Abstract
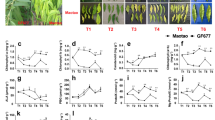
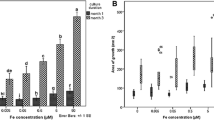
The present study demonstrates that chlorophyll biosynthesis can be effectively used for identifying iron (Fe) deficiency tolerant cultivars from a large population. Fifty genetically diverse wheat cultivars were screened on the basis of the leaf greenness index, i.e. chlorophyll retention under iron deficient (Fe−) compared to iron sufficient (Fe+) conditions, and three Fe deficiency tolerant and two Fe deficiency susceptible cultivars were identified. These two groups of cultivars were further tested for their efficiency for Fe uptake and translocation and biomass production per unit Fe under Fe deficient condition in the nutrient solution culture. Fe deficiency tolerant lines were found capable of translocating more Fe to the shoot and produced greater biomass per unit of Fe when compared with the susceptible group. Efficiency for chlorophyll biosynthesis per unit shoot Fe was also higher for the tolerant group of cultivars. Fe use efficient cultivars produced and released a larger amount of phytosiderophore (PS) and differed from the inefficient cultivars in terms of the PS release but not in the PS biosynthesis. Thus, indicating that the limitation at the level of release of the PS was responsible for low Fe use efficiency of the Fe deficiency susceptible cultivars. Further the diurnal variation in the PS release was similar for all the investigated wheat cultivars and did not influence the genotypic variation in the Fe use efficiency.






Similar content being viewed by others
References
Aciksoz, B. S., Ozturk, L., Gokmen, O. O., Romheld, V., & Cakmak, I. (2011). Effect of nitrogen on root release of phytosiderophores and root uptake of Fe(III) phytosiderophore in Fe-deficient wheat plant. Physiologia Plantarum, 142, 287–296.
Bhargava, B. S., & Raghupathi, H. B. (1993). Analysis of plant materials for macro and micrnutrients. In H. L. S. Tandon (Ed.), Methods of analysis of soils, plants, water and fertilizers (pp. 49–82). New Delhi: Fertilization Department Consultant Organization.
Brian, M. W., & Grace, C. T. (2012). Natural variation in iron use efficiency and mineral remobilization in cucumber (Cucumis sativus). Plant and Soil, 352, 185–197.
Cakmak, I. (2008). Enrichment of cereal grains with Zinc: agronomic or genetic biofortification? Plant and Soil, 302, 1–1.
Chaignon, V., Malta, D., & Hinsinger, P. (2002). Fe-deficiency increases Cu acquisition by wheat cropped in a Cu contaminated vineyard soil. New Phytologist, 154, 121–130.
Crowley, D. E., Wu, C. L., Gries, D., Brunn, S., & Parker, D. R. (2002). Quantitative traits associated with adaptation of three barley (H. vulgare L.) cultivars to suboptimal iron supply. Plant soil, 241, 57–65.
Dunisijević, B. D., Dukić, M., Maksimović, V., Skočajić, D., & Suručić, L. (2012). The effects of iron deficiency on lead accumulation in Ailanthus altissima (Mill.) Swingle seedlings. Journal of Environmental Quality, 41(5), 1517–1524.
Gries, D., Brunn, S., Crowley, D. E., & Parker, D. R. (1995). Phytosiderophore release in relation to metal micronutrient deficiencies in barley. Plant and Soil, 172, 299–308.
Hansen, N. C., Jolley, V. D., Naeve, S. L., & Goos, R. J. (2004). Iron deficiency of soybean in the North Central US and associated soil properties. Soil Science and Plant Nutrition, 50(7), 983–987.
Kawai, S., Takagi, S., & Sato, Y. (1988). Mugineic acid-family phytosiderophores in root-secretions of barley, corn and sorghum varieties. Journal of Plant Nutrition, 11, 633–642.
Krouma, A., & Abdelly, C. (2003). Importance of iron use-efficiency of nodules in common bean (Phaseolus vulgaris L.) for iron deficiency chlorosis resistance. Journal of Plant Nutrition and Soil Science, 166(4), 1–4.
Ma, J. F., Ueno, H., Ueno, D., Rambola, A. D., & Iwashita, T. (2003). Characterization of phytosiderophore secretion under Fe deficiency stress in Festuca rubra. Plant and Soil, 256, 131–137.
Mahmoudi, H., Labidi, N., Ksouri, R., Gharsalli, M., & Abdelly, C. (2007). Differential tolerance to iron deficiency of chickpea varieties and Fe resupply effects. Comptes Rendus Biologies, 330, 237–246.
