Abstract
Making a sizeable contribution to worldwide cancer mortality, adenocarcinomas and squamous cell carcinomas of the esophagus appear to be caused largely by environmental factors. Descriptive epidemiologic analyses demonstrate that these cancers occur at markedly different rates in populations around the world. Moreover, their incidences have changed greatly over time, strongly suggesting that their causes are modifiable. For adenocarcinomas, the risk factors identified consistently in analytic studies include gastroesophageal acid reflux, obesity, smoking, and male sex. Squamous cell carcinomas in Western countries have been attributed largely to smoking, high levels of alcohol consumption, and poor nutrition. In other parts of the world, poor-quality diets, thermal injury, exposure to polycyclic aromatic hydrocarbons, and poor oral health have been implicated. Prevention strategies targeting these exposures offer a logical foundation for action, although the outcomes of prevention trials have been frustratingly ineffective to date. Novel approaches to early detection are currently under development and may prove useful for disease control in high-incidence settings.
Similar content being viewed by others
Introduction
Cancers of the esophagus are the eighth most commonly occurring cancers worldwide and the sixth most common cause of cancer mortality, accounting for an estimated 400,000 deaths each year [1]. Two main types of esophageal cancer are recognized histologically—namely, esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC). This review discusses the key epidemiologic features of each cancer, briefly outlines the risk factors for both types, and assesses opportunities for prevention in the broader sense, including strategies directed toward primary prevention and early detection.
Descriptive Epidemiology of Esophageal Cancer
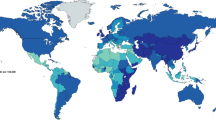
The incidences and relative proportions of ESCC and EAC vary enormously across populations (see Fig. 1a, b). In most regions, ESCC accounts for the vast majority of all diagnoses of esophageal cancer (Fig. 1a); however, in a relatively small number of industrialized countries of predominantly European heritage, the numbers of cases of EAC now exceed the numbers of ESCC cases. In most populations, the combined incidences of cancers of the esophagus are between 4 and 8 cases per 100,000 per year, but much higher rates are observed in some populations (Fig. 1b). Two belts of particularly high incidence are recognized, one following the old Silk Road from northern and central China, across central Asia and into northeastern Iran, the other extending in a broad swath from eastern to southern Africa. While a number of environmental factors have been causally associated with each type of esophageal cancer and likely explain much of the variation in type-specific incidence across populations, marked differences in EAC and ESCC incidence rates have been observed between white, black, Asian, and Hispanic populations within the USA (Fig. 2), suggesting that heritable factors may also be at play.
Frequencies of esophageal cancers in different populations: a relative proportions; b age-standardized incidence of esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC) in populations from Oceania, Asia, Europe, and North and South America [GLOBOCAN, International Agency for Research on Cancer (IARC) data]
It is important to note that the relative excess of EAC cases in industrialized populations is a recent phenomenon. Prior to about 1970, EAC was a rare cancer, but then the incidence of EAC began to climb in several countries around the world, including the USA, the UK, and Australia, and in Scandinavia. Even so, it was not until the mid- to late 1990s that the numbers of EAC cases exceeded the numbers of ESCC cases. In most of these populations, the rate of the increase in the incidence of EAC was greater than for any other major cancer, averaging about 7 % per year [2]. While some reports suggest that the incidence may now be stabilizing [3, 4], others suggest that the rates are continuing to rise and speculate that birth cohort effects will continue to drive incidence rises in the foreseeable future [5].
In Western populations, esophageal cancers tend to occur most commonly in the sixth and seventh decades, with a median age of diagnosis of 67 years in the US population [6]. Both types of esophageal cancer are more common in men than in women in Western populations; for EAC, the sex ratio is very high, averaging around 5:1 in most registries that report type-specific data.
Risk Factors for Cancers of the Esophagus
Esophageal Adenocarcinoma
Established risk factors for EAC include frequent gastroesophageal acid reflux, obesity, the presence of hiatus hernia, and smoking; factors that have been inversely associated with EAC include frequent use of aspirin or nonsteroidal anti-inflammatory drugs (NSAIDs) and infection with Helicobacter pylori. The evidence underpinning these associations is reviewed briefly below. A host of other factors have been investigated as potentially conferring increased risks of EAC, including poor diet, low or high intakes of specific nutrients, alcohol intake, and various medications that might promote reflux, with inconsistent findings overall.
Gastroesophageal Acid Reflux
Clinicians have long observed that patients with EAC typically report a history of frequent and severe acid regurgitation, and this impression has been confirmed subsequently in epidemiologic studies using various methods for assessing past history of reflux [7–12]. The chronic inflammatory state induced by repeated exposure to gastric acid is assumed to establish a microenvironment with a high turnover of basal epithelial cells, coupled with a propensity for mutation through generation of reactive oxygen species and other molecular pathways [13, 14]. The low-pH environment is also hypothesized to promote the production of mutagens from exogenous and endogenous contents of the esophagus (such as nitrosamines from saliva [15–17]). Conditions that predispose to gastroesophageal acid reflux—notably hiatus hernia—have also been strongly associated with the risk of EAC [10].
Barrett’s Esophagus
A small proportion of patients with chronic reflux develop a metaplastic columnar epithelial lining in the distal esophagus, known as Barrett’s esophagus, which is widely regarded as a precursor lesion to adenocarcinoma [18]. A detailed description of Barrett’s esophagus and its malignant potential is beyond the scope of this review; suffice it to say that identifying patients with Barrett’s esophagus has been the principal preventive strategy for controlling morbidity and mortality from EAC (see below).
Obesity
Epidemiologic studies of case–control [9, 11, 19–21] and cohort designs [22, 23] have all reported strong, positive associations between measures of body size and the risk of EAC. A recent pooled analysis reported 54 % and 139 % increased relative risks of EAC in people who were overweight [body mass index (BMI) 25–29.0] or obese (BMI 30.0–34.9), respectively, compared with those in the healthy weight range (BMI <25). The risks increased monotonically with increasing levels of obesity, such that those with BMI values >40 had more than 350 % increased relative risks of EAC [24]. Several studies have reported synergistic interactions whereby obese people who also suffer frequent reflux have greatly increased risks of EAC, compared with obese people who are free of reflux [11, 25]. Most infer a causal association, for which the favored explanation currently is that male-pattern obesity (abdominal or visceral adiposity) has dual effects on raising the risk of EAC. First, there are the mechanical effects of increasing the pressure on the esophagogastric sphincter, thereby promoting reflux of gastric acid into the esophagus [26–28]. Second, the visceral fat deposits that predominate in men are metabolically active—more so than the subcutaneous fat deposits that tend to accumulate preferentially in women [29]. It is hypothesized that the metabolic disturbances engendered by visceral fat [including insulin resistance and increased production of adipocytokines and insulin-like growth factors (IGFs)], may promote tumor development through their anti-apoptotic and pro-proliferative effects [13, 30, 31].
