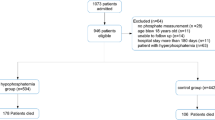
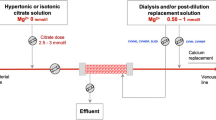
Abstract
Hypophosphatemia is a common but often underestimated electrolyte derangement among intensive care unit (ICU) patients. Low phosphate levels can lead to cellular dysfunction with potentially relevant clinical manifestations (e.g., muscle weakness, respiratory failure, lethargy, confusion, arrhythmias). In critically ill patients with severe acute kidney injury (AKI) renal replacement therapies (RRTs) represent a well-known risk factor for hypophosphatemia, especially if the most intensive and prolonged modalities of RRT, such as continuous RRT or prolonged intermittent RRT, are used. Currently, no evidence-based specific guidelines are available for the treatment of hypophosphatemia in the critically ill; however, considering the potentially negative impact of hypophosphatemia on morbidity and mortality, strategies aimed at reducing its incidence and severity should be timely implemented in the ICUs. In the clinical setting of critically ill patients on RRT, the most appropriate strategy could be to anticipate the onset of RRT-related hypophosphatemia by implementing the use of phosphate-containing solutions for RRT through specifically designed protocols. The present review is aimed at summarizing the most relevant evidence concerning epidemiology, prognostic impact, prevention and treatment of hypophosphatemia in critically ill patients with AKI on RRT, with a specific focus on RRT-induced hypophosphatemia.



Similar content being viewed by others
References
Suzuki S, Egi M, Schneider AG, Bellomo R, Hart GK, Hegarty C (2013) Hypophosphatemia in critically ill patients. J Crit Care 28:536
Ronco C, Ricci Z, De Backer D, Kellum JA, Taccone FS, Joannidis M, Pickkers P, Cantaluppi V, Turani F, Saudan P, Bellomo R, Joannes-Boyau O, Antonelli M, Payen D, Prowle JR, Vincent JL (2015) Renal replacement therapy in acute kidney injury: controversy and consensus. Crit Care 19:146
Bugg NC, Jones JA (1998) Hypophosphataemia. Pathophysiology, effects and management on the intensive care unit. Anaesthesia 53:895–902
Kim SY, Kim YN, Shin HS, Jung Y, Rim H (2017) The influence of hypophosphatemia on outcomes of low- and high-intensity continuous renal replacement therapy in critically ill patients with acute kidney injury. Kidney Res Clin Pract 36:240–249
Geerse DA, Bindels AJ, Kuiper MA, Roos AN, Spronk PE, Schultz MJ (2010) Treatment of hypophosphatemia in the intensive care unit: a review. Crit Care 14:R147
Troyanov S, Geadah D, Ghannoum M, Cardinal J, Leblanc M (2004) Phosphate addition to hemodiafiltration solutions during continuous renal replacement therapy. Intensive Care Med 30:1662–1665
VA/NIH Acute Renal Failure Trial Network, Palevsky PM, Zhang JH, O’Connor TZ, Chertow GM, Crowley ST, Choudhury D, Finkel K, Kellum JA, Paganini E, Schein RM, Smith MW, Swanson KM, Thompson BT, Vijayan A, Watnick S, Star RA, Peduzzi P (2008) Intensity of renal support in critically ill patients with acute kidney injury. N Engl J Med 359:7–20
RENAL Replacement Therapy Study Investigators, Bellomo R, Cass A, Cole L, Finfer S, Gallagher M, Lo S, McArthur C, McGuinness S, Myburgh J, Norton R, Scheinkestel C, Su S (2009) Intensity of continuous renal-replacement therapy in critically ill patients. N Engl J Med 361:1627–1638
Santiago MJ, López-Herce J, Urbano J, Bellón JM, del Castillo J, Carrillo A (2009) Hypophosphatemia and phosphate supplementation during continuous renal replacement therapy in children. Kidney Int 75:312–316
