Abstract
Vitiligo is a chronic skin disorder characterised by the loss of melanocytes and subsequent skin depigmentation. Although many theories have been proposed in the literature, none alone explains the pathogenesis of vitiligo. Oxidative stress has been identified as a potential factor in the pathogenesis of vitiligo. A growing body of evidence suggests that antioxidant therapies may offer a promising approach to managing this condition. This review summarises the potential mechanisms of oxidative stress and the types of melanocyte death in vitiligo. We also provide a brief overview of the most commonly studied antioxidants. Melanocytes in vitiligo are thought to be damaged by an accumulation of reactive oxygen species to destroy the structural and functional integrity of their DNA, lipids, and proteins. Various causes, including exogenous and endogenous stress factors, an imbalance between prooxidants and antioxidants, disruption of antioxidant pathways, and gene polymorphisms, lead to the overproduction of reactive oxygen species. Although necroptosis, pyroptosis, ferroptosis, and oxeiptosis are newer types of cell death that may contribute to the pathophysiology of vitiligo, apoptosis remains the most studied cell death mechanism in vitiligo. According to studies, vitamin E helps to treat lipid peroxidation of the skin caused by psoralen ultra-violet A treatment. In addition, Polypodium leucotomos increased the efficacy of psoralen ultra-violet A or narrow-band ultraviolet B therapy. Our review provides valuable insights into the potential role of oxidative stress in pathogenesis and antioxidant-based supporting therapies in treating vitiligo, offering a promising avenue for further research and the development of effective treatment strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Melanocytes naturally contain significant amounts of reactive oxygen species. If their degradation is disturbed, this leads to the inhibition of melanin synthesis and the death of melanocytes, resulting in the hypopigmented lesions of vitiligo. |
Oxidative stress-induced death of melanocytes in vitiligo is primarily triggered by apoptosis via the mitochondrial pathway by the release of cytochrome c. However, other mechanisms such as pyroptosis, necroptosis, ferroptosis and oxeiptosis may also be involved. |
Clinical studies have shown that certain antioxidant supplements, including Ginkgo biloba, Polypodium leucotomos and vitamin E, show promising results. In addition, the combination of phototherapy and antioxidant supplements has been shown to be more effective than phototherapy alone in the treatment of vitiligo. |
1 Introduction
Vitiligo is an acquired chronic depigmentation disorder of the skin affecting about 0.5–2% of the population worldwide. It is characterised by demarcated white patches resulting from the selective loss of epidermal melanocytes [1]. Although vitiligo can appear at any age, it most frequently affects young people aged between 10 and 30 years [2]. Treatment options for vitiligo vary and range from topical and oral drugs and phototherapy to cellular and tissue grafts [3]. Understanding etiopathogenesis is crucial for finding new treatment modalities. Various hypotheses have been developed to explain the pathogenesis of vitiligo. These include genetic, oxidative stress, autoimmune, autoinflammatory, neural, apoptotic, molecular adhesion disorder and multifactorial mechanisms [1]. None of the hypotheses proposed so far explains the pathogenesis of vitiligo entirely. Numerous studies in recent years have shown that oxidative stress hypersensitivity plays a significant role in determining melanocyte degeneration, leading to the development of the disease [4]. Oxidative stress and defects in metabolic pathways can promote dysregulation of immune responses in vitiligo [5]. Redox disequilibrium can cause melanocyte death by activating cytotoxic T cells and other death modalities such as apoptosis, necroptosis, and necrosis or oxeiptosis [6]. Because of their properties of neutralising reactive oxygen species, supplementation with antioxidants could be a potential treatment support option [7]. Unfortunately, there is limited evidence of antioxidant efficacy [8]. Therefore, in this review, we provide an overview of the mechanisms of vitiligo-associated oxidative stress and the most commonly studied antioxidants, describe the proposed mechanisms of action, and discuss the underlying evidence for their use or ineffectiveness.
2 Oxidative Stress
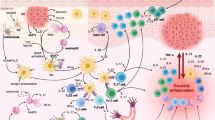
Oxidative and nitrosative stress are thought to play an important role in the development of vitiligo [9]. Oxidative stress refers to the excessive production of highly active molecules such as reactive oxygen species (ROS) in the body [2]. Melanocytes are thought to be damaged by an accumulation of ROS by destroying the structural and functional integrity of their DNA, lipids and proteins [10]. Exogenous and endogenous stimuli both play a role in excessive ROS production. Considering the external stressor, environmental triggers, including ultraviolet radiation, cytotoxic chemicals, trauma, pregnancy, stress, and immunisation, can contribute to the overproduction of ROS [11, 12]. However, when endogenous stimuli are considered, excessive ROS may also stem from melanogenesis. Melanin production is accompanied by active production of ROS and melanin itself absorbs ultraviolet light, which causes oxidative stress. The conversion of DOPA to dopaquinone and dopaquinone to dopachrome results in the formation of ROS during the melanin synthesis process. Hence, melanocytes are one of the different types of cutaneous cells that contain massive amounts of ROS [13]. It is worth noting that the energy-intensive aspect of melanogenesis suggests a high demand for adenosine triphosphate. In mitochondria, ROS are self-produced in the biosynthesis of adenosine triphosphate, another example of endogenous stimuli of excessive ROS production. Because of their control over oxidative stress-related ageing and apoptosis, mitochondria are considered a dominant source of ROS [14]. In a normal physiological state, the small amount of ROS produced by the body is converted into non-toxic substances by the antioxidant system so that it does no harm. However, when ROS production increases in pathological conditions, it creates an imbalance between prooxidants and antioxidants in the skin and the blood [3]. Patients with vitiligo have been found to have not only significantly increased average levels of hydrogen peroxide (H2O2) in their skin lesions, but also significantly lower measured activities of catalase, superoxide dismutase, glutathione peroxidase and glutathione S-transferase than in the control group, as well as significantly increased levels of malondialdehyde [9, 15, 16]. Compared with the segmental vitiligo group, these changes in prooxidants and antioxidants were particularly pronounced in non-segmental vitiligo. However, no significant correlation with the duration of vitiligo was found [15].
Oxidative stress can lead to vitiligo through various mechanism (Fig. 1). First, it disrupts the process of melanin synthesis by inducing the release of high mobility group box 1 (HMGB1) protein, thus downregulating the melanin biosynthesis-related proteins: melanocyte-inducing transcription factor, tyrosinase, tyrosinase-related protein-1, tyrosinase-related protein-2, RAB27A and fascin actin-bundling protein 1 [17, 18]. Furthermore, the excessive accumulation of ROS not only disrupts the process of melanin synthesis, but also destabilises, synthesises and disrupts the circulation of lipids in melanocytes, resulting in damage to the mitochondrial electron transport chain and increased ROS production, creating a vicious cycle and destroying melanocytes [19]. Oxidative stress is a critical factor in inducing the release of chemokines from keratinocytes that direct CD8+ T cells and lead to migration into skin tissue, causing melanocyte death [20]. In addition, cytokines produced by skin-invading T cells can further amplify the immune response [21]. Remarkably high levels of proinflammatory interleukins are found in perilesional vitiligo skin and in the peripheral bloodstream [22]. The most prominent interleukins (ILs) are IL-15, C-X-C motif ligand 16 (CXCL16), CXCL9, IL -17 and IL-1b [19, 20, 22,23,24]. Most of those interleukins potentiate the activation and expression of cytotoxic proteins in CD8+ T cells [22]. As for interleukin, IL-17 and IL-1b act by inhibiting cell-specific melanosomal proteins and activating T helper-17 cells, which increase cell damage by causing mitochondrial dysfunction [19]. Inhibition of the chemotactic axis could be a promising therapeutic option [20, 22].
Nuclear factor E2-related factor 2 (Nrf2), activated by ROS, protects against oxidative stress and chemically induced cell damage. The Kelch-like ECH-associated protein 1 (KEAP1) regulates Nrf2 activity. The KEAP1-Nrf2 complex breaks down when the cell is exposed to oxidative stress, and the released Nrf2 is then transported into the nucleus and binds to antioxidant response elements [25, 26]. Jian et al. found that induction of haem oxygenase-1 through activation of the Nrf2-ARE pathway protected melanocytes from H2O-induced toxicity and that upregulation of Nrf2 expression reduced H2O2-induced apoptosis of human melanocytes [27]. It suggests that the KEAP1/Nrf2/ARE pathway may be one of the most important signalling pathways in protecting cells from oxidative stress. Furthermore, abnormalities in the Nrf2-p62 pathway also impair autophagy in vitiligo melanocytes under oxidative stress [28, 29]. The role of Nrf2 gene polymorphisms in the susceptibility of patients to vitiligo and in altering the clinical presentation of the disease is also suspected [30].