Smoking
Smoking is a cause of both histologic subtypes of esophageal cancer [32], although studies consistently report stronger associations with ESCC (up to 4-fold increased risks) than with EAC (approximate 2-fold increased risks). For example, a recent pooled analysis of ten epidemiologic studies reported a summary odds ratio (OR) of 1.96 (95 % CI 1.64–2.34) for ever- versus never-smoking, with a trend of increasing risks with increasing cumulative exposure to tobacco smoke [33]. Among ever-smokers, those who had quit for more than 10 years had significantly reduced risks of EAC (0.71, 95 % CI 0.56–0.89), although the risks for this group remained 72 % higher than those for never-smokers. The mechanisms through which smoking may cause EAC are many and include directly genotoxicity to esophageal cells, promotion of gastroesophageal reflux, or the presence of hazardous compounds, such as polycyclic aromatic hydrocarbons (PAHs) and nitrosamines [32].
Medications
Given the strong positive associations between gastroesophageal acid reflux and EAC, there has been speculation that drugs designed specifically to reduce gastric acid secretion [such as histamine (H2) receptor antagonists and proton pump inhibitors (PPIs)] should reduce the risk of EAC. A competing hypothesis has proposed that the risks of cancer may be increased by bacterial overgrowth in the achlorhydric stomach, leading to production of carcinogenic nitrosamines. While there is some evidence of increased bacterial loads in long-term users of acid-suppressant medications [34], there is no evidence to suggest increased risks of EAC via this mechanism. A recent meta-analysis of seven observational studies reported a 71 % reduction in the risk of EAC or high-grade dysplasia among people with Barrett’s esophagus treated with PPIs, compared with those not receiving PPIs [35•]. That said, it is extraordinarily difficult to untangle medicinal effects through observational studies because of confounding by indication, hence the outcomes of chemoprevention trials are keenly awaited [36, 37].
Infection
H. pylori is a species of Gram-negative bacteria that colonizes the human stomach. Conclusively established as a cause of non-cardia gastric cancers [38–40], this organism has been associated with reduced risks of EAC. A recent meta-analysis identified 19 studies that assessed the association between past infection with H. pylori and the risk of EAC through serologic analyses, generating a summary risk estimate of 0.56 (95 % CI 0.46–0.68) [41]. The Cag-A+ strains of H. pylori are associated with higher pathogenicity for gastric cancer; the small number of studies that have assessed strain-specific associations for EAC suggest strong inverse associations with Cag-A+ infection (summary OR 0.41, 95 % CI 0.28–0.62) but no association with Cag-A− infections (summary OR 1.08, 95 % CI 0.76–1.53) [41]. Several mechanisms have been proposed for the apparent protective effect of H. pylori on esophageal neoplasia, including reduced production of gastric acid and ghrelin [42].
Nonsteroidal Anti-inflammatory Drugs
NSAIDs are a broad class of drugs that inhibit one or more cyclooxygenase enzymes. These enzymes are central to many inflammatory pathways, and there has been long-standing interest in their potential for preventing the development of epithelial tumors, particularly in the gastrointestinal tract [43, 44]. A recent pooled analysis of six case–control studies reported reduced risks of EAC associated with ever-use of aspirin or NSAIDs (summary OR 0.68, 95 % CI 0.56–0.83) [45•]. Larger risk reductions were observed among daily users (summary OR 0.56, 95 % CI 0.43–0.73). Because of concerns about reverse-causality bias, in which inverse associations might be induced spuriously because patients with symptomatic reflux might avoid using NSAIDs, findings from observational studies have been interpreted cautiously. Long-term follow-up of aspirin trials also suggests reductions in the risk of EAC, however, lending credence to the observational findings [46••]. A large chemoprevention trial is under way to test this hypothesis formally [37].
Dietary Factors
The majority of studies investigating associations between diet and EAC have been case–control designs, raising concerns regarding systematic misclassification, since the disease process is likely to induce dietary changes in the prediagnostic period. While relatively few prospective studies have been powered sufficiently to report on EAC outcomes, recent reports from large cohorts suggest that diets high in fruits and vegetables, or characterized as being “healthy” or “Mediterranean,” confer lower risks of EAC [47–49]. Although the risk reductions were modest and there are acknowledged limitations even for prospective dietary studies [50], these findings provide the strongest evidence to date that diet can shape future risks of EAC.
Alcohol
Many studies have investigated the role of alcohol in EAC development, and most have found no association [51–56]. The most comprehensive analysis to date, combining records from more than 1,800 cancer patients and 10,000 control subjects, derived a summary risk estimate of 0.97 (95 % CI 0.68–1.36) for those who drank more than seven drinks per day, versus none [57•]. There was some evidence that modest consumption of alcohol may be associated with slight reductions in risk, although such findings should be interpreted with caution.
Esophageal Squamous Cell Carcinoma
The patterns of occurrence of ESCC are quite distinctive across populations, likely reflecting distinctive causal pathways operating in different parts of the world. In Western populations, ESCC is associated most strongly with smoking, alcohol, poor diet, and poor oral health. In high-incidence areas of Asia, Africa, and South America, other factors appear to operate, and these are reviewed separately below.
Smoking
Tobacco is recognized as a cause of esophageal cancer by the International Agency for Research on Cancer (IARC). In the most recent IARC monograph [32], 30 cohort studies and 55 case–control studies were reviewed, and all were found to report positive associations with ESCC. Most studies have shown increasing risks with increasing duration and dosage of cigarette smoking, and all studies examining the effects of quitting smoking found reduced cancer risks with time since quitting.
