Abstract
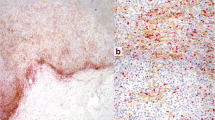
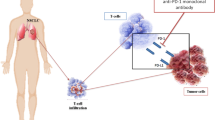
Immunotherapy with programmed death 1 (PD-1)- and programmed death-ligand 1 (PD-L1)-targeted monoclonal antibodies has dramatically changed the therapeutic and prognostic landscape for several types of malignancy. PD-1 and PD-L1 are immune checkpoint proteins whose binding ultimately result in T cell exhaustion and self-tolerance. Blocking this pathway ‘releases the brakes’ on the immune system and allows for attack of tumor cells that express PD-L1. The clinical trials that led to the US Food and Drug Administration (FDA) approval of these agents used different immunohistochemical (IHC) platforms with various PD-L1 antibodies to assess for PD-L1 expression on either tumor cells or tumor-infiltrating immune cells. There are four PD-L1 IHC assays registered with the FDA, using four different PD-L1 antibodies (22C3, 28-8, SP263, SP142), on two different IHC platforms (Dako and Ventana), each with their own scoring systems. Attempts at harmonization of PD-L1 IHC antibodies and staining platforms are underway. While PD-L1 IHC can be used to predict the likelihood of response to anti-PD-1 or anti-PD-L1 therapy, a proportion of patients that are negative can have a response and identification of alternative biomarkers is critical to further refine selection of patients most likely to respond to these therapies.
Similar content being viewed by others
References
Patel SP, Kurzrock R. PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther. 2015;14(4):847–56.
Swaika A, Hammond WA, Joseph RW. Current state of anti-PD-L1 and anti-PD-1 agents in cancer therapy. Mol Immunol. 2015;67(2 Part A):4–17.
Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti–PD-1 antibody in cancer. N Engl J Med. 2012;366(26):2443–54.
Taube JM, Klein A, Brahmer JR, et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to Anti–PD-1 therapy. Clin Cancer Res. 2014;20(19):5064–74.
Brahmer J, Reckamp KL, Baas P, et al. Nivolumab versus docetaxel in advanced squamous-cell non–small-cell lung cancer. N Engl J Med. 2015;373(2):123–35.
Borghaei H, Paz-Ares L, Horn L, et al. Nivolumab versus docetaxel in advanced nonsquamous non–small-cell lung cancer. N Engl J Med. 2015;373(17):1627–39.
Borghaei H, Brahmer JR, Horn L, et al. Nivolumab (nivo) vs docetaxel (doc) in patients (pts) with advanced NSCLC: CheckMate 017/057 2-y update and exploratory cytokine profile analyses [abstract]. J Clin Oncol. 2016;34(15 suppl):9025.
Garon EB, Rizvi NA, Hui R, et al. KEYNOTE-001 Investigators. Pembrolizumab for the treatment of non–small-cell lung cancer. N Engl J Med. 2015;372(21):2018–28.
Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–50.
Reck M, Rodriguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N Engl J Med. 2016;375:1823–33.
Langer CJ, Gadgeel SM, Borghaei H, KEYNOTE-021 investigators, et al. Carboplatin and pemetrexed with or without pembrolizumab for advanced, non-squamous non-small-cell lung cancer: a randomised, phase 2 cohort of the open-label KEYNOTE-021 study. Lancet Oncol. 2016;17(11):1497–508.
Socinski M, Creelan B, Horn L, et al. NSCLC, metastaticCheckMate 026: a phase 3 trial of nivolumab vs investigator’s choice (IC) of platinum-based doublet chemotherapy (PT-DC) as first-line therapy for stage iv/recurrent programmed death ligand 1 (PD-L1) − positive NSCLC [abstract]. Ann Oncol 2016;27(suppl 6). https://doi.org/10.1093/annonc/mdw435.39 (LBA7_PR-LBA7_PR).
Carbone DP, Reck M, Paz-Ares L, et al. First-line nivolumab in stage IV or recurrent non–small-cell lung cancer. N Engl J Med. 2017;376(25):2415–26.
Fehrenbacher L, Spira A, Ballinger M, POPLAR Study Group, et al. Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet. 2016;387(10030):1837–46.
Roche, Media Release, Basel, 27 September 2015. Two positive studies of Roche’s investigational cancer immunotherapy atezolizumab in specific type of lung cancer presented at 2015 European Cancer Congress. F. Hoffmann-La Roche Ltd. 2015. http://www.roche.com/media/store/releases/med-cor-2015-09-27.htm. Accessed 24 Oct 2017.
