari N, Oguntolu FA, Oshinubi K, Musa R (2021) Transmission dynamics of monkeypox virus: a mathematical modelling approach. Model Earth Syst Environ. https://doi.org/10.1007/s40808-021-01313-2
Petrova V, Kristiansen P, Norheim G, Yimer SA (2020) Rift valley fever: diagnostic challenges and investment needs for vaccine development. BMJ Glob Health 5:e002694. https://doi.org/10.1136/bmjgh-2020-002694
Pépin M, Bouloy M, Bird BH, Kemp A, Paweska J (2010) Rift Valley fever virus (Bunyaviridae: Phlebovirus): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. et Res 41:61
Pontryagin LS, Boltyanskii VG, Gamkrelidze RV, Mishchenko EF (1962) The mathematical theory of optimal processes. Wiley, New York
Schreur PJ, Oymans J, Kant J, van de Water S, Kollar A, Dehon Y, Soos P, Penzes Z, van Keulen L, Kortekaas J (2021) A single vaccination with four-segmented Rift Valley fever virus prevents vertical transmission of the wild-type virus in pregnant ewes. npj Vaccines 6:8. https://doi.org/10.1038/s41541-020-00271-7
Scott GR, Weddell W, Reid D (1956) Preliminary finding on the prevalence of Rift Valley fever in Kenya cattle. Bull Epizoot Dis Afr 4:17–25
Shadi R, Fakharian A, Khaloozadeh H (2022) Transmission dynamics and optimal control strategy to mitigate the spread of novel coronovirus: the case of Iran. Int Conf Elect Eng 30:356–361. https://doi.org/10.1109/ICEE55646.2022.9827037
Sharma S, Samanta GP (2015) Stability analysis and optimal control of an epidemic model with vaccination. Int J Biomath 8(3):1550030. https://doi.org/10.1142/S1793524515500308
Srivastav AK, Ghosh M (2021) Modeling the transmission dynamics of malaria with saturated treatment: a case study of India. J Appl Math Comput 67(1):519–540. https://doi.org/10.1007/s12190-020-01469-7
Tennant WSD, Cardinale E, Cetre-Sossah C, Moutroifi Y, Le Godais G, Colombi D, Spencer SEF, Tildesley MJ, Keeling MJ, Charafouddine O, Colizza V, Edmunds WJ, Metras R (2021) Modelling the persistence and control of Rift Valley fever virus in a spatially heterogeneous landscape. Nat Commun 12:5593. https://doi.org/10.1038/s41467-021-25833-8
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48. https://doi.org/10.1016/S0025-5564(02)00108-6
Acknowledgements
Authors are thankful to anonymous reviewers and the handling editor for their detailed checking and constructive suggestions that improved the original manuscript.
Funding
There is no funding to declare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Falowo, O.D., Olaniyi, S. & Oladipo, A.T. Optimal control assessment of Rift Valley fever model with vaccination and environmental sanitation in the presence of treatment delay. Model. Earth Syst. Environ. 9, 457–471 (2023). https://doi.org/10.1007/s40808-022-01508-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40808-022-01508-1