Alcohol
Alcohol is a Group 1 carcinogen and a recognized cause of ESCC [58]. There is strong epidemiologic evidence that the effects of alcohol and smoking are multiplicative on a relative risk scale [59], although alcohol confers increased risks even in the absence of alcohol. Significantly elevated risks among never-smokers have been reported for moderate levels of alcohol intake (1–4 drinks/day; summary OR 1.54) and high levels of alcohol intake (4+ drinks/day; summary OR 3.09) [60]; the risks associated with “light” alcohol consumption (<1 drink/day) suggest no effect on ESCC (summary OR 0.74, 95 % CI 0.47–1.16). The mechanism through which alcohol causes ESCC is not well understood and is probably multifaceted. Alcohol is not directly genotoxic and does not bind to DNA [58]; however, it undergoes metabolism to acetaldehyde, a known mutagen and carcinogen [61, 62]. Moreover, the risks of ESCC are increased among those carrying variants in aldehyde dehydrogenase (ALDH-1), the enzyme responsible for catabolizing acetaldehyde to acetic acid [63]. In addition to these hazardous metabolic properties, alcohol may act as a solvent for other carcinogens and can also lead to nutritional deficiency [58, 61].
Diet
Dietary factors have long been implicated in ESCC development, with postulated mechanisms ranging from deficiencies of specific micronutrients, contamination by carcinogens (e.g., through preparation or storage), or overall poor-quality diet. Despite much research, however, conclusive evidence for any dietary factors as causal (or protective) agents for ESCC has proven disappointingly elusive [64]. In northern and central China, where the rates of ESCC are the highest in the world, observational studies suggested that deficiencies in micronutrients were likely to underlie the burden of ESCC. To test these hypotheses, two large chemoprevention trials were implemented—one in the general population [65] and the other in patients with high-risk precursor lesions [66]—to test a range of nutritional interventions, but they delivered essentially null results [67, 68••]. When these findings were placed alongside null findings from other trials [69, 70], enthusiasm for further nutritional interventions for ESCC was tempered. More recently, a small number of cohort studies conducted in both Western and Chinese populations have reported that diets with high intakes of fruits or vegetables, or with indices of high dietary quality, are associated with reduced risks of ESCC [47–49, 71]. Thus, while no specific micronutrients can be implicated with certainty, there remains considerable scientific interest in the role of dietary factors in ESCC prevention [50].
Maté
An infusion of the herb Ilex paraguayensis¸ maté (also known as yerba maté) is a popular hot beverage in parts of South America, including southern Brazil, northeastern Argentina, Uruguay, and Paraguay. The infusion has been suspected as a cause of ESCC for decades [72]; a recent systematic review identified nine epidemiologic studies testing the hypothesis [73] and reported a summary OR of 2.95 (95 % CI 1.70–5.13) associated with ever- vs never-drinking maté. There are reasonable grounds for inferring a causal association, including consistency of effect, dose response, temporality, and biologic plausibility. The postulated mechanisms for maté carcinogenicity include thermal damage from high-temperature infusions and exposure to PAHs. Very high PAH concentrations have been detected in both hot and cold infusions from commercial brands of yerba maté [74], and maté drinkers have high excretion of urinary PAH [75].
Thermal Injury
Chronic consumption of hot fluids (particularly tea and maté) has been hypothesized to induce thermal injury to the esophageal epithelium [72]. Novel studies in high-incidence regions of northern Iran, incorporating measures of drinking temperatures, have provided the strongest evidence yet that those who drink “hot” or “very hot” tea have significantly higher risks of ESCC (2-fold and 8-fold, respectively) than those who prefer their tea “warm” [76]. Similar associations have been reported recently from studies conducted in high-incidence regions of China [77].
Opium
Consumed rarely in most parts of the world, opium is smoked or ingested by substantial numbers of people in high-incidence regions in northeastern Iran, where exposure to alcohol or tobacco is very low. Case–control studies have suggested approximately 2-fold increased risks of ESCC associated with smoking or ingesting both crude and refined opium, with the relative risks being similarly elevated regardless of whether people also smoked tobacco or not [78]. The mechanisms are unclear, but opium smoke is likely to contain high levels of carcinogenic PAHs [32].
Poor Oral Health
Tooth loss, periodontal disease, and other markers of poor oral hygiene have been associated with increased risks of ESCC and its precursor lesion, esophageal squamous dysplasia (ESD), in many populations [79–81]. Because poor oral health is also associated with other risk factors for ESCC—notably smoking, alcohol, and poor nutrition—confounding is difficult to exclude, even after adjustment. Poor oral hygiene has also been consistently positively associated with ESCC among non-smoking, non-drinking patients in Iran [82], arguing against confounding by those factors in that setting. It is speculated that oral pathogens may underlie this association, producing metabolites such as acetaldehyde, nitrites, and nitrosamines, all of which have been implicated as epithelial carcinogens [83, 84].
Aspirin/Nonsteroidal Anti-inflammatory Drugs
Aspirin and NSAIDs have been associated with reduced risks of epithelial cancers of the gastrointestinal tract, including ESCC. Case–control studies have reported risk reductions for ESCC of up to 40 % among frequent consumers of aspirin and NSAIDs [85, 86], which have been reported similarly in long-term follow-up of prospective studies and clinical trials [46••].
Infection
Human papillomaviruses (HPVs) cause cancers of the anogenital tract and oropharynx, and there is intuitive appeal in the notion that this family of viruses may also cause ESCC. Numerous case–control studies have reported positive associations with ESCC [87, 88]; however, more recent studies conducted under sterile conditions with very careful ascertainment of tumor tissue have failed to replicate those earlier findings [89, 90]. Large-scale serologic analyses pooling data from diverse populations have also failed to demonstrate consistently that cases have higher antibody titers than control subjects [91]. On balance, there is no strong evidence that HPV causes ESCC.
Obesity
In marked contrast to EAC patients, ESCC patients tend to be significantly smaller than control subjects [22, 92, 93]. While reverse causality and residual confounding by smoking are often cited as possible explanations, reduced risks of ESCC with increasing BMI have been reported in analyses restricted to never-smokers [92] and in long-standing cohort studies restricted to people with high self-rated health [93]. In meta-analyses, summary estimates have suggested approximately 25 % lower risks per unit increase in BMI (5 kg/m2) [23]. It is likely that the association is real, but it is too early to infer causality, since a low BMI can also be a marker of poor nutrition, an acknowledged risk factor for ESCC.