Sirtuins (SIRTs) are a protein family with versatile functions, such as metabolic control, epigenetic modification and lifespan extension [31]. It has been observed that the expression and deacetylase activity of SIRT3 are attenuated in vitiligo melanocytes. Oxidative stress impairs SIRT3 activity and transcription by regulating protein carbonylation and transcriptional function of peroxisome proliferator-activated receptor-γ coactivator-1α, leading to mitochondrial dysfunction and activation of the mitochondrial apoptotic pathway via optic atrophy 1 to induce cell death of vitiligo melanocytes [32].
A genetic predisposition to oxidative stress and immune-mediated destruction of melanocytes may influence the development of vitiligo. Several human leukocyte antigen (HLA) alleles associated with an increased risk of vitiligo can be distinguished between different nations and ethnic groups [33]. Human leukocyte antigen alleles encode histocompatibility glycoproteins that enable the initiation of a specific immune response through the presentation of antigens to T cells and have been identified as risk factors for other skin diseases such as psoriasis [34]. Hayran et al. showed that serum total antioxidant capacity is significantly lower in patients without the HLA-DRB1*01 allele. The HLA-DRB1*01 allele was more common in the control group and in patients with vitiligo with high serum total antioxidant capacity levels, suggesting a protective role of this allele in vitiligo [33].
3 Forms of Melanocyte Death Associated with Oxidative Stress
Cell death may occur in multiple forms in response to different stressors, particularly oxidative stress [35], which is also firmly implicated in several pathways leading to vitiligo melanocyte loss. Apoptosis is the mechanism of cell death that has been most studied in vitiligo. However, other types of cell death, such as typical necrosis and autophagy, also appear to be implicated in the death of melanocytes in patients with vitiligo. The pathogenesis of vitiligo may also be influenced by the emerging processes of regulated cell death known as ferroptosis, necroptosis and pyroptosis [25]. Cells can die through regulated cell death or accidental cell death [35]. Spontaneous attacks and damage beyond possible control mechanisms can trigger accidental cell death, such as necrosis. Regulated cell death apoptosis, in contrast, has different biochemical, functional and immunological effects and is carried out by a group of identified effector molecules (apoptosis, necroptosis, pyroptosis, oxeiptosis, ferroptosis, parthanatos) [36]. Several diseases, including cancer, neurodegenerative diseases, autoimmune diseases, infectious diseases and vitiligo, are caused by a loss of control over single or combined modes of cell death. In recent years, understanding of the aetiology of the disease has improved with a number of studies investigating the molecular mechanism behind the loss of melanocytes in vitiligo [37].
3.1 Apoptosis
Apoptosis is the primary mechanism by which ROS destroys melanocytes in vitiligo [38]. Apoptosis occurs in all multi-cellular organisms and is the best studied form of programmed cell death. Several molecular mechanisms are involved in the induction of apoptosis in melanocytes in vitiligo. Nevertheless, we can mainly divide them into the extrinsic and intrinsic apoptosis signalling pathways regulated by activated caspases and molecular systems such as Bcl-2/Bax and Fas/Fas ligand [39, 40]. The mitochondrial signalling pathway of intrinsic apoptosis is particularly relevant to the pathogenesis of vitiligo (Fig. 2) [41]. Among the members of the caspase family, caspase-3 is a crucial modulator of apoptosis in mammalian cells [42]. The Bcl-2 family, in contrast, includes Bcl-2, which can protect cells from various apoptotic triggers, and Bax, an apoptosis agonist that acts through heterodimerisation with Bcl-2 [43]. It has been shown that perilesional melanocytes have a lower ratio of Bcl-2/Bax than control melanocytes, suggesting that melanocytes from patients with vitiligo are more susceptible to apoptosis activation due to altered Bcl-2 and Bax expression and that the balance between pro-apoptotic and anti-apoptotic proteins in vitiligo melanocytes is shifted towards apoptosis [44].
In this pathway, oxidative stress can trigger the release of cytochrome c from mitochondria into the cytosol. Cytochrome c binds to apoptotic protease activation factor-1, a cytosolic protein that forms an oligomeric apoptosome complex with procaspase-9. The apoptosome complex activates caspase-9, which cleaves and activates downstream effector caspases such as caspase-3 and caspase-7. The activated effector caspases then cleave various cellular substrates, leading to apoptosis.
The endoplasmic reticulum (ER) is essential for the synthesis, folding and transport of proteins. Any disruption of the normal function of the ER, especially oxidative stress, can lead to ER stress, which is characterised by an accumulation of misfolded or unfolded proteins in the ER lumen. Endoplasmic reticulum stress triggers a cellular response known as the unfolded protein response (UPR), which aims to restore ER homeostasis. However, suppose the stress is prolonged or severe. In this case, the UPR can activate apoptotic signalling pathways that lead to cell death [45, 46]. Homocysteine has been shown to cause vitiligo-related death of melanocytes by triggering ER stress and UPR activation in a ROS-dependent manner [47]. Moreover, the ROS-induced increase in the activity of CXCL16, a chemokine essential for the migration of cytotoxic T cells in vitiligo, is also caused by UPR activation [23]. In addition to intrinsic apoptosis, oxidative stress may also contribute to extrinsic apoptosis in vitiligo. The extrinsic pathway is triggered by the binding of a death ligand such as tumour necrosis factor (TNF) or Fas ligand to the corresponding death receptors such as TNF receptor 1 and Fas receptor, respectively [48]. Furthermore, cytokines such as TNF-α and interferon-gamma have been shown to induce extrinsic apoptosis in melanocytes in addition to ROS. These cytokines are produced by activated T cells that invade the skin in vitiligo [49, 50]. It was shown that keratinocytes can influence melanocyte function and survival through several mechanisms and are involved in a vitiligo potential patho-mechanism. In some cases, keratinocytes can promote melanocyte loss. Keratinocytes release cytokines to attract autoreactive T cells. They interfere with signal transduction in melanocytes, leading to the death of these cells and acting as a mediator of the damaging effects of oxidative stress [22]. Caspase-3-mediated death of melanocytes can also be triggered by ROS-induced secretion of adenosine triphosphate from keratinocytes, which can also induce nearby keratinocytes to produce CXCL9 to chemically attract CD8+ T cells [24]. One of these microRNAs (miRNAs), miR-25, has been shown to have important functions in keratinocytes. MiR-25 is overexpressed in both keratinocytes and melanocytes in response to oxidative stress, which decreases the activity of antioxidant enzymes and impairs melanosome transport, leading to redox imbalance [51]. Furthermore, oxidative stress activates the release of HMGB1 signal transduction in keratinocytes by decreasing the level of melanogenetic molecules (including gp100) and inducing apoptosis. In addition to inducing apoptosis, the transfer of HMGB1 from the melanocyte nucleus to the cytosol under oxidative stress can also suppress Nrf2 expression [52, 53].
3.2 Pyroptosis
In contrast to apoptosis, pyroptosis is an inflammatory form of programmed cell death that often occurs during infection with intracellular pathogens [54]. Activation of gasdermin D and the caspase-1/4/5/11 series distinguishes pyroptosis from other forms of death. However, the main regulator of pyroptosis is caspase 1, which is activated by the inflammasome, a large supramolecular complex. The activated caspases then cleave gasdermin, form pores in the cell membrane, and lead to cell lysis and secretion of IL-1β [55, 56]. An essential element of the NLRP1 inflammasome that activates IL-1 is the protein pyrin domain-containing protein 1 [57, 58]. It has been suggested that the inflammasome and IL-1β are involved in melanocyte loss, as staining for these markers was highly positive in the perilesional skin of patients with active vitiligo [59]. Pyroptosis was discovered to be involved in cell death via granzyme A, which cleaves gasdermin B. These findings support the hypothesis that pyroptosis may be involved in melanocyte death associated with ROS in vitiligo [12, 60].
3.3 Necroptosis
Necroptosis is a programmed cell death response to a specific environmental signal, morphologically resembling necrosis. It differs from the processes of apoptosis and autophagy in its particular mechanism of action and physiological significance [61]. Receptor-interacting proteins (RIPK) 1 and RIPK3 are crucial components of this pathway. The mixed lineage kinase-like domain, identified as a critical mediator in necroptosis, is then activated by RIPK3 [62]. Numerous stimulants, including activation of death receptors (such as Fas), Toll-like receptors (e.g. TLR3 and TLR4) and adhesion receptors, can lead to necroptosis [63]. Studies have shown that ROS is involved in necroptosis, particularly mitochondrial ROS, which promotes the autophosphorylation of RIP1 necessary for the inscription of RIP3 into necrosomes, and also ROS, which is dependent on RIP3, promotes necroptosis [64]. Necroptosis has been shown to be strongly associated with vitiligo, as melanocytes from perilesional skin affected by vitiligo were significant for necroptosis markers, such as phosphorylated RIP3 and a phosphorylated mixed-lineage kinase-like domain. Some studies found that mitochondrial ROS production mediated by RIP1 contributes to the development of a necrosome in melanocytes. The RIP1 pathway was also shown to contribute specifically to the promotion of oxidative stress-induced necroptosis, which leads to melanocyte death. This means that melanocytes exposed to oxidative stress may undergo necroptosis when the RIP1 pathway is activated [37, 65].
