Abstract
Purpose
Multiple factors influence intrauterine growth and lead to low birth sizes. The impact of genetic alterations on both pre- and post-natal growth is still largely unknown. The aim of this study was to investigate the prevalence of CNVs in an Italian cohort of SGA children with persistent short stature and complex clinical phenotype. rhGH treatment efficacy was evaluated according to the different genotypes.
Subjects and methods
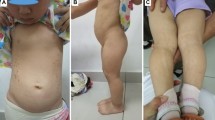
Twenty-four SGA children (10F/14M) with persistent short stature associated with dysmorphic features and/or developmental delay underwent CNV evaluation.
Results
CNVs were present in 14/24 (58%) SGA children. Six patients had a microdeletion involving the following regions: 3q24q25.1, 8p21.2p12, 15q26, 19q13.11, 20q11.21q12, 22q11.2. In three females, the same microdeletion involving 17p13.3 region was identified. In two different patients, two microduplications involving 10q21.3 and Xp11.3 region were observed. A further female patient showed both an 11q12.1 and an Xq27.1 microduplication, inherited from her mother and from her father, respectively. In a boy, the presence of a 12p13.33 microdeletion and a 19q13.43 microduplication was found. GH treatment efficacy, expressed by height gain and height velocity in the first 12 months of therapy, was similar in subjects with and without CNVs.
Conclusions
These results show that pathogenic CNVs are common in SGA children with short stature associated with additional clinical features. Interestingly, the involvement of 17p13.3 region occurs with a relative high frequency, suggesting that genes located in this region could play a key role in pre- and post-natal growth. rhGH therapy has similar efficacy in the short term whether CNVs are present or not.


Similar content being viewed by others
References
van der Steen M, Hokken-Koelega AC (2016) Growth and metabolism in children born small for gestational age. Endocrinol Metab Clin N Am 45:283–294. https://doi.org/10.1016/j.ecl.2016.01.008
Lee PA, Chernausek SD, Hokken-Koelega AC, Czernichow P (2003) International small for gestational age advisory board consensus development conference statement: management of short children born small for gestational age, April 24-October 1, 2001. Pediatrics 111:1253–1261. https://doi.org/10.1542/peds.111.6.1253
Clayton PE, Cianfarani S, Czernichow P, Johannsson G, Rapaport R, Rogol A (2007) Management of the child born small for gestational age through to adulthood: a consensus statement of the International Societies of Pediatric Endocrinology and the Growth Hormone Research Society. J Clin Endocrinol Metab 92:804–810. https://doi.org/10.1210/jc.2006-2017
Van Pareren Y, Mulder P, Houdijk M, Jansen M, Reeser M, Hokken-Koelega A (2003) Adult height after long-term, continuous growth hormone (GH) treatment in short children born small for gestational age: results of a randomized, double-blind, dose-response GH trial. J Clin Endocrinol Metab 88:3584–3590. https://doi.org/10.1210/jc.2002-021172
Lunde A, Melve KK, Gjessing HK, Skjaerven R, Irgens LM (2007) Genetic and environmental influences on birth weight, birth length, head circumference, and gestational age by use of population-based parent-offspring data. Am J Epidemiol 165:734–741. https://doi.org/10.1093/aje/kwk107
Stalman SE, Solanky N, Ishida M, Aleman-Charlet C, Abu-Amero S, Alders M et al (2018) Genetic analyses in small-for-gestational-age newborns. J Clin Endocrinol Metab 103:917–925. https://doi.org/10.1210/jc.2017-01843
Alkan C, Coe BP, Eichler EE (2011) Genome structural variation discovery and genotyping. Nat Rev Genet 12:363–376. https://doi.org/10.1038/nrg2958