Primary Prevention
This brief review has enumerated many factors that are associated with higher or lower risks of EAC and ESCC, many of which are amenable to intervention. For EAC, it seems prudent to recommend smoking abstinence for everyone and weight loss for overweight or obese people. Control of acid reflux through medical or surgical means, for which observational studies suggest benefits in reducing the risk of EAC [94], has clinical appeal and brings symptomatic relief.
For ESCC, it is clear that primary prevention strategies need to be tailored to the attributable fractions in each target population. In Western countries, population-wide abstinence from smoking and alcohol would reduce ESCC incidence substantially—perhaps by as much as 75–90 % [95, 96]. In China, large-scale nutritional interventions have been trialed in high-risk populations, albeit with limited success in terms of reducing incidence or mortality [66, 67]. The interventions to date have been focused on specific micronutrients or food groups, however, as opposed to overall improvement in dietary quality. In Iran, localized interventions to reduce consumption of opium and scalding tea might prove beneficial. In all populations, potentially harmful cooking practices that contaminate food with PAHs should be eliminated, and programs to encourage good oral hygiene and to enhance the availability, pricing, and consumption of fresh fruit and vegetables would have clear health benefits beyond ESCC. Finally, while daily intake of aspirin appears to confer reduced risks of many cancers, including both types of esophageal cancer, there is insufficient evidence that its benefits outweigh its harms to recommend this as a population-wide strategy.
Secondary Prevention
Esophageal Adenocarcinoma
Efforts to define strategies to detect EAC early in its course have been shaped strongly by the paradigm of the inflammation–metaplasia–dysplasia–neoplasia sequence. It is widely believed, although not proven, that most if not all EAC arises from metaplastic Barrett’s epithelium. First described more than 50 years ago, the presence of Barrett’s esophagus has long been known to confer markedly increased risks of EAC. Initial estimates suggested rates of progression as high as 1–4 % per year [97], although with increasing awareness of the diagnosis and the advent of population-based linkage studies, estimates of the rates of progression have been scaled downward [98] toward 0.1 % per year [99•]. In spite of these lower rates of progression, early detection efforts have focused on identifying the pool of patients with Barrett’s esophagus, with the aim of entering them into periodic surveillance. A full discussion of the issues is beyond the scope of this review; however, there are substantial challenges with this approach, due to the so-called Barrett’s paradox [100]. First, Barrett’s esophagus is a reasonably common condition—up to 1 in 50 people may be affected, yet the vast majority are unaware of their diagnosis. Second, up to half of all patients with EAC report no previous symptoms of acid reflux, and more than 90 % of EAC patients have no prior diagnosis of Barrett’s esophagus. Third, although Barrett’s esophagus is a “precursor” to EAC, the rates of progression are low, and 95 % of Barrett’s esophagus patients die from unrelated causes. For these reasons, most guidelines advise against routine endoscopic screening of patients with reflux [101, 102]. Mindful of the costs and hazards of invasive tissue-based screening programs, new population-based strategies are emerging that attempt to stratify people’s risk on the basis of easy-to-measure clinical, anthropometric, and environmental factors [103, 104].
For the minority of patients diagnosed with Barrett’s esophagus and thus at risk of progression to cancer, there is as yet no trial-based evidence for interventions that demonstrably reduce risk. Retrospective and prospective observational studies suggest that aspirin may have a role to play in inhibiting progression, but this awaits confirmation in clinical trials [36, 37]. There is also observational evidence that Barrett’s patients who regularly use PPIs [35•, 94] or statins [105] may have lower rates of progression to cancer. In terms of lifestyle modifications, several small trials have delivered ambitious exercise [106] and dietary [107] interventions to Barrett’s patients and followed them up for changes in biomarkers. While those trials successfully delivered their respective interventions and brought about changes in some anthropometric parameters, there was no evidence that they had any effect on neoplastic biomarkers.
For patients with dysplastic Barrett’s esophagus, new ablation technologies have demonstrated efficacy in reducing progression to high-grade dysplasia and cancer [108, 109]. Larger trials with extended follow-up are required to demonstrate the long-term effectiveness of these techniques.
Esophageal Squamous Cell Carcinoma
Secondary prevention of ESCC has been pursued more vigorously in high-incidence areas of China and Iran than elsewhere. Prospective studies in China provide strong evidence that ESD is the precursor lesion for ESCC [110], with 13-year risks of developing cancer of 24 %, 50 %, and 75 % for biopsy-proven mild, moderate, and severe ESD, respectively [111]. ESD can be detected on endoscopy using Lugol’s iodine staining. (Readers are referred to the excellent review by Taylor et al. [68••] for a full discussion of the strategies to prevent ESCC by targeting ESD). A number of endoscopic methods are available to treat ESD, including endoscopic mucosal resection, electrocoagulation, and radiofrequency ablation. Clinical trials are under way, evaluating the effectiveness of these measures.
Summary and Conclusions
Cancers of the esophagus are common, have poor survival, and exact a high mortality worldwide. The two main histological types differ markedly in their distribution and risk factors, and so they require different strategies for their control. Given the challenges of early detection and secondary prevention for both types of esophageal cancer, as well as the high attributable fractions of known causal factors [95, 96, 112, 113], primary prevention is arguably the preferred strategy for reducing the population burden of these tumors.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
GLOBOCAN 2012 v1.0, cancer incidence and mortality worldwide: IARC CancerBase No. 11. Lyon: International Agency for Research on Cancer; 2012 [cited 28 March 2014]. Available from: http://globocan.iarc.fr.
Pohl H, Welch HG. The role of overdiagnosis and reclassification in the marked increase of esophageal adenocarcinoma incidence. J Natl Cancer Inst. 2005;97(2):142–6.
Lagergren J, Mattsson F. No further increase in the incidence of esophageal adenocarcinoma in Sweden. Int J Cancer. 2011;129(2):513–6.
Pohl H, Sirovich B, Welch HG. Esophageal adenocarcinoma incidence: are we reaching the peak? Cancer Epidemiol Biomarkers Prev. 2010;19(6):1468–70.
Thrift A, Whiteman D. The incidence of esophageal adenocarcinoma continues to rise: analysis of period and birth cohort effects on recent trends. Ann Oncol. 2012;23(12):3155–62.
Howlader N, Noone AM, Krapcho M, Garshell J, Neyman N, Altekruse SF, et al. SEER cancer statistics review, 1975–2010. Bethesda: National Cancer Institute; 2013. [cited 27 March 2014]; Available from: http://seer.cancer.gov/csr/1975_2010/.