Demirjian S, Teo BW, Guzman JA, Heyka RJ, Paganini EP, Fissell WH, Schold JD, Schreiber MJ (2011) Hypophosphatemia during continuous hemodialysis is associated with prolonged respiratory failure in patients with acute kidney injury. Nephrol Dial Transplant 26:3508–3514
Broman M, Carlsson O, Friberg H, Wieslander A, Godaly G (2011) Phosphate-containing dialysis solution prevents hypophosphatemia during continuous renal replacement therapy. Acta Anaesthesiol Scand 55:39–45
Maynar Moliner J, Honore PM, Sánchez-Izquierdo Riera JA, Herrera Gutiérrez M, Spapen HD (2012) Handling continuous renal replacement therapy-related adverse effects in intensive care unit patients: the dialytrauma concept. Blood Purif 34:177–185
Yang Y, Zhang P, Cui Y, Lang X, Yuan J, Jiang H, Lei W, Lv R, Zhu Y, Lai E, Chen J (2013) Hypophosphatemia during continuous veno-venous hemofiltration is associated with mortality in critically ill patients with acute kidney injury. Crit Care 17:R205
Park JT, Lee H, Kee YK, Park S, Oh HJ, Han SH, Joo KW, Lim CS, Kim YS, Kang SW, Yoo TH, Kim DK, HICORES Investigators (2016) High-dose versus conventional-dose continuous venovenous hemodiafiltration and patient and kidney survival and cytokine removal in sepsis-associated acute kidney injury: a randomized controlled trial. Am J Kidney Dis 68:599–608
Lim C, Tan HK, Kaushik M (2017) Hypophosphatemia in critically ill patients with acute kidney injury treated with hemodialysis is associated with adverse events. Clin Kidney J 10:341–347
Talakoub R, Bahrami M, Honarmand A, Abbasi S, Gerami H (2017) The predicting ability of serum phosphorus to assess the duration of mechanical ventilation in critically ill patients. Adv Biomed Res 6:51
Bech A, Blans M, Raaijmakers M, Mulkens C, Telting D, de Boer H (2013) Hypophosphatemia on the intensive care unit: individualized phosphate replacement based on serum levels and distribution volume. J Crit Care 28:838–843
Agarwal B, Walecka A, Shaw S, Davenport A (2014) Is parenteral phosphate replacement in the intensive care unit safe? Ther Apher Dial 18:31–36
Chua HR, Baldwin I, Ho L, Collins A, Allsep H, Bellomo R (2012) Biochemical effects of phosphate-containing replacement fluid for continuous venovenous hemofiltration. Blood Purif 34:306–312
Chua HR, Schneider AG, Baldwin I, Collins A, Ho L, Bellomo R (2013) Phoxilium vs hemosol-B0 for continuous renal replacement therapy in acute kidney injury. J Crit Care 28:884.e7–884.e14
Morabito S, Pistolesi V, Tritapepe L, Zeppilli L, Polistena F, Fiaccadori E, Pierucci A (2013) Regional citrate anticoagulation in CVVH: a new protocol combining citrate solution with a phosphate-containing replacement fluid. Hemodial Int 17:313–320
Morabito S, Pistolesi V, Tritapepe L, Vitaliano E, Zeppilli L, Polistena F, Fiaccadori E, Pierucci A (2013) Continuous veno-venous hemofiltration using a phosphate-containing replacement fluid in the setting of regional citrate anticoagulation. Int J Artif Organs 36:845–852
Morabito S, Pistolesi V, Tritapepe L, Vitaliano E, Zeppilli L, Polistena F, Fiaccadori E, Pierucci A (2013) Continuous venovenous hemodiafiltration with a low citrate dose regional anticoagulation protocol and a phosphate-containing solution: effects on acid-base status and phosphate supplementation needs. BMC Nephrol 14:232
Godaly G, Carlsson O, Broman M (2016) Phoxilium(®) reduces hypophosphataemia and magnesium supplementation during continuous renal replacement therapy. Clin Kidney J 9:205–210
Pistolesi V, Zeppilli L, Polistena F, Sacco MI, Pierucci A, Tritapepe L, Regolisti G, Fiaccadori E, Morabito S (2017) Preventing continuous renal replacement therapy-induced hypophosphatemia: an extended clinical experience with a phosphate-containing solution in the setting of regional citrate anticoagulation. Blood Purif 44:8–15