Weise A, Mrasek K, Klein E, Mulatinho M, Llerena JC Jr, Hardekopf D et al (2012) Microdeletion and microduplication syndromes. J Histochem Cytochem 60:346–358. https://doi.org/10.1369/0022155412440001
Inzaghi E, Reiter E, Cianfarani S (2019) The challenge of defining and investigating the causes of idiopathic short stature and finding an effective therapy. Horm Res Paediatr 92:71–83. https://doi.org/10.1159/000502901
Seaver LH, Irons M (2009) ACMG practice guideline: genetic evaluation of short stature. Genet Med 11:465–470. https://doi.org/10.1097/GIM.0b013e3181a7e8f8
Miller DT, Adam MP, Aradhya S, Biesecker LG, Brothman AR, Carter NP et al (2010) Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet 86:749–764. https://doi.org/10.1016/j.ajhg.2010.04.006
Homma TK, Krepischi ACV, Furuya TK, Honjo RS, Malaquias AC, Bertola DR et al (2018) Recurrent copy number variants associated with syndromic short stature of unknown cause. Horm Res Paediatr 89:13–21. https://doi.org/10.1159/000481777
Wit JM, van Duyvenvoorde HA, van Klinken JB, Caliebe J, Bosch CA, Lui JC et al (2014) Copy number variants in short children born small for gestational age. Horm Res Paediatr 82:310–318. https://doi.org/10.1159/000367712
Kearney HM, Thorland EC, Brown KK, Quintero-Rivera F, South ST (2011) American College of Medical Genetics standards and guidelines for interpretation and reporting of postnatal constitutional copy number variants. Genet Med 13:680–685. https://doi.org/10.1097/GIM.0b013e3182217a3a
Canton AP, Costa SS, Rodrigues TC, Bertola DR, Malaquias AC, Correa FA et al (2014) Genome-wide screening of copy number variants in children born small for gestational age reveals several candidate genes involved in growth pathways. Eur J Endocrinol 171:253–262. https://doi.org/10.1530/EJE-14-0232
Bruno DL, Anderlid BM, Lindstrand A, van Ravenswaaij-Arts C, Ganesamoorthy D, Lundin J et al (2010) Further molecular and clinical delineation of co-locating 17p13.3 microdeletions and microduplications that show distinctive phenotypes. J Med Genet 47:299–311. https://doi.org/10.1136/jmg.2009.069906
Østergaard JR, Graakjær J, Brandt C, Birkebæk NH (2012) Further delineation of 17p13.3 microdeletion involving CRK. The effect of growth hormone treatment. Eur J Med Genet 55:22–26. https://doi.org/10.1016/j.ejmg.2011.09.004
Hirasawa-Inoue A, Takeshita E, Shimizu-Motohashi Y, Ishiyama A, Saito T, Komaki H et al (2019) Static leukoencephalopathy associated with 17p13.3 microdeletion syndrome: a case report. Neuropediatrics 50:387–390. https://doi.org/10.1055/s-0039-1693972
Veenma DC, Eussen HJ, Govaerts LC, de Kort SW, Odink RJ, Wouters CH et al (2010) Phenotype-genotype correlation in a familial IGF1R microdeletion case. J Med Genet 47:492–498. https://doi.org/10.1136/jmg.2009.070730
Forzano F, Napoli F, Uliana V, Malacarne M, Viaggi C, Bloise R et al (2012) 19q13 microdeletion syndrome: further refining the critical region. Eur J Med Genet 55:429–432. https://doi.org/10.1016/j.ejmg.2012.03.002
Loddo S, Alesi V, Genovese S, Orlando V, Calacci C, Restaldi F et al (2018) First report of low-rate mosaicism for 20q11.21q12 deletion and delineation of the associated disorder. Cytogenet Genome Res. https://doi.org/10.1159/000493935
Mikhail FM, Burnside RD, Rush B, Ibrahim J, Godshalk R, Rutledge SL et al (2014) The recurrent distal 22q11.2 microdeletions are often de novo and do not represent a single clinical entity: a proposed categorization system. Genet Med 16:92–100. https://doi.org/10.1038/gim.2013.79
Ben-Shachar S, Ou Z, Shaw CA, Belmont JW, Patel MS, Hummel M et al (2008) 22q11.2 distal deletion: a recurrent genomic disorder distinct from DiGeorge syndrome and velocardiofacial syndrome. Am J Hum Genet 82:214–221. https://doi.org/10.1016/j.ajhg.2007.09.014