3.4 Ferroptosis
Ferroptosis is a form of regulated cell death characterised by the accumulation of iron and lipid peroxides in cells [66]. It is driven by iron-dependent phospholipid peroxidation and regulated by several cellular metabolic events, including redox homeostasis, iron handling, mitochondrial activity, and metabolism of amino acids, lipids and sugars [67]. Although ferroptosis has been extensively studied in many diseases [68,69,70]. It is still unclear whether ferroptosis contributes to melanocytes loss in vitiligo. Oxidative stress is one of the main factors that trigger ferroptosis, as shown by the significantly increased levels of LIP-ROS and ROS [71]. Melanocytes in the skin have more accessible iron than keratinocytes. The level of Fe2+ in melanocytes increases in response to external oxidative stimulation. This significantly increases the sensitivity of melanocytes to ferroptosis [72]. Ferroptosis triggers can be divided into those that act through the cystine/glutamate antiporter system, such as erastin, and those that act by inhibiting the activity of glutathione peroxidase, such as Ras Selective Lethal 3 [73, 74]. Some studies have shown that glutathione peroxidase is decreased in the serum and skin of patients with vitiligo, although the role of glutathione peroxidase 4 in vitiligo has not been fully elucidated [75]. Yang et al. found that Ras Selective Lethal 3 can induce ferroptosis in primary melanocytes, as reflected by suppressed cell growth, mitochondrial dysfunction, increased ROS production and iron ion accumulation [76]. According to studies by Wu et al., the level of ferroptosis markers, especially erastin, changed dramatically in the epidermis of patients with vitiligo. In in vitro conditions, erastin increased iron overload, oxidative stress and lipid peroxide production in human epidermal melanocytes [77]. This provides new evidence that ferroptosis may occur during the vitiligo process.
3.5 Oxeiptosis
In 2018, Holze et al. discovered that high concentrations of ozone (O3) or H2O can trigger an apoptosis-like death process called oxeiptosis [78]. Oxeiptosis is a caspase-independent cell death that is triggered by oxidative stress and acts in the elimination of pathogens. Oxeiptosis is mediated by KEAP1, phosphoglycerate mutase family member 5, and apoptosis-inducing factor mitochondrion-associated 1 under oxidative stress [6]. The reduction of KEAP1 and phosphoglycerate mutase family member 5 demonstrated that the KEAP1-phosphoglycerate mutase family member 5-apoptosis-inducing factor mitochondrion-associated 1 signalling pathway is responsible for the death of melanocytes initiated by oxidative stress [6]. In the perilesional skin of patients with vitiligo, Chen et al. demonstrated oxeiptosis in melanocytes exposed to ROS [12]. However, it is still unclear how melanocyte apoptosis is involved in the aetiology of vitiligo [37]. Mitochondria may play a crucial role in controlling oxeiptosis, as they are an important source of ROS and a component of the oxeiptosis locus. However, this relationship needs further investigation [79].
4 Potential Therapeutic Approaches to Reduce Oxidative Stress
An antioxidant is a compound that considerably lessens or stops the harmful effects of free radicals on human tissue. Antioxidants can be categorised based on their structure, solubility and the kinetics of the processes in which they are involved [80]. The skin is endowed with natural antioxidants because it is exposed to numerous environmental factors. Vitamin E, catalase, superoxide dismutases and glutathione peroxidases are present in the viable layer of the epidermis. Glutathione, vitamin C, uric acid, alpha-tocopherol, squalene and coenzyme Q10 are present in the stratum corneum of human skin [81]. Antioxidant-based supporting treatment has been proposed as a therapy to prevent photo-ageing of the skin [82, 83], skin cancers [84] and autoimmune diseases of the skin [85]. To date, numerous drugs containing antioxidant-like substances have been investigated to either support or replace standard treatments for vitiligo (Table 1). Insufficient knowledge of antioxidants has been reported as a significant predictor of a lower frequency of antioxidant use by dermatologists in China [7].
4.1 Vitamin C
Vitamin C eliminates oxidative stress by oxidising ascorbate to monodehydroascorbate and dehydroascorbate. It is also a cofactor for the enzymatic activity of prolyl hydroxylase in collagen synthesis. Vitamin C and its derivatives inhibit tyrosinase activity and melanin content in a dose-dependent manner [86]. In a study with 30 patients with vitiligo, significantly lower vitamin C levels were found in patients with vitiligo [87]. In Madagascar, a combination of a cream containing clobetasol propionate 0.05%, oral vitamins C and B12 and microtrauma suppression proved to be an effective therapy. Response to treatment was better in young people, in lesions younger than 1 year, and when vitiligo was localised to the face and neck [88].
4.2 Vitamin E
Vitamin E is a photoprotective antioxidant that absorbs certain wavelengths and protects against lipid peroxidation [89]. A meta-analysis by Liu et al. showed low plasma and tissue vitamin E levels in patients with vitiligo [90]. Studies suggest that the combination of vitamin E with psoralen and ultraviolet A (PUVA) phototherapy may prevent oxidative disorders resulting from PUVA therapy, but did not improve clinical improvement of vitiligo lesions [91]. However, the combination of narrow-band ultraviolet B (NB-UVB) with oral supplementation of vitamin E may increase the efficacy of therapy [92,93,94].
4.3 Selenium
Selenium is considered as an important trace element in the modulation of oxidative stress [95].The main role of selenium is attributed to its presence in glutathione peroxidase, which protects cells from the damaging effects of free radicals. A meta-analysis by Iv et al. examined ten studies of changes in selenium levels in patients with vitiligo. Ultimately, it was found that patients with vitiligo had similar selenium levels to healthy controls. Another subgroup analysis showed that patients with vitiligo had lower selenium levels in skin and hair compared with controls, but no difference in serum levels [96]. Another study found that the total selenium levels of patients with vitiligo and healthy controls were similar. Subsequently, a subgroup analysis showed that selenium levels were lower in patients with vitiligo than in Asian controls, while there was no significant difference between Caucasians and healthy controls [95]. By comparison, other studies have shown an increase in serum selenium levels in patients with vitiligo compared with the control group [97].
4.4 Zinc
Zinc can protect melanocytes through its anti-apoptotic and antioxidant effects. It may also regulate melanin production by the release of the α-melanocyte stimulating hormone and the precipitation of zinc-α2-glycoprotein in tissues. Zinc is also one of the non-specific elements that contribute to the regulation of cell-mediated immunity and the control of gene expression [98]. A prospective study by Zaki et al. found a negative correlation between serum zinc levels and the severity and extent of vitiligo [99]. An analysis of three articles comparing serum zinc levels in patients with vitiligo and controls found that higher serum zinc levels may reduce the risk of vitiligo [97]. The average serum zinc level was significantly decreased in patients with vitiligo. In contrast, the levels of cytokines involved in the control of melanocyte proliferation and differentiation and the inhibition of melanogenesis, IL-17, IL-4 and IL-6, were significantly increased in the vitiligo group compared with the control group. Serum zinc levels correlated significantly negatively with IL-6, IL-4 and IL-17 [98]. A combination of oral zinc and a topical corticosteroid was also found to be more effective in repigmentation than the topical corticosteroid alone [100].
4.5 Copper
Copper (Cu) plays two main roles: it promotes melanogenesis and provides physiological protection against oxidative stress by participating in the destruction of free radicals through the formation of superoxide dismutase, which catalyses the dismutation of reactive oxygen species to oxygen (O2) or H2O2. Together with zinc, they catalyse the rearrangement of dopachrome to 5,6-dihydroxyindole-2-carboxylic acid in the final phase of eumelanin formation in melanogenesis [101, 102]. In a study of a Chinese population, serum Cu levels were significantly lower in patients with vitiligo than in healthy controls [103]. In Iranian patients, mean serum Cu and zinc levels were also significantly lower in patients with lower segmental and non-segmental vitiligo than in the control group [104]. By comparison, in the study by Wacewicz et al., serum Cu levels did not differ significantly between patients with vitiligo and control subjects [102]. A case-control study of 100 patients with vitiligo from India showed increased Cu levels compared with healthy individuals [105].
4.6 Ginkgo Biloba
Ginkgo biloba (GB) is a traditional Chinese herb. Ginkgo biloba extract protects melanocytes from oxidative damage by inhibiting H2O2-induced apoptosis and suppresses the autoimmune response to melanocytes by reducing the release of heat shock protein 70 [106]. In another study, GB’s protective effect on melanocytes was shown to be dependent on activation of the Nrf-ARE pathway and downstream antioxidant genes, including NQO-1, SOD2 and haem oxygenase-1 [107]. Ginkgo biloba also has anti-inflammatory properties, reducing cyclooxygenase activity and decreasing IL-8 and vascular endothelial growth factor release in response to TNF-α [108]. A double-blind, placebo-controlled, clinical trial in 47 patients with vitiligo showed that the administration of GB 40 mg three times daily could halt disease progression and cause repigmentation of most perifacial vitiligo lesions [108]. Another prospective non-randomised study showed that a GB 60-mg dose twice daily can reduce disease severity in patients with vitiligo [109]. Although there was no evidence of severe side effects in the above studies, GB may be associated with the risk of clotting disorders or interactions with other medications. Therefore, the risks of taking GB may outweigh the known benefits of this supplement [110].