Ribet ME, Mensier EA. Reflux esophagitis and carcinoma. Surg Gynecol Obstet. 1992;175(2):121–5.
Lagergren J, Bergstrom R, Lindgren A, Nyren O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma [see comments]. N Engl J Med. 1999;340(11):825–31.
Mayne ST, Navarro SA. Diet, obesity and reflux in the etiology of adenocarcinomas of the esophagus and gastric cardia in humans. J Nutr. 2002;132(11 Suppl):3467S–70.
Wu AH, Tseng CC, Bernstein L. Hiatal hernia, reflux symptoms, body size, and risk of esophageal and gastric adenocarcinoma. Cancer. 2003;98(5):940–8.
Whiteman DC, Sadeghi S, Pandeya N, Smithers BM, Gotley DC, Bain CJ, et al. Combined effects of obesity, acid reflux and smoking on the risk of adenocarcinomas of the oesophagus. Gut. 2008;57(2):173–80.
Pandeya N, Webb PM, Sadeghi S, Green AC, Whiteman DC, Australian Cancer Study. Gastro-oesophageal reflux symptoms and the risks of oesophageal cancer: are the effects modified by smoking, NSAIDs or acid suppressants? Gut. 2010;59(1):31–8.
Sharma A, Elebiary A, Chowdhury S, Buttar N. Inflammation, obesity, Barrett’s esophagus, and esophageal adenocarcinoma. In: Dannenberg AJ, Berger NA, editors. Obesity, inflammation and cancer. Energy balance and cancer. Volume 7. New York: Springer; 2013. p. 133–45.
Kavanagh ME, O’Sullivan KE, O’Hanlon C, O’Sullivan JN, Lysaght J, Reynolds JV. The esophagitis to adenocarcinoma sequence; the role of inflammation. Cancer Lett. 2014;345(2):182–9.
Winter JW, Paterson S, Scobie G, Wirz A, Preston T, McColl KE. N-nitrosamine generation from ingested nitrate via nitric oxide in subjects with and without gastroesophageal reflux. Gastroenterology. 2007;133(1):164–74.
Iijima K, Henry E, Moriya A, Wirz A, Kelman AW, McColl KE. Dietary nitrate generates potentially mutagenic concentrations of nitric oxide at the gastroesophageal junction. Gastroenterology. 2002;122(5):1248–57.
McColl KE. When saliva meets acid: chemical warfare at the oesophagogastric junction. Gut. 2005;54(1):1–3.
Sharma P, McQuaid K, Dent J, Fennerty MB, Sampliner R, Spechler S, et al. A critical review of the diagnosis and management of Barrett’s esophagus: the AGA Chicago Workshop. Gastroenterology. 2004;127(1):310–30.
Tretli S, Robsahm TE. Height, weight and cancer of the oesophagus and stomach: a follow-up study in Norway. Eur J Cancer Prev. 1999;8(2):115–22.
Lagergren J, Bergstrom R, Nyren O. Association between body mass and adenocarcinoma of the esophagus and gastric cardia. Ann Intern Med. 1999;130(11):883–90.
Chow WH, Blot WJ, Vaughan TL, Risch HA, Gammon MD, Stanford JL, et al. Body mass index and risk of adenocarcinomas of the esophagus and gastric cardia. J Natl Cancer Inst. 1998;90(2):150–5.
Reeves GK, Pirie K, Beral V, Green J, Spencer E, Bull D. Cancer incidence and mortality in relation to body mass index in the Million Women Study: cohort study. Br Med J. 2007;335(7630):1134.
Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008;371(9612):569–78.
Hoyo C, Cook MB, Kamangar F, Freedman ND, Whiteman DC, Bernstein L, et al. Body mass index in relation to oesophageal and oesophagogastric junction adenocarcinomas: a pooled analysis from the International BEACON Consortium. Int J Epidemiol. 2012;41(6):1706–18.
Lagergren J, Mattsson F, Nyrén O. Gastroesophageal reflux does not alter effects of body mass index on risk of esophageal adenocarcinoma. Clin Gastroenterol Hepatol. 2014;12(1):45–51.
El-Serag HB, Ergun GA, Pandolfino J, Fitzgerald S, Tran T, Kramer JR. Obesity increases oesophageal acid exposure. Gut. 2007;56(6):749–55.
El-Serag HB, Graham DY, Satia JA, Rabeneck L. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am J Gastroenterol. 2005;100(6):1243–50.
El-Serag HB, Tran T, Richardson P, Ergun G. Anthropometric correlates of intragastric pressure. Scand J Gastroenterol. 2006;41(8):887–91.
Després J-P. Assessing the cardiometabolic risk of obesity: importance of visceral/ectopic fat and of the use of hypertriglyceridemic waist. In: Haslam DW, Sharma AM, le Roux CW, editors. Controversies in obesity. London: Springer; 2014. p. 127–35.
Roberts DL, Dive C, Renehan AG. Biological mechanisms linking obesity and cancer risk: new perspectives. Annu Rev Med. 2010;61:301–16.
Renehan AG, Frystyk J, Flyvbjerg A. Obesity and cancer risk: the role of the insulin-IGF axis. Trends Endocrinol Metab. 2006;17(8):328–36.
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. A review of human carcinogens. Part E: Personal habits and indoor combustions. Lyon: International Agency for Research on Cancer; 2012.
Cook MB, Kamangar F, Whiteman DC, Freedman ND, Gammon MD, Bernstein L, et al. Cigarette smoking and adenocarcinomas of the esophagus and esophagogastric junction: a pooled analysis from the International BEACON Consortium. J Natl Cancer Inst. 2010;102(17):1344–53.
Sheen E, Triadafilopoulos G. Adverse effects of long-term proton pump inhibitor therapy. Dig Dis Sci. 2011;56(4):931–50.
Singh S, Garg SK, Singh PP, Iyer PG, El-Serag HB. Acid-suppressive medications and risk of oesophageal adenocarcinoma in patients with Barrett’s oesophagus: a systematic review and meta-analysis. Gut. 2013. doi:10.1136/gutjnl-2013-305997. A comprehensive summary of the studies examining the association between proton pump inhibitors and EAC.
Jankowski J, Moayyedi P. Re: cost-effectiveness of aspirin chemoprevention for Barrett’s esophagus. J Natl Cancer Inst. 2004;96(11):885–7. author reply 887.