Ma S, Tang KH, Chan YP, Lee TK, Kwan PS, Castilho A et al (2010) miR-130b Promotes CD133(+) liver tumor-initiating cell growth and self-renewal via tumor protein 53-induced nuclear protein 1. Cell Stem Cell 7:694–707. https://doi.org/10.1016/j.stem.2010.11.010
Bengoa-Alonso A, Artigas-López M, Moreno-Igoa M, Cattalli C, Hernández-Charro B, Ramos-Arroyo MA (2016) Delineation of a recognizable phenotype for the recurrent LCR22-C to D/E atypical 22q11.2 deletion. Am J Med Genet A 170:1485–1494. https://doi.org/10.1002/ajmg.a.37614
Hori O, Miyazaki M, Tamatani T, Ozawa K, Takano K, Okabe M et al (2006) Deletion of SERP1/RAMP4, a component of the endoplasmic reticulum (ER) translocation sites, leads to ER stress. Mol Cell Biol 26:4257–4267. https://doi.org/10.1128/MCB.02055-05
Rooryck C, Stef M, Burgelin I, Simon D, Souakri N, Thambo JB et al (2009) 2.3 Mb terminal deletion in 12p13.33 associated with oculoauriculovertebral spectrum and evaluation of WNT5B as a candidate gene. Eur J Med Genet 52:446–449. https://doi.org/10.1016/j.ejmg.2009.08.005
Abdelmoity AT, Hall JJ, Bittel DC, Yu S (2011) 1.39 Mb inherited interstitial deletion in 12p13.33 associated with developmental delay. Eur J Med Genet 54:198–203. https://doi.org/10.1016/j.ejmg.2010.11.010
Sauter SM, Böhm D, Bartels I, Burfeind P, Laccone FA, Neesen J et al (2007) Partial trisomy of distal 19q detected by quantitative real-time PCR and FISH in a girl with mild facial dysmorphism, hypotonia and developmental delay. Am J Med Genet A 143a:1091–1099. https://doi.org/10.1002/ajmg.a.31686
Tsukahara M, Murano I, Aoki Y, Kajii T, Furukawa S (1995) Interstitial deletion of 8p: report of two patients and review of the literature. Clin Genet 48:41–45. https://doi.org/10.1111/j.1399-0004.1995.tb04052.x
Klopocki E, Fiebig B, Robinson P, Tönnies H, Erdogan F, Ropers HH et al (2006) A novel 8 Mb interstitial deletion of chromosome 8p12-p21.2. Am J Med Genet A 140:873–877. https://doi.org/10.1002/ajmg.a.31163
Willemsen MH, de Leeuw N, Pfundt R, de Vries BB, Kleefstra T (2009) Clinical and molecular characterization of two patients with a 6.75 Mb overlapping deletion in 8p12p21 with two candidate loci for congenital heart defects. Eur J Med Genet 52:134–139. https://doi.org/10.1016/j.ejmg.2009.03.003
Arghir A, Papuc SM, Tutulan-Cunita AC, Erbescu A, Loddo S, Genovese S et al (2020) Autism and severe clinical phenotype in a patient with 8p21.2p11.21 deletion: case report and literature review. Clin Case Rep 9:314–321. https://doi.org/10.1002/ccr3.3523
Nagamani SC, Erez A, Bader P, Lalani SR, Scott DA, Scaglia F et al (2011) Phenotypic manifestations of copy number variation in chromosome 16p13.11. Eur J Hum Genet 19:280–286. https://doi.org/10.1038/ejhg.2010.184
Redaelli S, Maitz S, Crosti F, Sala E, Villa N, Spaccini L et al (2019) Refining the phenotype of recurrent rearrangements of chromosome 16. Int J Mol Sci. https://doi.org/10.3390/ijms20051095
Jung H, Rosilio M, Blum WF, Drop SL (2008) Growth hormone treatment for short stature in children born small for gestational age. Adv Ther 25:951–978. https://doi.org/10.1007/s12325-008-0101-3
Funding
This work was supported by Ministero della Salute grant, 2019: RRC-2019-2366867.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Appropriate informed consent was acquired from the participating subject.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Inzaghi, E., Deodati, A., Loddo, S. et al. Prevalence of copy number variants (CNVs) and rhGH treatment efficacy in an Italian cohort of children born small for gestational age (SGA) with persistent short stature associated with a complex clinical phenotype. J Endocrinol Invest 45, 79–87 (2022). https://doi.org/10.1007/s40618-021-01617-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-021-01617-1