Rittmeyer A, Balesi F, Waterkamp D, et al. OAK Study Group. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. 2017;389:255–65.
Rizvi N, Brahmer JR, Ou S-HI, et al. Safety and clinical activity of MEDI4736, an anti-programmed cell death-ligand 1 (PD-L1) antibody, in patients with non-small cell lung cancer (NSCLC) [abstract]. J Clin Oncol. 2015;33(Suppl):8032.
Gulley JL, Rajan A, Spigel DR, et al. Avelumab for patients with previously treated metastatic or recurrent non-small-cell lung cancer (JAVELIN Solid Tumor): dose-expansion cohort of a multicentre, open-label, phase 1b trial. Lancet Oncol. 2017;18(5):599–610.
Hui R, Gandhi L, Carcereny Costa E, et al. Long-term OS for patients with advanced NSCLC enrolled in the KEYNOTE-001 study of pembrolizumab (pembro) [abstract]. J Clin Oncol. 2016;34(15 suppl):9026.
Baas P, Garon EB, Herbst RS, et al. Relationship between level of PD-L1 expression and outcomes in the KEYNOTE-010 study of pembrolizumab vs docetaxel for previously treated, PD-L1-Positive NSCLC [abstract]. J Clin Oncol. 2016;34(15 suppl):9015.
Rizvi NA, Hellmann MD, Brahmer JR, et al. Nivolumab in combination with platinum-based doublet chemotherapy for first-line treatment of advanced non–small-cell lung cancer. J Clin Oncol. 2016;34(25):2969–79.
Gettinger S, Rizvi NA, Chow LQ, et al. Nivolumab monotherapy for first-line treatment of advanced non–small-cell lung cancer. J Clin Oncol. 2016;34(25):2980–7.
Smith DA, Vansteenkiste JF, Fehrenbacher L, et al. Updated survival and biomarker analyses of a randomized phase II study of atezolizumab vs docetaxel in 2L/3L NSCLC (POPLAR) [abstract]. J Clin Oncol. 2016;34(15 suppl):9028.
Barlesi F, Park K, Ciardiello F, et al. Primary analysis from OAK, a randomized phase III study comparing atezolizumab with docetaxel in 2L/3L NSCLC [abstract]. Ann Oncol 2016;27(suppl 6):9): ix139–ix156. https://doi.org/10.1093/annonc/mdw594 (LBA44_PR).
Gubens MA, Sequist LV, Stevenson J, et al. Phase I/II study of pembrolizumab (pembro) plus ipilimumab (ipi) as second-line therapy for NSCLC: KEYNOTE-021 cohorts D and H [abstract]. J Clin Oncol. 2016;34(15 suppl):9027.
Antonia S, Goldberg SB, Balmanoukian A, et al. Safety and antitumour activity of durvalumab plus tremelimumab in non-small cell lung cancer: a multicentre, phase 1b study. Lancet Oncol. 2016;17(3):299–308.
Hellmann MD, Rizvi NA, Goldman JW, et al. Nivolumab plus ipilimumab as first-line treatment for advanced non-small-cell lung cancer (CheckMate 012): results of an open-label, phase 1, multicohort study. Lancet Oncol. 2017;18(1):31–41.
Planchard D, Yokoi T, McCleod MJ, et al. A phase III study of durvalumab (MEDI4736) with or without tremelimumab for previously treated patients with advanced NSCLC: rationale and protocol design of the ARCTIC study. Clin Lung Cancer. 2016;17(3):232–236.e1.
Spranger S, Spaapen RM, Zha Y, et al. Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med. 2013;5(200):200ra116.
McLaughlin J, Han G, Schalper KA, et al. Quantitative assessment of the heterogeneity of pd-l1 expression in non–small-cell lung cancer. JAMA Oncol. 2016;2(1):46–54.
Gadiot J, Hooijkaas AI, Kaiser AD, et al. Overall survival and PD-L1 expression in metastasized malignant melanoma. Cancer. 2011;117(10):2192–201.
Sholl LM, Aisner DL, Allen TC, et al. Members of Pulmonary Pathology Group. Programmed death ligand-1 immunohistochemistry—a new challenge for pathologists: a perspective from members of the Pulmonary Pathology Society. Arch Pathol Lab Med. 2016;140(4):341–4.
