Abstract
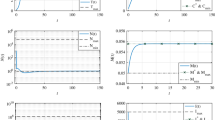
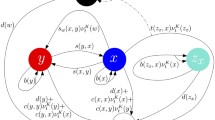
In this paper, we have considered competition models describing tumor–normal–immune cell interaction with the added effects of periodically pulsed chemotherapy. The parametric conditions needed to prevent relapse following attempts to remove the tumor or tumor metastasis are obtained. The effects of resistant tumor sub-populations are also investigated and recurrence prevention strategies are discussed. Our analytical findings are explained through numerical simulation which show the reliability of our models from the epidemiological point of view.






Similar content being viewed by others
References
Knolle H (1988) Cell kinetic modelling and the chemotherapy of cancer. Lecture notes in biomathematics, Vol 75. Springer, New York
Miller FR, Medina D, Heppner GH (1981) Preferential growth of mammary tumors in intact mammary fatpads. Cancer Res 41:3863–3867
Aroesty J, Lincoln T, Shapiro N, Boccia G (1973) Tumor growth and chemotherapy: mathematical methods, computer simulations, and experimental foundations. Math Biosci 17:243–300
Berenbaum MC (1969) Dose–response curves for agents that impair cell reproductive integrity. Br J Cancer 23:434–445
Murray JM (1990) Some optimal control problems in cancer chemotherapy with a toxicity limit. Math Biosci 100:49–67
Panetta JC (1996) A mathematical model of periodically pulsed chemotherapy tumor recurrence and metastasis in a competitive environment. Bull Math Biol 58(3):425–447
Kuznetsov V, Makalkin I (1992) Bifurcation analysis of mathematical model of interactions between cytotoxic lymphocytes and tumor cells—effect of immunological amplification of tumor growth and its connection with other phenomena of oncoimmunology. Biofizika 37(6):1063–1070
Kuznetsov V, Knott GD (2001) Modeling tumor regrowth and immunotherapy. Math Comput Model 33:1275–1287
Kuznetsov V, Makalkin I, Taylor M, Perelson A (1994) Nonlinear dynamics of immunogenic tumors: parameter estimation and global bifurcation analysis. Bull Math Biol 56(2):295–321
Kirschner D, panetta J (1998) Modelling immunotherapy of the tumor–immune interaction. J Math Biol 37:235–252
Kolev M (2003) Mathematical modelling of the competition between tumors and immune system considering the role of the antibodies. Math Comput Model 37:1143–1152
de Pillis LG, Gu W, Radunskaya AE (2006) Mixed immunotherapy and chemotherapy of tumors: modeling, applications and biological interpretations. J Theor Biol 238:841–862
de Pillis LG, Radunskaya AE (2001) A mathematical tumor model with immune resistance and drug therapy: an optimal control approach. J Theor Med 3:79–100
Arciero JC, Jackson TL, Kirschner DE (2004) A mathematical model of tumor-immune evasion and siRNA treatment. Discret Contin Dyn Syst Ser B 4(1):39–58
Bellomo N, Bellouquid A, Delitala M (2004) Mathematical topics on the modelling of multicellular systems in competition between tumor and immune cells. Math Models Methods Appl Sci 14:1683–1733
Bellomo N, Preziosi L (2000) Modelling and mathematical problems related to tumor evolution and its interaction with the immune system. Math Comput Model 32:413–452
Chan BS, Yu P (2012) Bifurcation analysis in a model of cytotoxic T-lymphocyte response to viral infections. Nonlinear Anal: Real World Appl 13:64–77
de Pillis LG, Radunskaya AE, Wiseman CL (2005) A validatted mathematical model of cell-mediated immune response to tumor growth. Cancer Res 61(17):7950–7958
Derbel L (2004) Analysis of a new model for tumor-immune system competition including long time scale effects. Math Models Methods Appl Sci 14:1657–1682
d’Onofrio A (2005) A general framework for modeling tumor-immune system competition and immunotherapy: mathematical analysis and biomedical references. Phys D 208:220–235
Nani F, Freedman HI (2000) A mathematical model of cancer treatment by immunotherapy. Math Biosci 163:159–199
Pinho STR, Bacelar FS, Andrade RFS, Freedman HI (2013) A mathematical model for the effect of anti-angiogenic therapy in the treatment of cancer tumors by chemotherapy. Nonlinear Anal: Real World Appl 14:815–828
Siu H, Vitetta ES, May RD, Uhr IW (1986) Tumor dormancy. I. Regression of bcl tumor and induction of a dormant tumor state in mice chimeric at the major histocompatibility complex. J Immunol 137:1376–1382
Takayanagi T, Ohuchi A (2001) A mathematical analysis of the interactions between immunogenic tumor cells and cytotoxic T lymphocytes. Microbiol Immunol 45:709–715
Yafia R (2006) Dynamics analysis and limit cycle in a delayed model for tumor growth with quiescene. Nonlinear Anal: Model Control 11:95–110
de Pillis LG, Gu W, Fister KR, Head T, Maples K, Murugan A, Neal T, Yoshida K (2007) Chemotherapy for tumors: an analysis of the dynamics and a study od quadratic and linear optimal controls. Math Biosci 209:292–315
