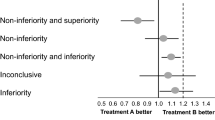
Abstract
Superiority trials are conducted to test the hypothesis that a treatment or strategy A is superior to (i.e., better than) treatment strategy B in reducing the impact of disease. However, A may be considerably safer, more convenient, or cheaper than B. These features may make A more attractive than B even if the burden of disease is reduced comparably by the two treatments, or even a bit worse by A over B. In this context, non-inferiority trials have become increasingly popular to test the hypothesis that a new treatment is not ‘unacceptably worse’ than an active comparator by more than a predefined non-inferiority margin. Non-inferiority trials have unique design features and methodology and require a different analysis than traditional superiority trials. The main aim of this overview is to analyze the role of non-inferiority trials in the development of new treatments, involving some scientific, statistical, and practical considerations. We elucidate some aspects of non-inferiority trials that contribute to the validity of the results. The unique design features and methodology of non-inferiority trials are summarized and key findings to consider when evaluating a non-inferiority trial are illustrated.



Similar content being viewed by others
References
Armitage P, Berry G. Statistical methods in medical research. 3rd ed. Oxford: Blackwell; 1994.
Head SJ, Kaul S, Bogers AJ, Kappetein AP. Non-inferiority study design: lessons to be learned from cardiovascular trials. Eur Heart J. 2012;33(11):1318–24. https://doi.org/10.1093/eurheartj/ehs099.
Bikdeli B, Welsh JW, Akram Y, Punnanithinont N, Lee I, Desai NR, et al. Non-inferiority designed cardiovascular trials in highest-impact journals. Circulation. 2019;140(5):379–89. https://doi.org/10.1161/circulationaha.119.040214.
Pocock SJ. The pros and cons of noninferiority trials. Fundam Clin Pharmacol. 2003;17(4):483–90.
Wangge G, Klungel OH, Roes KC, de Boer A, Hoes AW, Knol MJ. Interpretation and inference in noninferiority randomized controlled trials in drug research. Clin Pharmacol Ther. 2010;88(3):420–3. https://doi.org/10.1038/clpt.2010.134.
Scott IA. Non-inferiority trials: determining whether alternative treatments are good enough. Med J Aust. 2009;190(6):326–30.
Kaji AH, Lewis RJ. Noninferiority trials: is a new treatment almost as effective as another? JAMA. 2015;313(23):2371–2. https://doi.org/10.1001/jama.2015.6645.
Piaggio G, Elbourne DR, Pocock SJ, Evans SJ, Altman DG, Group C. Reporting of noninferiority and equivalence randomized trials: extension of the CONSORT 2010 statement. JAMA. 2012;308(24):2594–604. https://doi.org/10.1001/jama.2012.87802.
Center for Biologics Evaluation and Research (CBER), Center for Drug Evaluation and Research (CDER), Food and Drug Administration, U.S. Department of Health and Human Services. Guidance for industry non-inferiority clinical trials. http://www.fdagov/downloads/Drugs/Guidances. Accessed 01 July 2019.
European Medicines Agency. Pre-authorisation evaluation of medicines for human use. Guideline on the choice of the non-inferiority margin. 2005. https://www.ema.europaeu/en/documents/scientific-guideline/guideline-choice-non-inferiority-margin_enpdf. Accessed 31 Jan 2020.
Fleiss JL. General design issues in efficacy, equivalency and superiority trials. J Periodontal Res. 1992;27(4 Pt 2):306–13 (discussion 23–7).
Pocock SJ, Clayton TC, Stone GW. Design of major randomized trials: part 3 of a 4-part series on statistics for clinical trials. J Am Coll Cardiol. 2015;66(24):2757–66. https://doi.org/10.1016/j.jacc.2015.10.036.
van Ravenzwaaij D, Monden R, Tendeiro JN, Ioannidis JPA. Bayes factors for superiority, non-inferiority, and equivalence designs. BMC Med Res Methodol. 2019;19(1):71. https://doi.org/10.1186/s12874-019-0699-7.
Koopmeiners JS, Hobbs BP. Detecting and accounting for violations of the constancy assumption in non-inferiority clinical trials. Stat Methods Med Res. 2018;27(5):1547–58. https://doi.org/10.1177/0962280216665418.
Hart RG, Benavente O, McBride R, Pearce LA. Antithrombotic therapy to prevent stroke in patients with atrial fibrillation: a meta-analysis. Ann Intern Med. 1999;131(7):492–501. https://doi.org/10.7326/0003-4819-131-7-199910050-00003.
Berlin JA, Laird NM, Sacks HS, Chalmers TC. A comparison of statistical methods for combining event rates from clinical trials. Stat Med. 1989;8(2):141–51.
Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361(12):1139–51. https://doi.org/10.1056/NEJMoa0905561.
Blech S, Ebner T, Ludwig-Schwellinger E, Stangier J, Roth W. The metabolism and disposition of the oral direct thrombin inhibitor, dabigatran, in humans. Drug Metab Dispos. 2008;36(2):386–99. https://doi.org/10.1124/dmd.107.019083.
Stangier J, Rathgen K, Stahle H, Gansser D, Roth W. The pharmacokinetics, pharmacodynamics and tolerability of dabigatran etexilate, a new oral direct thrombin inhibitor, in healthy male subjects. Br J Clin Pharmacol. 2007;64(3):292–303. https://doi.org/10.1111/j.1365-2125.2007.02899.x.
Verdecchia P, Angeli F, Aita A, Bartolini C, Reboldi G. Why switch from warfarin to NOACs? Intern Emerg Med. 2016;11(3):289–93. https://doi.org/10.1007/s11739-016-1411-0.
Holmgren EB. Establishing equivalence by showing that a specified percentage of the effect of the active control over placebo is maintained. J Biopharm Stat. 1999;9(4):651–9.
Rothmann M, Li N, Chen G, Chi GY, Temple R, Tsou HH. Design and analysis of non-inferiority mortality trials in oncology. Stat Med. 2003;22(2):239–64. https://doi.org/10.1002/sim.1400.
Angeli F, Verdecchia P, Reboldi G. Non-inferiority trial design in drug development: a primer for cardiovascular healthcare professionals. Am J Cardiovasc Drugs. 2019. https://doi.org/10.1007/s40256-019-00378-w.
Wangge G, Klungel OH, Roes KC, de Boer A, Hoes AW, Knol MJ. Room for improvement in conducting and reporting non-inferiority randomized controlled trials on drugs: a systematic review. PLoS One. 2010;5(10):e13550. https://doi.org/10.1371/journal.pone.0013550.
Schumi J, Wittes JT. Through the looking glass: understanding non-inferiority. Trials. 2011;12:106. https://doi.org/10.1186/1745-6215-12-106.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No external funding was used in the preparation of this article.
Conflict of interest
Fabio Angeli Paolo Verdecchia, Gaetano Vaudo, Sergio Masnaghetti, and Gianpaolo Reboldi have no conflicts of interest that are directly relevant to the content of this article.
Rights and permissions
About this article
Cite this article
Angeli, F., Verdecchia, P., Vaudo, G. et al. Optimal Use of the Non-Inferiority Trial Design. Pharm Med 34, 159–165 (2020). https://doi.org/10.1007/s40290-020-00334-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40290-020-00334-z