Abdel-Rahman O. Correlation between PD-L1 expression and outcome of NSCLC patients treated with anti-PD-1/PD-L1 agents: a meta-analysis. Crit Rev Oncol Hematol. 2016;101:75–85.
Aguiar P Jr, Lopes G, Santoro I, et al. P2.47 (also presented as PD1.02): the role of PD-L1 expression as a predictive biomarker in advanced NSCLC: an update of a network meta-analysis. J Thorac Oncol. 2016;11(10):S247–8.
Zhang T, Xie J, Arai S, et al. The efficacy and safety of anti-PD-1/PD-L1 antibodies for treatment of advanced or refractory cancers: a meta-analysis. Oncotarget. 2016;7(45):73068–79.
Villaruz LC, Socinski MA. The clinical utility of PD-L1 testing in selecting non-small cell lung cancer patients for PD1/PD-L1-directed therapy. Clin Pharmacol Ther. 2016;100(3):212–4.
Hirsch FR, McElhinny A, Stanforth D, et al. PD-L1 immunohistochemistry assays for lung cancer: results from phase 1 of the Blueprint PD-L1 IHC Assay Comparison Project. J Thorac Oncol. 2017;12(2):208–22.
Ratcliffe MJ, Sharpe A, Midha A, et al. Agreement between programmed cell death ligand-1 diagnostic assays across multiple protein expression cut-offs in non-small cell lung cancer. Clin Cancer Res. 2017;23(14):3585–91.
NCCN. NCCN collaborates with Bristol-Myers Squibb to study PD-L1 expression and test interpretation in lung cancer. 2016. https://www.nccn.org/about/news/newsinfo.aspx?NewsID=559. Accessed 24 Oct 2017.
Scheel AH, Dietel M, Heukamp LC, et al. Harmonized PD-L1 immunohistochemistry for pulmonary squamous-cell and adenocarcinomas. Mod Pathol. 2016;29(10):1165–72.
Rimm DL, Han G, Taube JM, et al. A prospective, multi-institutional, pathologist-based assessment of 4 immunohistochemistry assays for pd-l1 expression in non–small cell lung cancer. JAMA Oncol. 2017;3(8):1051–8. https://doi.org/10.1001/jamaoncol.2017.0013.
Rizvi NA, Hellmann MD, Snyder A, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non–small cell lung cancer. Science. 2015;348(6230):124–8.
Peters S. Impact of tumor mutation burden on the efficacy of first-line nivolumab in stage IV or recurrent non-small cell lung cancer: an exploratory analysis of CheckMate-026 [abstract no. CT082]. In: AACR Annual Meeting 2017; 1–5 Apr 2017; Washington, DC.
Chaft JE, Forde PM, Smith KN, et al. Neoadjuvant nivolumab in early-stage, resectable non-small cell lung cancers. J Clin Oncol. 2017;35(15 suppl):8508.
Higgs BW, et al. 15LBA High tumoral IFN mRNA, PD-L1 protein, and combined IFN mRNA/PD-L1 protein expression associates with response to durvalumab (anti-PD-L1) monotherapy in NSCLC patients [abstract no. 15LBA]. Eur J Cancer. 2015;51:S717.
Shukuya T, Carbone DP. Predictive markers for the efficacy of anti PD-1/PD-L1 antibodies in lung cancer. J Thorac Oncol. 2016;11(7):976–88.
Gopalakrishnan V, Spencer C, Reuben A, et al. Association of diversity and composition of the gut microbiome with differential responses to PD-1 based therapy in patients with metastatic melanoma [abstract]. J Clin Oncol 2017; 35(suppl 7S) (abstract 2).
Gnjatic S, Bronte V, Brunet LR, et al. Identifying baseline immune-related biomarkers to predict clinical outcome of immunotherapy. J Immunother Cancer. 2017;5(1):44.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for the preparation of this manuscript.
Conflict of interest
Mark Socinski has received consulting fees or honorarium, and payment for lectures including service on speaker bureaus from Bristol-Meyers Squibb and Genentech. Katerina Ancevski Hunter and Liza Villaruz declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ancevski Hunter, K., Socinski, M.A. & Villaruz, L.C. PD-L1 Testing in Guiding Patient Selection for PD-1/PD-L1 Inhibitor Therapy in Lung Cancer. Mol Diagn Ther 22, 1–10 (2018). https://doi.org/10.1007/s40291-017-0308-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-017-0308-6