Fiaccadori E, Coffrini E, Ronda N, Vezzani A, Cacciani G, Fracchia C, Rampulla C, Borghetti A (1990) Hypophosphatemia in course of chronic obstructive pulmonary disease. Prevalence, mechanisms, and relationships with skeletal muscle phosphorus content. Chest 97:857–868
Fiaccadori E, Coffrini E, Fracchia C, Rampulla C, Montagna T, Borghetti A (1994) Hypophosphatemia and phosphorus depletion in respiratory and peripheral muscles of patients with respiratory failure due to COPD. Chest 105:1392–1398
Jung SY, Kim H, Park S, Jhee JH, Yun HR, Kim H, Kee YK, Yoon CY, Oh HJ, Chang TI, Park JT, Yoo TH, Kang SW, Lee H, Kim DK, Han SH (2016) Electrolyte and mineral disturbances in septic acute kidney injury patients undergoing continuous renal replacement therapy. Medicine (Baltimore) 95:e4542
Thongprayoon C, Cheungpasitporn W, Mao MA, Sakhuja A, Erickson SB (2018) Admission hyperphosphatemia increases the risk of acute kidney injury in hospitalized patients. J Nephrol 31:241–247
Barak V, Schwartz A, Kalickman I, Nisman B, Gurman G, Shoenfeld Y (1998) Prevalence of hypophosphatemia in sepsis and infection: the role of cytokines. Am J Med 104:40–47
Berger MM, Rothen C, Cavadini C, Chiolero RL (1997) Exudative mineral losses after serious burns: a clue to the alterations of magnesium and phosphate metabolism. Am J Clin Nutr 65:1473–1481
Polderman KH, Bloemers FW, Peerdeman SM, Girbes AR (2000) Hypomagnesemia and hypophosphatemia at admission in patients with severe head injury. Crit Care Med 28:2022–2025
Buell JF, Berger AC, Plotkin JS, Kuo PC, Johnson LB (1998) The clinical implications of hypophosphatemia following major hepatic resection or cryosurgery. Arch Surg 133:757–761
Cohen J, Kogan A, Sahar G, Lev S, Vidne B, Singer P (2004) Hypophosphatemia following open heart surgery: incidence and consequences. Eur J Cardiothorac Surg 26:306–310
Salem RR, Tray K (2005) Hepatic resection-related hypophosphatemia is of renal origin as manifested by isolated hyperphosphaturia. Ann Surg 241:343–348
Morimatsu H, Uchino S, Bellomo R, Ronco C (2002) Continuous veno-venous hemodiafiltration or hemofiltration: impact on calcium, phosphate and magnesium concentrations. Int J Artif Organs 25:512–519
Song YH, Seo EH, Yoo YS, Jo YI (2019) Phosphate supplementation for hypophosphatemia during continuous renal replacement therapy in adults. Ren Fail 41:72–79
Sun Z, Ye H, Shen X, Chao H, Wu X, Yang J (2014) Continuous venovenous hemofiltration versus extended daily hemofiltration in patients with septic acute kidney injury: a retrospective cohort study. Crit Care 18:R70
Albino BB, Balbi AL, Abrão JM, Ponce D (2015) Dialysis complications in acute kidney injury patients treated with prolonged intermittent renal replacement therapy sessions lasting 10 versus 6 hours: results of a randomized clinical trial. Artif Organs 39:423–431
Terlevich A, Hearing SD, Woltersdorf WW, Smyth C, Reid D, McCullagh E, Day A, Probert CS (2003) Refeeding syndrome: effective and safe treatment with phosphates polyfusor. Aliment Pharmacol Ther 17:1325–1329
Chanchal R, Gupta S, Kanta C, Singh K, Koonwar S (2018) Hypophosphataemia in severe acute malnutrition: a prospective observational study. Br J Nutr 17:1–6
Bollaert PE, Levy B, Nace L, Laterre PF, Larcan A (1995) Hemodynamic and metabolic effects of rapid correction of hypophosphatemia in patients with septic shock. Chest 107:1698–1701
Sykes L, Nipah R, Kalra P, Green D (2018) A narrative review of the impact of interventions in acute kidney injury. J Nephrol 31:523–535
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group (2012) KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl S2:1–138