4.7 Catalase
Catalase is a haem-containing enzyme that catalyses the decomposition of H2O2 into water and molecular oxygen. During the enzymatic process leading to the dissociation of H2O2, catalase is first oxidised to a hypervalent iron intermediate known as compound I and then reduced back to its resting state by a second H2O2 molecule. Catalases are classified into three types based on their structure and sequence: monofunctional catalase or conventional catalase, catalase peroxidase and pseudocatalase or manganese catalase [111]. High concentrations of H2O2 in the epidermis are toxic to melanocytes because they inhibit tyrosinase and deactivate catalase. Some studies have shown a reduction in the catalase level compared with normal healthy controls [112, 113] In other studies, the catalase activity of erythrocytes from patients with vitiligo was not significantly different from that of healthy individuals [114, 115]. However, clinical trials showed that topical pseudocatalase/superoxide dismutase gel was ineffective in treating vitiligo [116, 117]. Similarly, a study combining pseudocatalase cream with NB-UVB did not appear to provide benefit for NB-UVB therapy [118]. The efficacy of pseudocatalase foam applied twice daily with NB-UVB twice weekly for 24 weeks has also not been proven [119], but in a study combining pseudocatalase cream twice daily with UVB irradiation and calcium chloride, complete repigmentation occurred on the face and dorsum of the hands in 90% of patients [120]. In the study with topical pseudocatalase cream (PC-KUS) with NB-UVB, repigmentation of more than 75% on the face and neck was achieved in 66 of 71 patients [121]. A faster onset of repigmentation was also achieved with a combination of pseudocatalase cream (PC-KUS), a Dead Sea bath and sun exposure over a treatment period of 21 days [122].
4.8 Superoxide Dismutase
Superoxide dismutase (SOD) is a metalloenzyme and performs the first step in the breakdown of the superoxide radical (O2−) to H2O2 and O2. Some studies have shown significantly lower activity of SOD in patients with generalised and localised vitiligo [15, 87, 113]. Opposing studies show higher levels of SOD, such as Dell’Anna et al. [93] or the meta-analysis by Shi et al. [123] and a study by Sravani et al. [112], which found a significant increase in the levels of SOD in the skin of people with vitiligo. However, there was no significant difference between the SOD levels in lesioned and normal skin in patients with vitiligo.
4.9 Alpha-Lipoic Acid
In its reduced dihydrolipoate form, alpha-lipoic acid (ALA) reacts with ROS and reactive nitrogen species and protects cell membranes by interfering with the vitamin C and E pathways [124]. Alpha-lipoic acid is found in small amounts in foods such as spinach and broccoli and is also synthesised in the liver [125]. In clinical trials, ALA has been tested in combination with other antioxidants such as vitamins C and E and polyunsaturated fatty acids in conjunction with NB-UVB phototherapy. Treatment with mixed antioxidants, in addition to NB-UVB, significantly improved the efficacy of phototherapy, with 47% of participants achieving > 75% repigmentation, compared with 18% in the control group [93]. Combination therapy with oral ALA, betamethasone injection and NB-UVB has also been shown to be beneficial in non-segmental progressive vitiligo [126]. It is known that ALA can accelerate the initial repigmentation response. However, combination therapy of ALA alone with NB-UVB has not shown any additional benefit in the treatment of non-segmental stable vitiligo [127].
4.10 Coenzyme Q10
Coenzyme Q10 (CoQ10) is a vitamin-like ubiquitous compound that is an electron transfer agent in the mitochondrial respiratory chain. It plays an important role in the metabolism of fatty acids, pyrimidines, and lysosomes and mediates the expression of several genes, including those involved in inflammation [128]. Although CoQ10 is widely recommended as a systemic antioxidant, topical application of creams containing CoQ10 may induce facial vitiligo in susceptible individuals. Coenzyme Q10-mediated reduction of ROS by activated PC-KUS leads to cessation of depigmentation and repigmentation [129]. In contrast, researchers in a randomised, single-blind, placebo-controlled trial in Iraq found a significant reduction in the Vitiligo Area Scoring Index after 8 weeks of topical gel application with CoQ10 [130].
4.11 Phyllanthus emblica
Phyllanthus emblica has a high antioxidant capacity owing to its high content of polyphenolic compounds and vitamin C. The fruit extract can be used as an antioxidant and can inhibit lipid peroxidation and scavenge free radicals such as superoxide anions, hydrogen radicals, hydrogen peroxide and nitric oxide radicals [131, 132]. One study investigated the effect of oral intake of fruit extracts in combination with vitamin E and carotenoids on repigmentation in patients with vitiligo after phototherapy [133]. A significantly higher repigmentation rate was observed when supplementing with a mixture of antioxidants. In combination with two or three servings of fresh P. emblica per day, the recommended daily amount of vitamin C can be achieved.
4.12 Polypodium leucotomos
Polypodium leucotomos (PL) is a fern species native to Central America. It was first studied in Honduras as a treatment for psoriasis [134]. Polypodium leucotomos primarily prevents lipid peroxidation and membrane damage in fibroblasts and keratinocytes. In addition, PLE inhibits the oxidation of glutathione and acts as a direct ROS absorber against superoxide anion, hydroxyl, oxygen and oxygen peroxide. It can stimulate IL-10 production and promote a shift in the cytokine profile from type 1 to type 2 T cells [135]. Middelkamp-Hup et al. compared the use of PL in combination with NB-UVB therapy in a double-blind, placebo-controlled, clinical trial [136]. The authors showed that patients taking the supplement had significantly more repigmentation in the head and neck area compared with the placebo. No significant differences were found in other body regions. In a study by Reyes et al. the researchers investigated the effect of adding PL to PUVA therapy. It was found that subjects receiving PUVA and PL had a higher proportion of patients with repigmentation of 50% or more [137]. A study by Pacifico et al. suggests that treatment with NB-UVB phototherapy in combination with PL increases the extent of repigmentation and thus shortens the repigmentation time in patients with diffuse vitiligo at lower cumulative doses [138].
5 Conclusions
Oxidative stress is an important factor in the destruction of melanocytes. Melanocytes are particularly susceptible to oxidative stress because they are involved in melanin formation, which produces ROS as a by-product. While apoptosis is widely recognised as the primary form of melanocyte death in vitiligo, other forms of cell death such as oxeiptosis and ferroptosis have also been considered, primarily because of their close association with oxidative stress. In-depth studies of the intricate relationships between oxidative stress and various forms of cell death in vitiligo are expected to provide crucial insights into the mechanisms underlying melanocyte loss. A comprehensive understanding of the mechanisms underlying the different types of cell death observed in vitiligo is a crucial first step in developing therapeutic strategies. Such studies may shed light on the underlying causes of melanocyte destruction and pave the way for interventions that prevent melanocyte degeneration and allow for long-lasting repigmentation, an important therapeutic target in vitiligo treatment. The use of antioxidants in the treatment of vitiligo may seem attractive because of their antioxidant-protective properties against oxidative stress. A literature review revealed that more research on such subjects is needed. In most cases, antioxidants taken as monotherapy fail to produce significant improvement in skin repigmentation. To achieve satisfactory results, they should be used in conjunction with standard pharmacology or phototherapy.
References
Bergqvist C, Ezzedine K. Vitiligo: a review. Dermatol Basel Switz. 2020;236:571–92. https://doi.org/10.1159/000506103.
Bergqvist C, Ezzedine K. Vitiligo: a focus on pathogenesis and its therapeutic implications. J Dermatol. 2021;48:252–70. https://doi.org/10.1111/1346-8138.15743.
Czajkowski R, Placek W, Flisiak I, Krasowska D, Maj J, Marchlewicz M, et al. Vitiligo. Diagnostic and therapeutic recommendations of the Polish Dermatological Society. Dermatol Rev/Przegl Dermatol. 2019;106:1–15. https://doi.org/10.5114/dr.2019.83440.
Zhang J, Hu W, Wang P, Ding Y, Wang H, Kang X. Research progress on targeted antioxidant therapy and vitiligo. Oxid Med Cell Longev. 2022;2022:1–10. https://doi.org/10.1155/2022/1821780.
Lyu C, Sun Y. Immunometabolism in the pathogenesis of vitiligo. Front Immunol. 2022;13:1055958. https://doi.org/10.3389/fimmu.2022.1055958.
Kang P, Chen J, Zhang W, Guo N, Yi X, Cui T, et al. Oxeiptosis: a novel pathway of melanocytes death in response to oxidative stress in vitiligo. Cell Death Discov. 2022;8:70. https://doi.org/10.1038/s41420-022-00863-3.