Marschner H (2011) Marschner’s Mineral Nutrition of Higher Plants, 3rd Edition. Petra Marschner (Ed). Academic Press, p 672.
Masuda, H., Suzuki, M., Morikawa, K. C., Kobayashi, T., Nakanishi, H., Takahashi, M., et al. (2008). Increase in iron and zinc concentrations in rice grains via the introduction of barley genes involved in phytosiderophore synthesis. Rice, 1, 100–108.
Meda, A. R., Scheuermann, E. B., Prechsl, U. E., Erenoglu, B. L., Schaaf, G., & von Wiren, N. (2007). Iron acquisition by phytosiderophores contributes to cadmium tolerance. Plant Physiology, 143, 1761–1773.
Mohammad, M. J., Najim, H., & Khresat, S. (1998). Nitric acid- and o-phenanthroline-extractable iron for diagnosis of iron chlorosis in citrus lemon trees. Communications in Soil Science and Plant Analysis, 29, 1035–1043.
Nagasaka, S., Takahashi, M., Nakanishi-Itai, R., Bashir, K., Nakanishi, H., Mori, S., et al. (2009). Time course analysis of gene expression over 24 hours in Fe-deficient barley roots. Plant Molecular Biology, 69, 621–631.
Neaman, A., & Aguirre, L. (2007). Comparison of different methods for diagnosis of iron deficiency in Avocado. Journal of Plant Nutrition, 30, 1097–1108.
Neelam, K., Tiwari, K. V., Rawat, N., Tripathi, K. S., Randhawa, S. G., & Dhaliwal, S. H. (2010). Identification of Aegilopsspecies with higher production of phytosiderophore and iron and zinc uptake under micronutrient-sufficient and -deficient conditions. Plant Genetic Resources: Characterization and Utilization, 8(2), 132–141.
Nozoye, T., Nakanishi, H., & Nishizawa, N. K. (2013). Characterizing the crucial components of iron homeostasis in the maize mutants ys1 and ys3. PLoS one, 8(5), e62567.
Pierson, E. E., Clark, R. B., Maranville, J. W., & Coyne, D. P. (1984). Plant genotype differences to ferrous and total iron in emerging leaves. I. Sorghum and maize. Journal of Plant Nutrition, 7(1–5), 371–385.
Römheld, V., & Marschner, H. (1990). Genotypical differences among graminaceous species in release of phytosiderophores and uptake of iron phytosiderophores. Plant and Soil, 123, 147–153.
Ruel, M. T., & Bouis, H. E. (1998). Plant breeding: a long–term strategy for the control of zinc deficiency in vulnerable populations. The American Journal of Clinical Nutrition, 68(2), 488S–494S.
Shen, J., Zhang, F., Chen, Q., Rengel, Z., Tang, C., & Song, C. (2002). Genotypic difference in seed iron content and early responses to iron deficiency in wheat. Journal of Plant Nutrition, 25(8), 1631–1643.
Singh, B., Kumar, S., & Singh, B. K. (2006). Phytosiderophore production and its release is related to Zn efficiency of wheat under Zn deficiency. In K. A. Nayeem (Ed.), Wheat for tropical areas (pp. 247–252). New Delhi: IARI Publication.
Suzuki, M., Takahashi, M., Tsukamoto, T., Watanabe, S., Matsuhashi, S., Yazaki, J., et al. (2006). Biosynthesis and secretion of mugineic acid family phytosiderophores in zinc-deficient barley. The Plant Journal, 48, 85–97.
Takagi, S. (1976). Naturally occurring Iron-chelating compound in oat and rice root washings. Soil Science and Plant Nutrition, 22, 423–433.
Ueno, D., Yamaji, N., & Ma, J. F. (2009). Future characterization of ferric–phytosiderophore transporters zmys1 and hvys1 in maize and barley. Journal of experimental botany, 60(12), 3513–3520.
Yousfi, S., Rabhi, M., Abdelly, C., & Gharsalli, M. (2009). Iron deficiency tolerance traits in wild (Hordium maritimum) and cultivated barley (Hordeum vulgare). Comptes Rendus Biologies, 332, 523–533.
Zhang, F., Romheld, V., & Marschner, H. (1991). Diurnal rhythm of release of phytosiderophores and uptake rate of zinc in iron deficient wheat. Soil Science and Plant Nutrition, 37(4), 671–678.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khobra, R., Ahuja, S. & Singh, B. Chlorophyll biosynthesis as the basis of iron use efficiency under iron deficiency and its relationship with the phytosiderophore synthesis and release in wheat. Ind J Plant Physiol. 19, 330–337 (2014). https://doi.org/10.1007/s40502-014-0112-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-014-0112-9