de Pillis LG, Radunskaya AE (2003) The dynamics of an optimally controlled tumor model: a case study. Math Comput Model 37:1221–1244
Engelhart M, Lebiedz D, Sager S (2011) Optimal control for selected cancer chemotherapy ODE models: a view on the potential of optimal schedules and choice of objective function. Math Biosci 229:123–134
Fister KR, Donnelly J (2005) Immunotherapy: an optimal control theory approach. Math Biosci Eng 2(3):499–510
Fister KR, Panetta JC (2000) Optimal control applied to cell-cycle specific cancer chemotherapy. SIAM J Appl Math 60(3):1059–1072
Fister KR, Panetta JC (2003) Optimal control applied to competing chemotherapeutic cell-kill strategies. SIAM J Appl Math 63(6):1954–1971
Matveev A, Savkin A (2002) Application of optimal control theory to analysis of cancer chemotherapy regimens. Syst Cont Lett 46:311–321
Swarnali S, Samanta GP (2013) Dynamical behaviour of a tumor-immune system with chemotherapy and optimal control. J Nonlinear Dyn. doi:10.1155/2013/608598
Swarnali S, Samanta GP (2015) Analysis of the dynamics of a tumor-immune system with chemotherapy and immunotherapy and quadratic optimal control. Differ Equ Dyn Syst. doi:10.1007/s12591-015-0250-1
Elmouki I, Saadi S (2014) BCG immunotherapy optimization on an isoperimetric optimal control problem for the treatment of superficial bladder cancer. Int J Dyn Control. doi:10.1007/s40435-014-0106-5
Gyori I, Michelson S, Leith JT (1988) Time-dependent subpopulation induction in heterogeneous tumors. Bull Math Biol 50:681–696
Michelson S, Leith JT (1995) Interlocking triads of growth control in tumors. Bull Math Biol 57:345–366
Michelson S, Leith JT (1988) Unexpected equilibria resulting from differing growth rates of subpopulations within heterogeneous tumors. Math Biosci 91:119–129
Michelson S, Leith JT (1993) Tumor heterogeneity: a review of the theory. Drug News Perspect 6:655–661
Burger RA, Grosen EA, Ioli GR, Van Eden ME, Brightbill HD, Gatanaga M, DiSaia PJ, Granger GA, Gatanaga T (1994) Host-tumor interaction in ovarian cancer spontaneous release of tumor necrosis factor and interleukin-1 inhibitors by purified cell populations from human ovarian carcinoma in vitro. Gynecol Oncol 55:294–303
Dotto GP, Weinberg A, Ariza A (1988) Malignant transformation of mouse primary keratinocytes by Harvey sarcoma virus and its modulation by surrounding normal cells. Proc Natl Acad Sci USA 85:6389–6393
La Rocca SA, Grossi M, Falcone G, Alema S, Tato F (1989) Interaction with normal cells suppresses the transformed phenotype of v-myc-transformed quail muscle cells. Cell 58:123–131
Cornil I, Theodorescu D, Man S, Herlyn M, Jambrosic J, Kerbel RS (1991) Fibroblast cell interactions with human melanoma cells affect tumor cell growth as a function of tumor progression. Proc Natl Acad Sci USA 88:6028–6032
Hale JK (1977) Theory of functional differential equations. Springer, New York
Goldie JH, Coldman AJ (1979) A mathematical model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat Rep 63:1727–1733
Michelson S, Miller BE, Glicksman AS, Leith JT (1987) Tumor micro-ecology and competitive interactions. J Theor Biol 128:233–246
Schimke RT (1984) Gene amplification, drug resistance, and cancer. Cancer Res 44:1735–1742
Schimke RT (1988) Gene amplification in cultured cells. J Biol Chem 263:5989–5992
Souhami RL, Gregory WM, Birkhead BG (1988) Mathematical models in high-dose chemotherapy. Antibiot Chemother 41:21–28
Birkhead BG, Gregory WM (1984) A mathematical model of the effects of drug resistance in cancer chemotherapy. Math Biosci 72:59–69
Martin RB, Fisher ME, Michin RF, Teo KL (1992) Optimal control of tumor size used to maximize survival time when cells are resistant to chemotherapy. Math Biosci 110:201–219
Costa MIS, Boldrini JL, Bassanezi RC (1995) Drug kinetics and drug resistance in optimal chemotherapy. Math Biosci 125:191–209
Acknowledgments
The authors are grateful to the anonymous referees and the Editor-in-Chief (Prof. Jian-Qiao Sun) for their careful reading, valuable comments and helpful suggestions, which have helped them to improve the presentation of this work significantly. The first author likes to thank TWAS, UNESCO and National Autonomous University of Mexico (UNAM) for financial support. He is grateful to Prof. José Antonio Seade Kuri and Prof. Marcelo Aguilar, Institute of Mathematics, National Autonomous University of Mexico for their helps and encouragements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samanta, G.P., Aíza, R.G. & Sharma, S. Analysis of a mathematical model of periodically pulsed chemotherapy treatment. Int. J. Dynam. Control 5, 842–857 (2017). https://doi.org/10.1007/s40435-015-0204-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40435-015-0204-z