Zhou Y, Khan M, Jiang L, Fu C, Dong Y, Luo L, et al. The current status of antioxidants in the treatment of vitiligo in China. Oxid Med Cell Longev. 2022;2022:2994558. https://doi.org/10.1155/2022/2994558.
Taieb A, Alomar A, Böhm M, Dell’Anna ML, De Pase A, Eleftheriadou V, et al. Guidelines for the management of vitiligo: the European Dermatology Forum consensus. Br J Dermatol. 2013;168:5–19. https://doi.org/10.1111/j.1365-2133.2012.11197.x.
Mulayim MK, Kurutas EB, Nazik H, Ozturk P. Assessment of oxidative/nitrosative stress and raftlin in vitiligo. Indian J Dermatol. 2022;67:624. https://doi.org/10.4103/ijd.ijd_917_20.
Mitra S, De Sarkar S, Pradhan A, Pati AK, Pradhan R, Mondal D, et al. Levels of oxidative damage and proinflammatory cytokines are enhanced in patients with active vitiligo. Free Radic Res. 2017;51:986–94. https://doi.org/10.1080/10715762.2017.1402303.
Xie H, Zhou F, Liu L, Zhu G, Li Q, Li C, et al. Vitiligo: how do oxidative stress-induced autoantigens trigger autoimmunity? J Dermatol Sci. 2016;81:3–9. https://doi.org/10.1016/j.jdermsci.2015.09.003.
Chen J, Li S, Li C, Li CC. Mechanisms of melanocyte death in vitiligo. Med Res Rev. 2020;41:1138–66. https://doi.org/10.1002/med.21754.
Denat L, Kadekaro AL, Marrot L, Leachman SA, Abdel-Malek ZA. Melanocytes as instigators and victims of oxidative stress. J Invest Dermatol. 2014;134:1512–8. https://doi.org/10.1038/jid.2014.65.
Lu Y, Tonissen KF, Di Trapani G. Modulating skin colour: role of the thioredoxin and glutathione systems in regulating melanogenesis. Biosci Rep. 2021;41:BSR20210427. https://doi.org/10.1042/BSR20210427.
Mathachan SR, Khurana A, Gautam RK, Kulhari A, Sharma L, Sardana K. Does oxidative stress correlate with disease activity and severity in vitiligo? An analytical study. J Cosmet Dermatol. 2021;20:352–9. https://doi.org/10.1111/jocd.13499.
Said ER, Nagui NAER, Rashed LA, Mostafa WZ. Oxidative stress and the cholinergic system in non-segmental vitiligo: effect of narrow band ultraviolet b. Photodermatol Photoimmunol Photomed. 2021;37:306–12. https://doi.org/10.1111/phpp.12653.
Nie X-J, Hao B-Z, Zhang B-L, Li Y-Y. GATA3 ameliorates melanocyte injuries in vitiligo through SIRT3-mediated HMGB1 deacetylation. J Dermatol. 2023;50:472–84. https://doi.org/10.1111/1346-8138.16634.
Hu Y, Huang J, Li Y, Jiang L, Ouyang Y, Li Y, et al. Cistanche deserticola polysaccharide induces melanogenesis in melanocytes and reduces oxidative stress via activating NRF2/HO-1 pathway. J Cell Mol Med. 2020;24:4023–35. https://doi.org/10.1111/jcmm.15038.
Zhou J, An X, Dong J, Wang Y, Zhong H, Duan L, et al. IL-17 induces cellular stress microenvironment of melanocytes to promote autophagic cell apoptosis in vitiligo. FASEB J. 2018;32:4899–916. https://doi.org/10.1096/fj.201701242RR.
He S, Xu J, Wu J. The promising role of chemokines in vitiligo: from oxidative stress to the autoimmune response. Oxid Med Cell Longev. 2022;2022:8796735. https://doi.org/10.1155/2022/8796735.
Li S, Zhu G, Yang Y, Guo S, Dai W, Wang G, et al. Oxidative stress-induced chemokine production mediates CD8(+) T cell skin trafficking in vitiligo. J Investig Dermatol Symp Proc. 2015;17:32–3. https://doi.org/10.1038/jidsymp.2015.8.
Chen X, Guo W, Chang Y, Chen J, Kang P, Yi X, et al. Oxidative stress-induced IL-15 trans-presentation in keratinocytes contributes to CD8+ T cells activation via JAK-STAT pathway in vitiligo. Free Radic Biol Med. 2019;139:80–91. https://doi.org/10.1016/j.freeradbiomed.2019.05.011.
Li S, Zhu G, Yang Y, Jian Z, Guo S, Dai W, et al. Oxidative stress drives CD8+ T-cell skin trafficking in patients with vitiligo through CXCL16 upregulation by activating the unfolded protein response in keratinocytes. J Allergy Clin Immunol. 2017;140:177-89.e9. https://doi.org/10.1016/j.jaci.2016.10.013.
Ahn Y, Seo J, Lee EJ, Kim JY, Park M-Y, Hwang S, et al. ATP-P2X7-induced inflammasome activation contributes to melanocyte death and CD8+ T-cell trafficking to the skin in vitiligo. J Invest Dermatol. 2020;140:1794-804.e4. https://doi.org/10.1016/j.jid.2019.12.035.
Chang W-L, Ko C-H. The role of oxidative stress in vitiligo: an update on its pathogenesis and therapeutic implications. Cells. 2023;12:936. https://doi.org/10.3390/cells12060936.
Xuan Y, Yang Y, Xiang L, Zhang C. The role of oxidative stress in the pathogenesis of vitiligo: a culprit for melanocyte death. Oxid Med Cell Longev. 2022;2022:8498472. https://doi.org/10.1155/2022/8498472.
Jian Z, Li K, Liu L, Zhang Y, Zhou Z, Li C, et al. Heme oxygenase-1 protects human melanocytes from H2O2-induced oxidative stress via the Nrf2-ARE pathway. J Invest Dermatol. 2011;131:1420–7. https://doi.org/10.1038/jid.2011.56.
Białczyk A, Czajkowski R. Autophagy in psoriasis and vitiligo. Forum Dermatol. 2022;8:153–7. https://doi.org/10.5603/FD.a2022.0022.
He Y, Li S, Zhang W, Dai W, Cui T, Wang G, et al. Dysregulated autophagy increased melanocyte sensitivity to H2O2-induced oxidative stress in vitiligo. Sci Rep. 2017;7:42394. https://doi.org/10.1038/srep42394.
Sorour NE, Abd El-Kareem HM, Ibrahim AE, Salem RM. Nuclear factor erythroid-2-related factor 2 gene polymorphisms in vitiligo. J Clin Aesthetic Dermatol. 2021;14:14–7.
Ji Z, Liu G-H, Qu J. Mitochondrial sirtuins, metabolism, and aging. J Genet Genomics Yi Chuan Xue Bao. 2022;49:287–98. https://doi.org/10.1016/j.jgg.2021.11.005.
Yi X, Guo W, Shi Q, Yang Y, Zhang W, Chen X, et al. SIRT3-dependent mitochondrial dynamics remodeling contributes to oxidative stress-induced melanocyte degeneration in vitiligo. Theranostics. 2019;9:1614–33. https://doi.org/10.7150/thno.30398.
Hayran Y, Özge Ergen G, Özmen F. The relationship between non-segmental Vitiligo, HLA genotype and oxidative stress. Int J Clin Pract. 2021;75: e14024. https://doi.org/10.1111/ijcp.14024.
Huang Y-W, Tsai T-F. HLA-Cw1 and psoriasis. Am J Clin Dermatol. 2021;22:339–47. https://doi.org/10.1007/s40257-020-00585-1.
Tang D, Kang R, Berghe TV, Vandenabeele P, Kroemer G. The molecular machinery of regulated cell death. Cell Res. 2019;29:347–64. https://doi.org/10.1038/s41422-019-0164-5.
Galluzzi L, Bravo-San Pedro JM, Vitale I, Aaronson SA, Abrams JM, Adam D, et al. Essential versus accessory aspects of cell death: recommendations of the NCCD 2015. Cell Death Differ. 2015;22:58–73. https://doi.org/10.1038/cdd.2014.137.
Wu X, Yang Y, Xiang L, Zhang C. The fate of melanocyte: mechanisms of cell death in vitiligo. Pigment Cell Melanoma Res. 2021;34:256–67. https://doi.org/10.1111/pcmr.12955.
Sastry KS, Naeem H, Mokrab Y, Chouchane AI. RNA-seq reveals dysregulation of novel melanocyte genes upon oxidative stress: implications in vitiligo pathogenesis. Oxid Med Cell Longev. 2019;2019:2841814. https://doi.org/10.1155/2019/2841814.
D’Arcy MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43:582–92. https://doi.org/10.1002/cbin.11137.
Elmore S. Apoptosis: a review of programmed cell death. Toxicol Pathol. 2007;35:495–516. https://doi.org/10.1080/01926230701320337.
Campioni M, Santini D, Tonini G, Murace R, Dragonetti E, Spugnini EP, et al. Role of Apaf-1, a key regulator of apoptosis, in melanoma progression and chemoresistance. Exp Dermatol. 2005;14:811–8. https://doi.org/10.1111/j.1600-0625.2005.00360.x.