Jankowski J, DeCaestecker J, Harrison R, Watson P, Barr H, Attwood S, et al. NSAID and oesophageal adenocarcinoma: randomised trials needed to correct for bias. Lancet Oncol. 2006;7(1):7–8. author reply 9.
Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345(11):784–9.
Huang J-Q, Sridhar S, Chen Y, Hunt RH. Meta-analysis of the relationship between Helicobacter pylori seropositivity and gastric cancer. Gastroenterology. 1998;114(6):1169–79.
Forman D, Burley V. Gastric cancer: global pattern of the disease and an overview of environmental risk factors. Best Pract Res Clin Gastroenterol. 2006;20(4):633–49.
Islami F, Kamangar F. Helicobacter pylori and esophageal cancer risk: a meta-analysis. Cancer Prev Res (Phila). 2008;1(5):329–38.
Jeffery PL, McGuckin MA, Linden SK. Endocrine impact of Helicobacter pylori: focus on ghrelin and ghrelin O-acyltransferase. World J Gastroenterol. 2011;17(10):1249–60.
Flower R. What are all the things that aspirin does? Br Med J. 2003;327(7415):572–3.
Cuzick J, Otto F, Baron JA, Brown PH, Burn J, Greenwald P, et al. Aspirin and non-steroidal anti-inflammatory drugs for cancer prevention: an international consensus statement. Lancet Oncol. 2009;10(5):501–7.
Liao LM, Vaughan TL, Corley DA, Cook MB, Casson AG, Kamangar F, et al. Nonsteroidal anti-inflammatory drug use reduces risk of adenocarcinomas of the esophagus and esophagogastric junction in a pooled analysis. Gastroenterology. 2012;142(3):442–52. A pooled analysis of observational data, demonstrating consistently lower risks of EAC associated with regular aspirin consumption.
Algra AM, Rothwell PM. Effects of regular aspirin on long-term cancer incidence and metastasis: a systematic comparison of evidence from observational studies versus randomised trials. Lancet Oncol. 2012;13(5):518–27. A detailed review of the evidence examining aspirin consumption and cancer incidence. This analysis compares risk estimates from observational cohort studies with those obtained from long-term follow-up of randomized controlled trials.
Freedman ND, Park Y, Subar AF, Hollenbeck AR, Leitzmann MF, Schatzkin A, et al. Fruit and vegetable intake and esophageal cancer in a large prospective cohort study. Int J Cancer. 2007;121(12):2753–60.
Li WQ, Park Y, Wu JW, Ren JS, Goldstein AM, Taylor PR, et al. Index-based dietary patterns and risk of esophageal and gastric cancer in a large cohort study. Clin Gastroenterol Hepatol. 2013;11(9):1130–6.e2.
Steevens J, Schouten LJ, Goldbohm RA, van den Brandt PA. Vegetables and fruits consumption and risk of esophageal and gastric cancer subtypes in the Netherlands Cohort Study. Int J Cancer. 2011;129(11):2681–93.
Vaughan TL. Diet and upper gastrointestinal cancers: in search of dark matter. Clin Gastroenterol Hepatol. 2013;11(9):1137–9.
Anderson LA, Cantwell MM, Watson RG, Johnston BT, Murphy SJ, Ferguson HR, et al. The association between alcohol and reflux esophagitis, Barrett’s esophagus, and esophageal adenocarcinoma. Gastroenterology. 2009;136(3):799–805.
Gammon MD, Schoenberg JB, Ahsan H, Risch HA, Vaughan TL, Chow WH, et al. Tobacco, alcohol, and socioeconomic status and adenocarcinomas of the esophagus and gastric cardia [see comments]. J Natl Cancer Inst. 1997;89(17):1277–84.
Kabat GC, Ng SK, Wynder EL. Tobacco, alcohol intake, and diet in relation to adenocarcinoma of the esophagus and gastric cardia. Cancer Causes Control. 1993;4(2):123–32.
Levi F, Ollyo JB, La-Vecchia C, Boyle P, Monnier P, Savary M. The consumption of tobacco, alcohol and the risk of adenocarcinoma in Barrett’s oesophagus. Int J Cancer. 1990;45(5):852–4.
Pandeya N, Williams G, Green AC, Webb PM, Whiteman DC, Australian Cancer Study. Alcohol consumption and the risks of adenocarcinoma and squamous cell carcinoma of the esophagus. Gastroenterology. 2009;136(4):1215–24.
Vaughan TL, Davis S, Kristal A, Thomas DB. Obesity, alcohol, and tobacco as risk factors for cancers of the esophagus and gastric cardia: adenocarcinoma versus squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 1995;4(2):85–92.
Freedman ND, Murray LJ, Kamangar F, Abnet CC, Cook MB, Nyren O, et al. Alcohol intake and risk of oesophageal adenocarcinoma: a pooled analysis from the BEACON Consortium. Gut. 2011;60(8):1029–37. An important study documenting the null association between alcohol intake and EAC.
IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Alcohol consumption and ethyl carbamate. Lyon: International Agency for Research on Cancer; 2007.
Zeka A, Gore R, Kriebel D. Effects of alcohol and tobacco on aerodigestive cancer risks: a meta-regression analysis. Cancer Causes Control. 2003;14(9):897–906.
Islami F, Fedirko V, Tramacere I, Bagnardi V, Jenab M, Scotti L, et al. Alcohol drinking and esophageal squamous cell carcinoma with focus on light-drinkers and never-smokers: a systematic review and meta-analysis. Int J Cancer. 2011;129(10):2473–84.
Seitz HK, Stickel F, Homann N. Pathogenetic mechanisms of upper aerodigestive tract cancer in alcoholics. Int J Cancer. 2004;108(4):483–7.
Secretan B, Straif K, Baan R, Grosse Y, El Ghissassi F, Bouvard V, et al. A review of human carcinogens—part E: tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol. 2009;10(11):1033–4.
Matsuo K, Hamajima N, Shinoda M, Hatooka S, Inoue M, Takezaki T, et al. Gene-environment interaction between an aldehyde dehydrogenase-2 (ALDH2) polymorphism and alcohol consumption for the risk of esophageal cancer. Carcinogenesis. 2001;22(6):913–6.
Kamangar F, Chow WH, Abnet CC, Dawsey SM. Environmental causes of esophageal cancer. Gastroenterol Clin North Am. 2009;38(1):27–57. vii.
