Abstract
Introduction
The role of macrolides for treatment of children with acute asthma or wheezing exacerbations is unclear.
Objective
The aim of this systematic review was to evaluate the effectiveness of macrolides in children with recurrent wheezing presenting with acute asthma or wheezing exacerbation.
Methods
We conducted an electronic search in MEDLINE, EMBASE, CINAHL, LILACS, CENTRAL, and ClinicalTrials.gov.
Study selection criteria
Randomized controlled trials of macrolides (any macrolide) compared with placebo or standard treatment in children up to 18 years with recurrent wheezing/asthma presenting with an acute exacerbation.
Outcomes
Primary outcomes were need for hospitalization and/or time of acute asthma/wheezing symptoms resolution; secondary outcomes were duration of stay in the emergency department (ED)/clinic, severity of symptoms of the index episode, use of additional systemic corticosteroids or short active β-2 agonists, changes in lung function measures, ED visit/hospitalization during first week after index episode, time to next exacerbation, or adverse effects (AEs).
Results
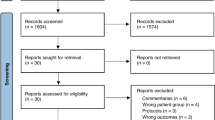
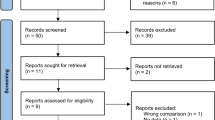
Only three studies met the inclusion criteria (n = 334 children, 410 treated episodes); two studies included recurrent wheezers and the third included asthmatic children. There was no difference in hospitalization between groups, but children treated with macrolides had a significantly lower time to symptoms resolution than controls, although the magnitude of benefit remains to be quantified due to no normal distribution data presented. There was no difference in time to next episode of exacerbation (HR 0.96; 95% CI 0.71–1.28; I2 = 0%; p = 0.77). In one study, children receiving macrolides had a significant decrease in the severity of symptoms, decrease use of salbutamol, and another study showed improved lung function. No study evaluated antibiotic resistance development.
Conclusions
Limited evidence support that a macrolide trial could be considered in children with acute asthma or recurrent wheezing exacerbation.



Similar content being viewed by others
Change history
13 March 2020
An Online First version of this article was made available online at https://link.springer.com/article/10.1007/s40272-019-00371-5 on 14 January 2020. An error was subsequently identified in the article, and the following correction should be noted:
References
Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention, 2018. http://www.ginasthma.org. Accessed Nov 2019.
Papi A, Brightling C, Pedersen SE, Reddel HK. Asthma. Lancet. 2018;391(17):783–800.
Nunes C, Pereira AM, Morais-Almeida M. Asthma costs and social impact. Asthma Res Pract. 2017;3(1):1.
Nurmagambetov T, Kuwahara R, Garbe P. The economic burden of asthma in the United States, 2008–2013. Ann Am Thorac Soc. 2018;15(3):348–56.
Garner R, Kohen D. Changes in the prevalence of asthma among Canadian children. Health Rep. 2008;19(2):45–50.
Lougheed MD, Garvey N, Chapman KR, Cicutto L, Dales R, Day AG, et al. The Ontario asthma regional variation study: emergency department visit rates and the relation to hospitalization rates. Chest. 2006;129(4):909–17.
Karaca-Mandic P, Jena AB, Joyce GF, Goldman DP. Out-of-pocket medication costs and use of medications and health care services among children with asthma. JAMA. 2012;307(12):1284–91.
Radhakrishnan DK, Dell SD, Guttmann A, Shariff SZ, Liu K, To T. Trends in the age of diagnosis of childhood asthma. J Allergy Clin Immunol. 2014;134(5):1057–62.
Szefler SJ, Zeiger RS, Haselkorn T, Mink DR, Kamath TV, Fish JE, et al. Economic burden of impairment in children with severe or difficult-to-treat asthma. Ann Allergy Asthma Immunol. 2011;107(2):110–9.
Castro-Rodriguez JA, Rodrigo GJ. Efficacy of inhaled corticosteroids in infants and preschoolers with recurrent wheezing and asthma: a systematic review with meta-analysis. Pediatrics. 2009;123(3):e519–25.
Kaiser SV, Huynh T, Bacharier LB, Rosenthal JL, Bakel LA, Parkin PC, et al. Preventing exacerbations in preschoolers with recurrent wheeze: a meta-analysis. Pediatrics. 2016;137(6):e20154496.