McIlwain DR, Berger T, Mak TW. Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol. 2013;5: a008656. https://doi.org/10.1101/cshperspect.a008656.
Raisova M, Hossini AM, Eberle J, Riebeling C, Wieder T, Sturm I, et al. The Bax/Bcl-2 ratio determines the susceptibility of human melanoma cells to CD95/Fas-mediated apoptosis. J Invest Dermatol. 2001;117:333–40. https://doi.org/10.1046/j.0022-202x.2001.01409.x.
Kumar R, Parsad D, Kanwar AJ, Kaul D. Altered levels of LXR-α: crucial implications in the pathogenesis of vitiligo. Exp Dermatol. 2012;21:853–8. https://doi.org/10.1111/exd.12017.
Park K, Lee SE, Shin K-O, Uchida Y. Insights into the role of endoplasmic reticulum stress in skin function and associated diseases. FEBS J. 2019;286:413–25. https://doi.org/10.1111/febs.14739.
Liu X, Hussain R, Mehmood K, Tang Z, Zhang H, Li Y. Mitochondrial-endoplasmic reticulum communication-mediated oxidative stress and autophagy. BioMed Res Int. 2022;2022:6459585. https://doi.org/10.1155/2022/6459585.
Chen J, Zhuang T, Chen J, Tian Y, Yi X, Ni Q, et al. Homocysteine induces melanocytes apoptosis via PERK-eIF2α-CHOP pathway in vitiligo. Clin Sci Lond. 2020;134:1127–41. https://doi.org/10.1042/CS20200218.
Lossi L. The concept of intrinsic versus extrinsic apoptosis. Biochem J. 2022;479:357–84. https://doi.org/10.1042/BCJ20210854.
Jimbo H, Nagai H, Fujiwara S, Shimoura N, Nishigori C. Fas-FasL interaction in cytotoxic T cell-mediated vitiligo: the role of lesional expression of tumor necrosis factor-α and interferon-γ in Fas-mediated melanocyte apoptosis. Exp Dermatol. 2020;29:61–70. https://doi.org/10.1111/exd.14053.
Tulic MK, Cavazza E, Cheli Y, Jacquel A, Luci C, Cardot-Leccia N, et al. Innate lymphocyte-induced CXCR3B-mediated melanocyte apoptosis is a potential initiator of T-cell autoreactivity in vitiligo. Nat Commun. 2019;10:2178. https://doi.org/10.1038/s41467-019-09963-8.
Shi Q, Zhang W, Guo S, Jian Z, Li S, Li K, et al. Oxidative stress-induced overexpression of miR-25: the mechanism underlying the degeneration of melanocytes in vitiligo. Cell Death Differ. 2016;23:496–508. https://doi.org/10.1038/cdd.2015.117.
Mou K, Liu W, Miao Y, Cao F, Li P. HMGB1 deficiency reduces H2O2-induced oxidative damage in human melanocytes via the Nrf2 pathway. J Cell Mol Med. 2018;22:6148–56. https://doi.org/10.1111/jcmm.13895.
Kim JY, Lee EJ, Seo J, Oh SH. Impact of high-mobility group box 1 on melanocytic survival and its involvement in the pathogenesis of vitiligo. Br J Dermatol. 2017;176:1558–68. https://doi.org/10.1111/bjd.15151.
Jorgensen I, Miao EA. Pyroptotic cell death defends against intracellular pathogens. Immunol Rev. 2015;265:130–42. https://doi.org/10.1111/imr.12287.
Shi J, Gao W, Shao F. Pyroptosis: gasdermin-mediated programmed necrotic cell death. Trends Biochem Sci. 2017;42:245–54. https://doi.org/10.1016/j.tibs.2016.10.004.
Liu X, Zhang Z, Ruan J, Pan Y, Magupalli VG, Wu H, et al. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature. 2016;535:153–8. https://doi.org/10.1038/nature18629.
Faria SS, Costantini S, de Lima VCC, de Andrade VP, Rialland M, Cedric R, et al. NLRP3 inflammasome-mediated cytokine production and pyroptosis cell death in breast cancer. J Biomed Sci. 2021;28:26. https://doi.org/10.1186/s12929-021-00724-8.
Kelley N, Jeltema D, Duan Y, He Y. The NLRP3 inflammasome: an overview of mechanisms of activation and regulation. Int J Mol Sci. 2019;20:3328. https://doi.org/10.3390/ijms20133328.
Marie J, Kovacs D, Pain C, Jouary T, Cota C, Vergier B, et al. Inflammasome activation and vitiligo/nonsegmental vitiligo progression. Br J Dermatol. 2014;170:816–23. https://doi.org/10.1111/bjd.12691.
Zhou Z, He H, Wang K, Shi X, Wang Y, Su Y, et al. Granzyme A from cytotoxic lymphocytes cleaves GSDMB to trigger pyroptosis in target cells. Science. 2020;368: eaaz7548. https://doi.org/10.1126/science.aaz7548.
Nirmala JG, Lopus M. Cell death mechanisms in eukaryotes. Cell Biol Toxicol. 2020;36:145–64. https://doi.org/10.1007/s10565-019-09496-2.
Mozaffari S, Abdollahi M. Chapter 3. Mitochondrial contribution to cell death. In: Pourahmad J, Rezaei M, editors. Mitochondrial metabolism. New York: Academic Press; 2021. p. 49–60. https://doi.org/10.1016/B978-0-12-822416-8.00004-X.
Chaouhan HS, Vinod C, Mahapatra N, Yu S-H, Wang I-K, Chen K-B, et al. Necroptosis: a pathogenic negotiator in human diseases. Int J Mol Sci. 2022;23:12714. https://doi.org/10.3390/ijms232112714.
Zhang Y, Su SS, Zhao S, Yang Z, Zhong C-Q, Chen X, et al. RIP1 autophosphorylation is promoted by mitochondrial ROS and is essential for RIP3 recruitment into necrosome. Nat Commun. 2017;8:14329. https://doi.org/10.1038/ncomms14329.
Li B, Yi X, Zhuang T, Zhang S, Li S, Yang Y, et al. RIP1-mediated necroptosis facilitates oxidative stress-induced melanocyte death, offering insight into vitiligo. J Invest Dermatol. 2021;141:2921-31.e6. https://doi.org/10.1016/j.jid.2020.06.042.
Li J, Cao F, Yin H, Huang Z, Lin Z, Mao N, et al. Ferroptosis: past, present and future. Cell Death Dis. 2020;11:88. https://doi.org/10.1038/s41419-020-2298-2.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology, and role in disease. Nat Rev Mol Cell Biol. 2021;22:266. https://doi.org/10.1038/S41580-020-00324-8.
Chen X, Kang R, Kroemer G, Tang D. Broadening horizons: the role of ferroptosis in cancer. Nat Rev Clin Oncol. 2021;18:280–96. https://doi.org/10.1038/s41571-020-00462-0.
Liu P, Wang W, Li Z, Li Y, Yu X, Tu J, et al. Ferroptosis: a new regulatory mechanism in osteoporosis. Oxid Med Cell Longev. 2022;2022:2634431. https://doi.org/10.1155/2022/2634431.
Wang J, Liu Y, Wang Y, Sun L. The cross-link between ferroptosis and kidney diseases. Oxid Med Cell Longev. 2021;2021: e6654887. https://doi.org/10.1155/2021/6654887.
Yu Y, Yan Y, Niu F, Wang Y, Chen X, Su G, et al. Ferroptosis: a cell death connecting oxidative stress, inflammation and cardiovascular diseases. Cell Death Discov. 2021;7:193. https://doi.org/10.1038/s41420-021-00579-w.
Xuan Y, Yang Y, Xiang L, Zhang C. The role of oxidative stress in the pathogenesis of vitiligo: a culprit for melanocyte death. Oxid Med Cell Longev. 2022;2022: 849847. https://doi.org/10.1155/2022/8498472.
Zhao Y, Li Y, Zhang R, Wang F, Wang T, Jiao Y. The role of erastin in ferroptosis and its prospects in cancer therapy. Onco Targets Ther. 2020;13:5429–41. https://doi.org/10.2147/OTT.S254995.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060–72. https://doi.org/10.1016/j.cell.2012.03.042.
Xiao B-H, Shi M, Chen H, Cui S, Wu Y, Gao X-H, et al. Glutathione peroxidase level in patients with vitiligo: a meta-analysis. BioMed Res Int. 2016;2016:3029810. https://doi.org/10.1155/2016/3029810.
Yang M, Li X, Li H, Zhang X, Liu X, Song Y. Baicalein inhibits RLS3-induced ferroptosis in melanocytes. Biochem Biophys Res Commun. 2021;561:65–72. https://doi.org/10.1016/J.BBRC.2021.05.010.
