Abstract
The complexity of neural circuits that control food intake and energy balance in the hypothalamic nuclei explains some of the constraints involved in the prevention and treatment of obesity. Two major neuronal populations present in the arcuate nucleus control caloric intake and energy expenditure: one population co-expresses orexigenic agouti-related peptide (AgRP) and neuropeptide Y and the other expresses the anorexigenic anorectic neuropeptides proopiomelanocortin and cocaine- and amphetamine-regulated transcript (POMC/CART). In addition to integrating signals from neurotransmitters and hormones, the hypothalamic systems that regulate energy homeostasis are affected by nutrients. Fat-rich diets, for instance, elicit hypothalamic inflammation (reactive activation and proliferation of microglia, a condition named gliosis). This process generates resistance to the anorexigenic hormones leptin and insulin, contributing to the genesis of obesity. Glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs) have increasingly been used to treat type 2 diabetes mellitus. One compound (liraglutide) was recently approved for the treatment of obesity. Although most studies suggest that GLP-1RAs promote weight loss mainly due to their inhibitory effect on food intake, other central effects that have been described for native GLP-1 and some GLP-1RAs in rodents and humans encourage future clinical trials to explore additional mechanisms that potentially underlie the beneficial effects observed with this drug class. In this article we review the most relevant data exploring the mechanisms involved in the effects of GLP-1RAs in the brain–adipocyte axis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
In addition to its well known action in glucose homeostasis GLP-1R can also modulate other important functions in the body, including cardiovascular, imune and nervous, and the control of caloric intake and energy expenditure. |
Experimental studies show that GLP-1RA promotes increased activity of brown adipose tissue through the activation of hypothalamic neurons. |
GLP-1RA are amongst the most promising agents that can act in the recruitment of brown adipose tissue in humans. |
Subcutaneously administered GLP-1RA have established efficacy in the treatment of obesity in adult patients. |
1 Introduction
Intestinal hormones have become an important therapeutic target in the management of obesity due to their involvement in energy homeostasis and satiety [1]. Among the intestinal L cell peptides, glucagon-like peptide-1 (GLP-1) has been highlighted because of the success of its recent clinical use in the treatment of diabetes mellitus and obesity [2]. GLP-1 receptor (GLP-1R) signaling contributes to increased glucose-dependent insulin secretion, β cell proliferation, islet size, portal glucose sensing, and postprandial lipid metabolism [3]. Moreover, it regulates cardiovascular function, glucose concentration, gut motility, immune function, neuronal physiology and repair, appetite, and energy expenditure, and therefore impacts on body mass control [3].
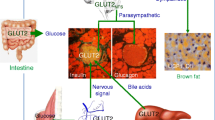
In addition to its extensively studied actions in peripheral tissues, studies have evaluated the distribution of GLP-1R in the central nervous system (CNS) of rodents, non-human primates, and humans, showing that it is widely diffused to multiple CNS neurons including neurons of the arcuate nucleus (ARC), which are crucial for the control of energy balance (Fig. 1) [4, 5]. Accordingly, one of the most remarkable central effects of GLP-1R signaling occurs in neuronal populations involved in the control of caloric intake by promoting anorexigenic effects. Furthermore, the activation of GLP-1R signaling in the ventromedial hypothalamus (VMH) can also control energy expenditure by promoting food intake-independent weight loss by inducing brown adipose tissue (BAT) thermogenesis and browning through a sympathetic drive to BAT [6]. BAT is a thermogenic mammalian organ composed of multilocular adipocytes specialized in generating heat instead of accumulating energy [7]. Besides the action of the GLP-1R agonist (GLP-1RA) on the brain–adipocyte axis, there is also recent evidence that its direct action (independent of its CNS actions) in the white adipose tissue induces browning and enhances the lipolytic capacity and mitochondrial biogenesis [8].