Pistolesi V, Di Napoli A, Fiaccadori E, Zeppilli L, Polistena F, Sacco MI, Regolisti G, Tritapepe L, Pierucci A, Morabito S (2016) Severe acute kidney injury following cardiac surgery: short-term outcomes in patients undergoing continuous renal replacement therapy (CRRT). J Nephrol 29:229–239
Ratanarat R, Brendolan A, Volker G, Bonello M, Salvatori G, Andrikos E, Yavuz A, Crepaldi C, Ronco C (2005) Phosphate kinetics during different dialysis modalities. Blood Purif 23:83–90
Schwartz A, Brotfain E, Koyfman L, Kutz R, Gruenbaum SE, Klein M, Zlotnik A (2014) Association between hypophosphatemia and cardiac arrhythmias in the early stage of sepsis: could phosphorus replacement treatment reduce the incidence of arrhythmias? Electrolyte Blood Press 12:19–25
Ariyoshi N, Nogi M, Ando A, Watanabe H, Umekawa S (2017) Cardiovascular consequences of hypophosphatemia. Panminerva Med 59:230–240
Chanutin A, Hermann E (1969) The interaction of organic and inorganic phosphates with hemoglobin. Arch Biochem Biophys 131:180–184
Lichtman MA, Miller DR, Cohen J, Waterhouse C (1971) Reduced red cell glycolysis, 2, 3-diphosphoglycerate and adenosine triphosphate concentration, and increased hemoglobin-oxygen affinity caused by hypophosphatemia. Ann Intern Med 74:562–568
Sharma S, Brugnara C, Betensky RA, Waikar SS (2015) Reductions in red blood cell 2,3 diphosphoglycerate concentration during continuous renal replacement therapy. Clin J Am Soc Nephrol 10:74–79
Rosen GH, Boullata JI, O’Rangers EA, Enow NB, Shin B (1995) Intravenous phosphate repletion regimen for critically ill patients with moderate hypophosphatemia. Crit Care Med 23:1204–1210
Shajahan A, Ajith Kumar J, Gireesh Kumar KP, Sreekrishnan TP, Jismy K (2015) Managing hypophosphatemia in critically ill patients: a report on an under-diagnosed electrolyte anomaly. J Clin Pharm Ther 40:353–354
Engwerda E, Van den Berg M, Blans M, Bech A, De Boer H (2018) Efficacy and safety of a phosphate replacement strategy for severe hypophosphatemia in the ICU. Neth J Med 76:437–441
Chow MT, Lin HJ, Mitra EA, Singh S, Lee E, Leehey DJ, Ing TS (1998) Hemodialysis-induced hypophosphatemia in a normophosphatemic patient dialyzed for ethylene glycol poisoning: treatment with phosphorus-enriched hemodialysis. Artif Organs 22:905–907
Dorval M, Pichette V, Cardinal J, Geadah D, Ouimet D, Leblanc M (1999) The use of an ethanol- and phosphate-enriched dialysate to maintain stable serum ethanol levels during haemodialysis for methanol intoxication. Nephrol Dial Transplant 14:1774–1777
Santiago MJ, López-Herce J, Muñoz R, del Castillo J, Urbano J, Solana MJ, Botrán M (2011) Stability of continuous renal replacement therapy solutions after phosphate addition: an experimental study. Ther Apher Dial 15:75–80
Heung M, Mueller BA (2018) Prevention of hypophosphatemia during continuous renal replacement therapy—an overlooked problem. Semin Dial 31:213–218
Shaw AR, Chaijamorn W, Clark JS, Mueller BA (2018) Preparation times and costs for various solutions used for continuous renal replacement therapy. Am J Health Syst Pharm 75:808–815
Author information
Authors and Affiliations
Contributions
VP, LZ and SM had the idea for the review, performed the literature search and data analysis, and drafted the work. EF, GR and LT critically revised the work.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pistolesi, V., Zeppilli, L., Fiaccadori, E. et al. Hypophosphatemia in critically ill patients with acute kidney injury on renal replacement therapies. J Nephrol 32, 895–908 (2019). https://doi.org/10.1007/s40620-019-00648-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-019-00648-5