Wu X, Jin S, Yang Y, Lu X, Dai X, Xu Z, et al. Altered expression of ferroptosis markers and iron metabolism reveals a potential role of ferroptosis in vitiligo. Pigment Cell Melanoma Res. 2022;35:328–41. https://doi.org/10.1111/pcmr.13032.
Holze C, Michaudel C, Mackowiak C, Haas DA, Benda C, Hubel P, et al. Oxeiptosis, a ROS-induced caspase-independent apoptosis-like cell-death pathway. Nat Immunol. 2018;19:130–40. https://doi.org/10.1038/s41590-017-0013-y.
Tian C, Liu Y, Li Z, Zhu P, Zhao M. Mitochondria related cell death modalities and disease. Front Cell Dev Biol. 2022;10: 832356. https://doi.org/10.3389/fcell.2022.832356.
Michalak M. Plant-derived antioxidants: significance in skin health and the ageing process. Int J Mol Sci. 2022;23:585. https://doi.org/10.3390/ijms23020585.
Pai VV, Shukla P, Kikkeri NN. Antioxidants in dermatology. Indian Dermatol Online J. 2014;5:210. https://doi.org/10.4103/2229-5178.131127.
Jung YY, Ha IJ, Lee M, Ahn KS. Skin improvement with antioxidant effect of Yuja (Citrus junos) peel fractions: wrinkles, moisturizing, and whitening. Antioxidants (Basel). 2022;12:51. https://doi.org/10.3390/antiox12010051.
Masaki H. Role of antioxidants in the skin: anti-aging effects. J Dermatol Sci. 2010;58:85–90. https://doi.org/10.1016/j.jdermsci.2010.03.003.
Sable KA, Shields BE. The role of dietary antioxidants in melanoma and nonmelanoma skin cancer. Cutis. 2023;111:33–48. https://doi.org/10.12788/cutis.0672.
Guarneri F, Bertino L, Pioggia G, Casciaro M, Gangemi S. Therapies with antioxidant potential in psoriasis, vitiligo, and lichen planus. Antioxidants. 2021;10:1087. https://doi.org/10.3390/antiox10071087.
Xing X, Dan Y, Xu Z, Xiang L. Implications of oxidative stress in the pathogenesis and treatment of hyperpigmentation disorders. Oxid Med Cell Longev. 2022;2022:7881717. https://doi.org/10.1155/2022/7881717.
Khan R, Satyam A, Gupta S, Sharma VK, Sharma A. Circulatory levels of antioxidants and lipid peroxidation in Indian patients with generalized and localized vitiligo. Arch Dermatol Res. 2009;301:731–7. https://doi.org/10.1007/s00403-009-0964-4.
Sendrasoa FA, Ranaivo IM, Sata M, Andrianarison M, Raharolahy O, Rakotoarisaona MF, et al. Treatment responses in patients with vitiligo to very potent topical corticosteroids combined with vitaminotherapy in Madagascar. Int J Dermatol. 2019;58:908–11. https://doi.org/10.1111/ijd.14510.
Sardana K, Sachdeva S. Role of nutritional supplements in selected dermatological disorders: a review. J Cosmet Dermatol. 2022;21:85–98. https://doi.org/10.1111/JOCD.14436.
Liu X, Yang G, Luo M, Lan Q, Shi X, Deng H, et al. Serum vitamin E levels and chronic inflammatory skin diseases: a systematic review and meta-analysis. PLoS ONE. 2021;16: e0261259. https://doi.org/10.1371/JOURNAL.PONE.0261259.
Akyol M, Celik VK, Ozcelik S, Polat M, Marufihah M, Atalay A. The effects of vitamin E on the skin lipid peroxidation and the clinical improvement in vitiligo patients treated with PUVA. Eur J Dermatol. 2002;12:24–6.
Elgoweini M, Din NNE. Response of vitiligo to narrowband ultraviolet B and oral antioxidants. J Clin Pharmacol/. 2009;49:852–5. https://doi.org/10.1177/0091270009335769.
Dell’Anna ML, Mastrofrancesco A, Sala R, Venturini M, Ottaviani M, Vidolin AP, et al. Antioxidants and narrow band-UVB in the treatment of vitiligo: a double-blind placebo controlled trial. Clin Exp Dermatol. 2007;32:631–6. https://doi.org/10.1111/J.1365-2230.2007.02514.X.
Nowowiejska L, Marek-Józefowicz L, Szewczyk-Golec K, Woźniak A, Basałygo M, Zegarska B, et al. Influence of oral supplementation of vitamins A and E on the effectiveness of vitiligo treatment. Dermatol Ther. 2023;2023:1–7. https://doi.org/10.1155/2023/3777109.
Dai T, Xiaoying S, Li X, Hongjin L, Yaqiong Z, Bo L. Selenium level in patients with vitiligo: a meta-analysis. BioMed Res Int. 2020;2020:7580939. https://doi.org/10.1155/2020/7580939.
Lv J, Ai P, Lei S, Zhou F, Chen S, Zhang Y. Selenium levels and skin diseases: systematic review and meta-analysis. J Trace Elem Med Biol. 2020;62: 126548. https://doi.org/10.1016/j.jtemb.2020.126548.
Huo J, Liu T, Huan Y, Li F, Wang R. Serum level of antioxidant vitamins and minerals in patients with vitiligo, a systematic review and meta-analysis. J Trace Elem Med Biol. 2020;62: 126570. https://doi.org/10.1016/j.jtemb.2020.126570.
Sanad EM, El-Fallah AA, Al-Doori AR, Salem RM. Serum zinc and inflammatory cytokines in vitiligo. J Clin Aesthetic Dermatol. 2020;13:S29.
Zaki AM, Nada AS, Elshahed AR, Abdelgawad NH, Jafferany M, Elsaie ML. Therapeutic implications of assessment of serum zinc levels in patients with vitiligo: a patient controlled prospective study. Dermatol Ther. 2020;33: e13998. https://doi.org/10.1111/dth.13998.
Bagherani N, Yaghoobi R, Omidian M. Hypothesis: zinc can be effective in treatment of vitiligo. Indian J Dermatol. 2011;56:480–4. https://doi.org/10.4103/0019-5154.87116.
Osredkar J. Copper and zinc, biological role and significance of copper/zinc imbalance. J Clin Toxicol. 2011;S3:001. https://doi.org/10.4172/2161-0495.S3-001.
Wacewicz M, Socha K, Soroczyńska J, Niczyporuk M, Aleksiejczuk P, Ostrowska J, et al. Selenium, zinc, copper, Cu/Zn ratio and total antioxidant status in the serum of vitiligo patients treated by narrow-band ultraviolet-B phototherapy. J Dermatol Treat. 2018;29:190–5. https://doi.org/10.1080/09546634.2017.1357797.
Zeng Q, Yin J, Fan F, Chen J, Zuo C, Xiang Y, et al. Decreased copper and zinc in sera of Chinese vitiligo patients: a meta-analysis. J Dermatol. 2014;41:245–51. https://doi.org/10.1111/1346-8138.12392.
Khoshdel Z, Gholijani N, Niknam M, Rahmani N, Hemmati-Dinarvand M, Naghibalhossaini F. Serum copper and zinc levels among Iranian vitiligo patients. Dermatol Pract Concept. 2022;12: e2022140. https://doi.org/10.5826/dpc.1204a140.
Narang I, Deb Barman K, Sahoo B, Lali P. Evaluation of serum levels of zinc and copper in vitiligo. Pigment Int. 2021;8:42–7. https://doi.org/10.4103/Pigmentinternational.Pigmentinternational.
Lu L, Wang S, Fu L, Liu D, Zhu Y, Xu A. Bilobalide protection of normal human melanocytes from hydrogen peroxide-induced oxidative damage via promotion of antioxidase expression and inhibition of endoplasmic reticulum stress. Clin Exp Dermatol. 2016;41:64–73. https://doi.org/10.1111/ced.12664.
Zhang S, Yi X, Su X, Jian Z, Cui T, Guo S, et al. Ginkgo biloba extract protects human melanocytes from H2O2-induced oxidative stress by activating Nrf2. J Cell Mol Med. 2019;23:5193–9. https://doi.org/10.1111/jcmm.14393.
Parsad D, Pandhi R, Juneja A. Effectiveness of oral Ginkgo biloba in treating limited, slowly spreading vitiligo. Clin Exp Dermatol. 2003;28:285–7.
Szczurko O, Shear N, Taddio A, Boon H. Ginkgo biloba for the treatment of vitiligo vulgaris: an open label pilot clinical trial. BMC Complement Altern Med. 2011;15:21. https://doi.org/10.1186/1472-6882-11-21.
Jamgochian M, Alamgir M, Rao B. Diet in dermatology: review of diet’s influence on the conditions of rosacea, hidradenitis suppurativa, herpes labialis, and vitiligo. Am J Lifestyle Med. 2021;17:152–60. https://doi.org/10.1177/15598276211026592.
Mahomoodally MF, Désiré A-LD, Rosette MA-LE. Catalase. In: Nabavi SM, Silva AS, editors. Antioxidants effects in health. Amsterdam: Elsevier; 2022. p. 81–90. https://doi.org/10.1016/B978-0-12-819096-8.00022-7.