Castro-Rodriguez JA, Beckhaus AA, Forno E. Efficacy of oral corticosteroids in the treatment of acute wheezing episodes in asthmatic preschoolers: systematic review with meta-analysis. Pediatr Pulmonol. 2016;51(8):868–76.
Beigelman A, Chipps BE, Bacharier LB. Update on the utility of corticosteroids in acute pediatric respiratory disorders. Allergy Asthma Proc. 2015;36(5):332–8.
da Silva Filho LV, Pinto LA, Stein RT. Uso de macrolídeos em doenças pulmonares: controvérsias da literatura recente. J Pediatr (Rio J). 2015;91(6 Suppl 1):S52–60.
Hernando-Sastre V. Macrolide antibiotics in the treatment of asthma. An update. Allergol Immunopathol (Madr). 2010;38(2):92–8.
Poole NM, Shapiro DJ, Fleming-Dutra KE, Hicks LA, Hersh AL, Kronman MP. Antibiotic prescribing for children in United States emergency departments: 2009–2014. Pediatrics. 2019;143(2):e20181056.
Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;350:g7647.
Clinical safety data management: definitions and standards for expedited reporting E2A. ICH Harmonised Tripartite Guideline. 1994. https://database.ich.org/sites/default/files/E2A_Guideline.pdf. Accessed Nov 2019.
Higgins JPT, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928.
Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Deeks JJ, Altman DG, Bradburn M. Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Davey G, Altman D, editors. Systematic reviews in health care: meta-analysis in context. 2nd ed. London: BMJ Publishing Group; 2001. p. 285–312.
Borenstein M, Hedges LV, Higgins JPT, Rothstein HR. Introduction to meta-analysis. Chichester: Wiley; 2009.
Volovitz B, Bilavsky E, Nussinovitch M. Effectiveness of high repeated doses of inhaled budesonide or fluticasone in controlling acute asthma exacerbations in young children. J Asthma. 2008;45(7):561–7.
Fonseca-Aten M, Okada PJ, Bowlware KL, Chavez-Bueno S, Mejias A, Rios AM, et al. Effect of clarithromycin on cytokines and chemokines in children with an acute exacerbation of recurrent wheezing: a double-blind, randomized, placebo-controlled trial. Ann Allergy Asthma Immunol. 2006;97(4):457–63.
Bacharier LB, Guilbert TW, Mauger DT, Boehmer S, Beigelman A, Fitzpatrick AM, et al. Early administration of azithromycin and prevention of severe lower respiratory tract illnesses in preschool children with a history of such illnesses a randomized clinical trial. JAMA. 2015;314(19):2034–44.
Koutsoubari I, Papaevangelou V, Konstantinou GN, Makrinioti H, Xepapadaki P, Kafetzis D, et al. Effect of clarithromycin on acute asthma exacerbations in children: an open randomized study. Pediatr Allergy Immunol. 2012;23(4):385–90.
Stokholm J, Chawes BL, Vissing NH, Bjarnadóttir E, Pedersen TM, Vinding RK, et al. Azithromycin for episodes with asthma-like symptoms in young children aged 1-3 years: a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2016;4(1):19–26.
Mandhane PJ, Paredes Zambrano de Silbernagel P, Aung YN, Williamson J, Lee BE, Spier S, et al. Treatment of preschool children presenting to the emergency department with wheeze with azithromycin: a placebo-controlled randomized trial. PLoS One. 2017;12(8):e0182411.
Stokholm J, Chawes BL, Vissing NH, Bjarnadóttir E, Pedersen TM, Vinding RK, et al. Supplement to: azithromycin for episodes with asthma-like symptoms in young children aged 1–3 years: a randomised, double-blind, placebo-controlled trial. Lancet Respir Med. 2016;4(1):19–26.
Normansell R, Sayer B, Waterson S, Dennett EJ, Del Forno M, Dunleavy A. Antibiotics for exacerbations of asthma. Cochrane Database Syst Rev. 2018. https://doi.org/10.1002/14651858.CD002741.pub2.
Lei W, Tsai M, Liu S, Yeh T. The effects of macrolides in children with reactive airway disease: a systematic review and meta- analysis of randomized controlled trials. Drug Des Dev Ther. 2018;12:3825–45.