Blot WJ, Li J-Y, Taylor PR, Guo W, Dawsey S, Wang G-Q, et al. Nutrition Intervention Trials in Linxian, China: supplementation with specific vitamin/mineral combinations, cancer incidence, and disease-specific mortality in the general population. J Natl Cancer Inst. 1993;85(18):1483–91.
Li J-Y, Taylor PR, Li B, Dawsey S, Wang G-Q, Ershow AG, et al. Nutrition Intervention Trials in Linxian, China: multiple vitamin/mineral supplementation, cancer incidence, and disease-specific mortality among adults with esophageal dysplasia. J Natl Cancer Inst. 1993;85(18):1492–8.
Qiao Y-L, Dawsey SM, Kamangar F, Fan J-H, Abnet CC, Sun X-D, et al. Total and cancer mortality after supplementation with vitamins and minerals: follow-up of the Linxian General Population Nutrition Intervention Trial. J Natl Cancer Inst. 2009;101(7):507–18.
Taylor PR, Abnet CC, Dawsey SM. Squamous dysplasia—the precursor lesion for esophageal squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2013;22(4):540–52. A comprehensive review of the precursor to ESCC.
Limburg PJ, Wei W, Ahnen DJ, Qiao Y, Hawk ET, Wang G, et al. Randomized, placebo-controlled, esophageal squamous cell cancer chemoprevention trial of selenomethionine and celecoxib. Gastroenterology. 2005;129(3):863–73.
Chen T, Yan F, Qian J, Guo M, Zhang H, Tang X, et al. Randomized phase II trial of lyophilized strawberries in patients with dysplastic precancerous lesions of the esophagus. Cancer Prev Res. 2012;5(1):41–50.
Tran GD, Sun XD, Abnet CC, Fan JH, Dawsey SM, Dong ZW, et al. Prospective study of risk factors for esophageal and gastric cancers in the Linxian General Population Trial cohort in China. Int J Cancer. 2005;113(3):456–63.
Craver LF. A clinical study of etiology of gastric and esophageal carcinoma. Am J Cancer. 1932;16(1):68–102.
Andrici J, Eslick GD. Maté consumption and the risk of esophageal squamous cell carcinoma: a meta-analysis. Dis Esophagus. 2013;26(8):807–16.
Kamangar F, Schantz MM, Abnet CC, Fagundes RB, Dawsey SM. High levels of carcinogenic polycyclic aromatic hydrocarbons in maté drinks. Cancer Epidemiol Biomarkers Prev. 2008;17(5):1262–8.
Fagundes RB, Abnet CC, Strickland PT, Kamangar F, Roth MJ, Taylor PR, et al. Higher urine 1-hydroxy pyrene glucuronide (1-OHPG) is associated with tobacco smoke exposure and drinking maté in healthy subjects from Rio Grande do Sul, Brazil. BMC Cancer. 2006;6:139.
Islami F, Pourshams A, Nasrollahzadeh D, Kamangar F, Fahimi S, Shakeri R, et al. Tea drinking habits and oesophageal cancer in a high risk area in northern Iran: population based case-control study. Br Med J. 2009;338:b929.
Gao Y, Hu N, Han XY, Ding T, Giffen C, Goldstein AM, et al. Risk factors for esophageal and gastric cancers in Shanxi Province, China: a case–control study. Cancer Epidemiol. 2011;35(6):e91–9.
Nasrollahzadeh D, Kamangar F, Aghcheli K, Sotoudeh M, Islami F, Abnet CC, et al. Opium, tobacco, and alcohol use in relation to oesophageal squamous cell carcinoma in a high-risk area of Iran. Br J Cancer. 2008;98(11):1857–63.
Abnet CC, Qiao YL, Mark SD, Dong ZW, Taylor PR, Dawsey SM. Prospective study of tooth loss and incident esophageal and gastric cancers in China. Cancer Causes Control. 2001;12(9):847–54.
Guha N, Boffetta P, Wunsch Filho V, Eluf Neto J, Shangina O, Zaridze D, et al. Oral health and risk of squamous cell carcinoma of the head and neck and esophagus: results of two multicentric case-control studies. Am J Epidemiol. 2007;166(10):1159–73.
Sepehr A, Kamangar F, Fahimi S, Saidi F, Abnet CC, Dawsey SM. Poor oral health as a risk factor for esophageal squamous dysplasia in northeastern Iran. Anticancer Res. 2005;25(1B):543–6.
Abnet CC, Kamangar F, Islami F, Nasrollahzadeh D, Brennan P, Aghcheli K, et al. Tooth loss and lack of regular oral hygiene are associated with higher risk of esophageal squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2008;17(11):3062–8.
Homann N, Tillonen J, Rintamäki H, Salaspuro M, Lindqvist C, Meurman JH. Poor dental status increases acetaldehyde production from ethanol in saliva: a possible link to increased oral cancer risk among heavy drinkers. Oral Oncol. 2001;37(2):153–8.
Eisenbrand G, Spiegelhalder B, Preussmann R. Nitrate and nitrite in saliva. Oncology. 1980;37(4):227–31.
Sadeghi S, Bain CJ, Pandeya N, Webb PM, Green AC, Whiteman DC. Aspirin, nonsteroidal anti-inflammatory drugs, and the risks of cancers of the esophagus. Cancer Epidemiol Biomarkers Prev. 2008;17(5):1169–78.
Corley DA, Kerlikowske K, Verma R, Buffler P. Protective association of aspirin/NSAIDs and esophageal cancer: a systematic review and meta-analysis. Gastroenterology. 2003;124(1):47–56.
Yong F, Xudong N, Lijie T. Human papillomavirus types 16 and 18 in esophagus squamous cell carcinoma: a meta-analysis. Ann Epidemiol. 2013;23(11):726–34.
Liyanage SS, Rahman B, Ridda I, Newall AT, Tabrizi SN, Garland SM, et al. The aetiological role of human papillomavirus in oesophageal squamous cell carcinoma: a meta-analysis. PLoS One. 2013;8(7):e69238.
Koshiol J, Wei WQ, Kreimer AR, Chen W, Gravitt P, Ren JS, et al. No role for human papillomavirus in esophageal squamous cell carcinoma in China. Int J Cancer. 2010;127(1):93–100.