Sravani PV, Babu NK, Gopal KVT, Rao GRR, Rao AR, Moorthy B, et al. Determination of oxidative stress in vitiligo by measuring superoxide dismutase and catalase levels in vitiliginous and non-vitiliginous skin. Indian J Dermatol Venereol Leprol. 2009;75:268–71. https://doi.org/10.4103/0378-6323.48427.
Li S, Dai W, Wang S, Kang P, Ye Z, Han P, et al. Clinical significance of serum oxidative stress markers to assess disease activity and severity in patients with non-segmental vitiligo. Front Cell Dev Biol. 2021;9: 739413. https://doi.org/10.3389/fcell.2021.739413.
Ines D, Sonia B, Riadh BM, Amel EG, Slaheddine M, Hamida T, et al. A comparative study of oxidant-antioxidant status in stable and active vitiligo patients. Arch Dermatol Res. 2006;298:147–52. https://doi.org/10.1007/s00403-006-0680-2.
Hazneci E, Karabulut AB, Ozturk C, Batcioglu K, Dogan G, Karaca S, et al. A comparative study of superoxide dismutase, catalase, and glutathione peroxidase activities and nitrate levels in vitiligo patients. Int J Dermatol. 2005;44:636–40. https://doi.org/10.1111/j.1365-4632.2004.02027.x.
Naini FF, Shooshtari AV, Ebrahimi B, Molaei R. The effect of pseudocatalase/superoxide dismutase in the treatment of vitiligo: a pilot study. J Res Pharm Pract. 2012;1:77–80. https://doi.org/10.4103/2279-042X.108375.
Alshiyab DM, Al-Qarqaz FA, Muhaidat JM, Alkhader YS, Al-Sheyab RF, Jafaar SI. Comparison of the efficacy of tacrolimus 0.1% ointment and tacrolimus 0.1% plus topical pseudocatalase/superoxide dismutase gel in children with limited vitiligo: a randomized controlled trial. J Dermatol Treat. 2022;33:146–9. https://doi.org/10.1080/09546634.2020.1729952.
Bakis-Petsoglou S, Le Guay JL, Wittal R. A randomized, double-blinded, placebo-controlled trial of pseudocatalase cream and narrowband ultraviolet B in the treatment of vitiligo. Br J Dermatol. 2009;161:910–7. https://doi.org/10.1111/j.1365-2133.2009.09252.x.
Patel DC, Evans AV, Hawk JLM. Topical pseudocatalase mousse and narrowband UVB phototherapy is not effective for vitiligo: an open, single-centre study. Clin Exp Dermatol. 2002;27:641–4. https://doi.org/10.1046/J.1365-2230.2002.01142.X.
Schallreuter KU, Wood JM, Lemke KR, Levenig C. Treatment of vitiligo with a topical application of pseudocatalase and calcium in combination with short-term UVB exposure: a case study on 33 patients. Dermatology. 1995;190:223–9. https://doi.org/10.1159/000246690.
Schallreuter KU, Krüger C, Würfel BA, Panske A, Wood JM. From basic research to the bedside: efficacy of topical treatment with pseudocatalase PC-KUS in 71 children with vitiligo. Int J Dermatol. 2008;47:743–53. https://doi.org/10.1111/j.1365-4632.2008.03660.x.
Schallreuter KU, Moore J, Behrens-Williams S, Panske A, Harari M. Rapid initiation of repigmentation in vitiligo with Dead Sea climatotherapy in combination with pseudocatalase (PC-KUS). Int J Dermatol. 2002;41:482–7. https://doi.org/10.1046/J.1365-4362.2002.01463.X.
Shi M-H, Wu Y, Li L, Cai Y-F, Liu M, Gao X-H, et al. Meta-analysis of the association between vitiligo and the level of superoxide dismutase or malondialdehyde. Clin Exp Dermatol. 2017;42:21–9. https://doi.org/10.1111/ced.12950.
Shakhbazova A, Wu H, Chambers CJ, Sivamani RK. A systematic review of nutrition, supplement, and herbal-based adjunctive therapies for vitiligo. J Altern Complement Med. 2021;27:294–311. https://doi.org/10.1089/acm.2020.0292.
Tóth F, Cseh EK, Vécsei L. Natural molecules and neuroprotection: kynurenic acid, pantethine and α-lipoic acid. Int J Mol Sci. 2021;22:403. https://doi.org/10.3390/ijms22010403.
Li L, Li L, Wu Y, Gao XH, Chen HD. Triple-combination treatment with oral α-lipoic acid, betamethasone injection, and NB-UVB for non-segmental progressive vitiligo. J Cosmet Laser Ther. 2016;18:182–5. https://doi.org/10.3109/14764172.2015.1114646.
Sun Y, Guan X, Wang H, Zhang J, Gu H, Lu H, et al. Randomized clinical trial of combined therapy with oral α-lipoic acid and NB-UVB for nonsegmental stable vitiligo. Dermatol Ther. 2021;34: e14610. https://doi.org/10.1111/dth.14610.
Hargreaves I, Heaton RA, Mantle D. Molecular sciences disorders of human coenzyme Q10 metabolism: an overview. Int J Mol Sci. 2020;21:6695. https://doi.org/10.3390/ijms21186695.
Schallreuter KU. Q10-triggered facial vitiligo. Br J Dermatol. 2013;169:1333–6. https://doi.org/10.1111/bjd.12587.
Hameed MF, Abu-Raghif AR, Farhood IG, Ali NM. The effectiveness of systemic co-enzyme Q10 in vitiligo. Iraqi J Med Sci. 2013;11:67–71.
Li Y, Sun HY, Yu XY, Liu D, Wan HX. Evaluation of cellular antioxidant and antiproliferative activities of five main Phyllanthus emblica L. cultivars in China. Indian J Pharm Sci. 2015;77:274. https://doi.org/10.4103/0250-474X.159608.
Di Nardo V, Barygina V, França K, Tirant M, Valle Y, Lotti T. Functional nutrition as integrated approach in vitiligo management. Dermatol Ther. 2019;32: e12625. https://doi.org/10.1111/dth.12625.
Colucci R, Dragoni F, Conti R, Pisaneschi L, Lazzeri L, Moretti S. Evaluation of an oral supplement containing Phyllanthus emblica fruit extracts, vitamin E, and carotenoids in vitiligo treatment. Dermatol Ther. 2015;28:17–21. https://doi.org/10.1111/dth.12172.
Thompson KG, Kim N. Dietary supplements in dermatology: a review of the evidence for zinc, biotin, vitamin D, nicotinamide, and Polypodium. J Am Acad Dermatol. 2021;84:1042–50. https://doi.org/10.1016/j.jaad.2020.04.123.
Calzari P, Vaienti S, Nazzaro G. Uses of Polypodium leucotomos extract in oncodermatology. J Clin Med. 2023;12:673. https://doi.org/10.3390/jcm12020673.
Middelkamp-Hup MA, Bos JD, Rius-diaz F, Gonzalez S, Westerhof W. Treatment of vitiligo vulgaris with narrow-band UVB and oral Polypodium leucotomos extract: a randomized double-blind placebo-controlled study. J Eur Acad Dermatol Venereol. 2007;21:942–50. https://doi.org/10.1111/J.1468-3083.2006.02132.X.
Reyes E, Jaén P, Heras EDL, Eusebio ED, Carrión F, Cuevas J, et al. Systemic immunomodulatory effects of Polypodium leucotomos as an adjuvant to PUVA therapy in generalized vitiligo: a pilot study. J Dermatol Sci. 2006;41:213–6. https://doi.org/10.1016/J.JDERMSCI.2005.12.006.
Pacifico A, Damiani G, Iacovelli P, Conic RRZ, Gonzalez S, Morrone A. NB-UVB plus oral Polypodium leucotomos extract display higher efficacy than NB-UVB alone in patients with vitiligo. Dermatol Ther. 2021;34: e14776. https://doi.org/10.1111/dth.14776.
Acknowledgements
Special thanks to Małgorzata Grochocka for her assistance in preparing the graphics and Julia Mackenzie for proofreading the article. Figure 2 was created with BioRender.com.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The open access fee was funded by Nicolaus Copernicus University in Torun.
Conflict of interest
Aleksandra Białczyk, Adam Wełniak, Barbara Kamińska and Rafał Czajkowski have no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
This article does not contain any studies with human participants or animals.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Author contributions
Conceptualisation: AB, AW; literature search and data analysis: AB, AW, BK; writing, original draft preparation: AB, AW, BK; writing, review and editing: RC; supervision: RC. All authors read and approved the final manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Białczyk, A., Wełniak, A., Kamińska, B. et al. Oxidative Stress and Potential Antioxidant Therapies in Vitiligo: A Narrative Review. Mol Diagn Ther 27, 723–739 (2023). https://doi.org/10.1007/s40291-023-00672-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-023-00672-z