Lin C, Yeh T, Liu S, Lin H, Cheng Y, Hung H, et al. Effects of macrolide treatment during the hospitalization of children with childhood wheezing disease : a systematic review and meta-analysis. J Clin Med. 2018;7(11):432.
Steel HC, Theron AJ, Cockeran R, Anderson R, Feldman C. Pathogen- and host-directed anti-inflammatory activities of macrolide antibiotics. Mediat Inflamm. 2012;2012:17.
Altenburg J, de Graaff CS, van der Werf TS, Boersma WG. Immunomodulatory effects of macrolide antibiotics—part 1: biological mechanisms. Respiration. 2011;81(1):67–74.
Beigelman A, Gunsten S, Mikols CL, Vidavsky I, Cannon CL, Brody SL, et al. Azithromycin attenuates airway inflammation in a noninfectious mouse model of allergic asthma. Chest. 2009;136(2):498–506.
Zimmermann P, Ziesenitz VC, Curtis N, Ritz N. The immunomodulatory effects of macrolides-A systematic review of the underlying mechanisms. Front Immunol. 2018;9:302.
Beigelman A, Isaacson-schmid M, Sajol G, Baty J, Rodriguez OM, Leege E, et al. Randomized trial to evaluate azithromycin ’ s effects on serum and upper airway IL-8 levels and recurrent wheezing in infants with respiratory syncytial virus bronchiolitis. J Allergy Clin Immunol. 2015;135(5):1171–8.
Wong EHC, Porter JD, Edwards MR, Johnston SL. The role of macrolides in asthma: current evidence and future directions. Lancet Respir. 2014;2600(14):1–14.
Kanoh S, Rubin BK. Mechanisms of action and clinical application of macrolides as immunomodulatory medications. Clin Microbiol Rev. 2010;23(3):590–615.
Asada M, Yoshida M, Suzuki T, Hatachi Y, Sasaki T, Yasuda H, et al. Macrolide antibiotics inhibit respiratory syncytial virus infection in human airway epithelial cells. Antiviral Res. 2009;83(2):191–200.
Min JY, Jang YJ. Macrolide therapy in respiratory viral infections. Mediat Inflamm. 2012;2012:9.
Gielen V, Johnston SL, Edwards MR. Azithromycin induces anti-viral responses in bronchial epithelial cells. Eur Respir J. 2010;36(3):646–54.
Papadopoulos NG, Christodoulou I, Rohde G, Agache I, Almqvist C, Bruno A, et al. Viruses and bacteria in acute asthma exacerbations—A GA2LEN-DARE systematic review. Allergy. 2011;66(4):458–68.
Hahn DL, Webley W. Chronic Chlamydia pneumoniae lung infection: a neglected explanation for macrolide effects in wheezing and asthma? Lancet Respir. 2016;4(3):e8.
Rollins DR, Beuther DA, Martin RJ. Update on infection and antibiotics in asthma. Curr Allergy Asthma Rep. 2010;10(1):67–73.
Cohen RT, Pelton SI. Individual benefit vs societal effect of antibiotic prescribing for preschool children with recurrent wheeze. JAMA. 2015;314(19):2027–9.
Fleming-Dutra KE, Friedman CR, Hicks LA. Early azithromycin treatment to prevent severe lower respiratory tract illnesses in children. JAMA. 2016;315(19):2121–2.
Hansen MP, Scott AM, McCullough A, Thorning S, Aronson JK, Beller EM, et al. Adverse events in people taking macrolide antibiotics versus placebo for any indication. Cochrane Database Syst Rev. 2019. https://doi.org/10.1002/14651858.CD011825.pub2.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Dr. Castro-Rodriguez’s contribution was funded in part by CONICYT PIA/ANILLO (Grant no. 170925013) from the Chilean Comisión Nacional de Investigación Científica y Tecnológica (CONICYT).
Financial disclosure
The authors have no financial relationships relevant to this article to disclose.
Conflict of interest
The authors declare that they have no conflict of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pincheira, M.A., Bacharier, L.B. & Castro-Rodriguez, J.A. Efficacy of Macrolides on Acute Asthma or Wheezing Exacerbations in Children with Recurrent Wheezing: A Systematic Review and Meta-analysis. Pediatr Drugs 22, 217–228 (2020). https://doi.org/10.1007/s40272-019-00371-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40272-019-00371-5