Antonsson A, Nancarrow DJ, Brown IS, Green AC, Drew PA, Watson DI, et al. High-risk human papillomavirus in esophageal squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2010;19(8):2080–7.
Sitas F, Egger S, Urban MI, Taylor PR, Abnet CC, Boffetta P, et al. InterSCOPE study: associations between esophageal squamous cell carcinoma and human papillomavirus serological markers. J Natl Cancer Inst. 2012;104(2):147–58.
Lahmann PH, Pandeya N, Webb PM, Green AC, Whiteman DC, Australian Cancer Study. Body mass index, long-term weight change, and esophageal squamous cell carcinoma: is the inverse association modified by smoking status? Cancer. 2012;118(7):1901–9.
Smith M, Zhou M, Whitlock G, Yang G, Offer A, Hui G, et al. Esophageal cancer and body mass index: results from a prospective study of 220,000 men in China and a meta-analysis of published studies. Int J Cancer. 2008;122(7):1604–10.
Kastelein F, Spaander MC, Steyerberg EW, Biermann K, Valkhoff VE, Kuipers EJ, et al. Proton pump inhibitors reduce the risk of neoplastic progression in patients with Barrett’s esophagus. Clin Gastroenterol Hepatol. 2013;11(4):382–8.
Pandeya N, Olsen CM, Whiteman DC. Sex differences in the proportion of esophageal squamous cell carcinoma cases attributable to tobacco smoking and alcohol consumption. Cancer Epidemiol. 2013;37(5):579–84.
Engel LS, Chow WH, Vaughan TL, Gammon MD, Risch HA, Stanford JL, et al. Population attributable risks of esophageal and gastric cancers. J Natl Cancer Inst. 2003;95(18):1404–13.
Williamson WA, Ellis FH, Gibb SP, Shahian DM, Aretz HT, Heatley GJ, et al. Barrett’s esophagus: prevalence and incidence of adenocarcinoma. Arch Intern Med. 1991;151(11):2212–6.
Yousef F, Cardwell C, Cantwell MM, Galway K, Johnston BT, Murray L. The incidence of esophageal cancer and high-grade dysplasia in Barrett’s esophagus: a systematic review and meta-analysis. Am J Epidemiol. 2008;168(3):237–49.
Hvid-Jensen F, Pedersen L, Drewes AM, Sørensen HT, Funch-Jensen P. Incidence of adenocarcinoma among patients with Barrett’s esophagus. N Engl J Med. 2011;365(15):1375–83. A population-based study documenting the rate of progression from Barrett’s oesophagus to cancer.
Reid BJ, Li X, Galipeau PC, Vaughan TL. Barrett’s oesophagus and oesophageal adenocarcinoma: time for a new synthesis. Nat Rev Cancer. 2010;10(2):87–101.
Fitzgerald RC, di Pietro M, Ragunath K, Ang Y, Kang J-Y, Watson P, et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett’s oesophagus. Gut. 2014;63(1):7–42.
Wang KK, Sampliner RE. Diagnosis, surveillance and therapy of Barrett’s esophagus. Am J Gastroenterol. 2008;103:788–97.
Thrift AP, Kendall BJ, Pandeya N, Vaughan TL, Whiteman DC. A clinical risk prediction model for Barrett esophagus. Cancer Prev Res (Phila). 2012;5(9):1115–23.
Thrift AP, Kendall BJ, Pandeya N, Whiteman DC. A model to determine absolute risk for esophageal adenocarcinoma. Clin Gastroenterol Hepatol. 2013;11(2):138–44.e2.
Kantor ED, Onstad L, Blount PL, Reid BJ, Vaughan TL. Use of statin medications and risk of esophageal adenocarcinoma in persons with Barrett’s esophagus. Cancer Epidemiol Biomarkers Prev. 2012;21(3):456–61.
Winzer BM, Paratz JD, Reeves MM, Whiteman DC. Exercise and the Prevention of Oesophageal Cancer (EPOC) study protocol: a randomized controlled trial of exercise versus stretching in males with Barrett’s oesophagus. BMC Cancer. 2010;10:292.
Kristal AR, Blount PL, Schenk JM, Sanchez CA, Rabinovitch PS, Odze RD, et al. Low-fat, high fruit and vegetable diets and weight loss do not affect biomarkers of cellular proliferation in Barrett esophagus. Cancer Epidemiol Biomarkers Prev. 2005;14(10):2377–83.
Shaheen NJ, Sharma P, Overholt BF, Wolfsen HC, Sampliner RE, Wang KK, et al. Radiofrequency ablation in Barrett’s esophagus with dysplasia. N Engl J Med. 2009;360(22):2277–88.
Phoa KN, van Vilsteren FG, Weusten BL, Bisschops R, Schoon EJ, Ragunath K, et al. Radiofrequency ablation vs endoscopic surveillance for patients with Barrett esophagus and low-grade dysplasia: a randomized clinical trial. JAMA. 2014;311(12):1209–17.
Li B, Taylor PR, Li JY, Dawsey SM, Wang W, Tangrea JA, et al. Linxian Nutrition Intervention Trials. Design, methods, participant characteristics, and compliance. Ann Epidemiol. 1993;3(6):577–85.
Wang GQ, Abnet CC, Shen Q, Lewin KJ, Sun XD, Roth MJ, et al. Histological precursors of oesophageal squamous cell carcinoma: results from a 13 year prospective follow up study in a high risk population. Gut. 2005;54(2):187–92.
Negri E, La-Vecchia C, Franceschi S, Decarli A, Bruzzi P. Attributable risks for oesophageal cancer in northern Italy. Eur J Cancer. 1992;28A(6–7):1167–71.
Olsen CM, Pandeya N, Green AC, Webb PM, Whiteman DC, Australian Cancer Study. Population attributable fractions of adenocarcinoma of the esophagus and gastroesophageal junction. Am J Epidemiol. 2011;174(5):582–90.
Compliance with Ethics Guidelines
Conflict of Interest
D.C. Whiteman declares no conflicts of interest.
Human and Animal Rights and Informed Consent
All studies by D.C. Whiteman involving animal and/or human subjects were performed after approval by the appropriate institutional review boards. When required, written informed consent was obtained from all participants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Whiteman, D.C. Esophageal Cancer: Priorities for Prevention. Curr Epidemiol Rep 1, 138–148 (2014). https://doi.org/10.1007/s40471-014-0015-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40471-014-0015-3