Glucagon-like peptide-1 (GLP-1) action in the central nervous system. The illustration of the whole brain depicts the main regions containing binding sites (GLP-1 receptors [GLP-1R], shown in blue) for GLP-1 (shown in green): hypothalamus (Hyp), ventral tegmental area (VTA), dorsal raphe nucleus (DR), brainstem (BS), nucleus of the solitary tract (NTS), and area postrema (AP). In the Hyp (upper box), GLP-1R has been detected in the paraventricular nucleus (PVN), lateral hypothalamic area (LHA), dorsomedial hypothalamus (DMH), ventromedial hypothalamus (VMH), and arcuate nucleus (ARC). Acting in the Hyp, GLP-1 can increase oxytocin and reduce hypothalamic gliosis (details in the right-hand side of the figure). In the ARC (box in the middle of the figure), GLP-1 reduces food intake by acting directly in proopiomelanocortin (POMC)/cocaine- and amphetamine-regulated transcript (CART) neurons and indirectly in neuropeptide Y (NPT)/agouti-related peptide (AgRP) neurons; the action in NPY/AgRP neurons is believed to occur through a hitherto unidentified γ-aminobutyric acid (GABA)-ergic neuron. In addition, acting in the Hyp, GLP-1 can increase energy expenditure by stimulating brown adipose tissue (BAT) activity and promoting browning of white adipose tissue (WAT) (details in the bottom right-hand side of the figure). The hypothalamic actions of GLP-1 increasing oxytocin and reducing gliosis can also contribute to reduction of food intake and increasing energy expenditure. In the VTA, GLP-1 can reduce dopamine, which contributes for reduction of consumption of highly palatable foods (details in the right-hand side of the figure). ↑ indicates increase, ↓ indicates decrease
Despite the rapidly increasing understanding of the central regulation of whole-body energy homeostasis, translating this knowledge into more efficient therapies for obesity has proved challenging. Most effort has been directed towards development of anorexigenic drugs [9]; however, compounds that exert additional effects, such as regulation of energy expenditure, are expected to act with larger efficiency. Thus, following the identification of active BAT in adult humans and the demonstration that certain depots of white adipose tissue can undergo browning, there has been recent interest in the development of approaches that induce BAT activity and promote browning [10]. Such strategies have undergone a rapid development due to translational research, with the emergence of new pharmacological agents. Among the new agents, GLP-1RAs are the most promising.
In this review, we focus on the actions of GLP-1RAs in the CNS, with emphasis on the contribution of GLP-1 to reset energy balance by promoting BAT recruitment.
2 Structure and Physiology of Glucagon-Like Peptide-1 (GLP-1)
The incretin hormone GLP-1 is derived from the processing of proglucagon that occurs in ileal L cells and the nucleus tractus solitarius (NTS) [11]. During meals, GLP-1 is secreted in two stages: a first peak occurs approximately 15 min after the beginning of the meal, when food in the stomach and in the initial portions of the intestine stimulates the release of hormones, such as glucose-dependent insulinotropic polypeptide (GIP), which acts by vagal pathways to stimulate L cells [12]; a second peak occurs after direct stimulation of L cells by nutrients [13].
The active forms of GLP-1 have a half-life of less than 2 min. Immediately after being secreted, GLP-1 enters the capillaries and is rapidly degraded by dipeptidyl peptidase-4 (DPP-4). Despite the loss of its insulinotropic effect, the GLP-1 products of DPP-4 catalysis exert other actions, such as suppression of hepatic glucose production and antioxidant activity in the cardiovascular system [14]. The effects of the active forms of GLP-1 are mediated by a G protein-coupled receptor, GLP-1R, which is expressed in several sites, including enteric and vagal nerves, the stomach, pancreas, intestine, and various brain regions [15].
Because of its very short half-life, native GLP-1 cannot be used for therapeutic purposes. The GLP-1RAs are resistant to DPP-4 degradation, have longer half-lives, and have been developed for the treatment of patients with type 2 diabetes mellitus (T2DM) [15]. More recently, they have been used for the treatment of obesity [16]. Among the GLP-1RAs, exenatide is the synthetic version of exendin-4, a molecule identified in the Gila monster salivary gland whose amino acid sequence shares 53% identity with human GLP-1; its relatively short half-life requires twice-daily administration [17]. Lixisenatide is also a synthetic version of exendin-4 that has modifications consisting of the deletion of one proline residue and the addition of six lysine residues at the C-terminal end, which increases its half-life and its binding affinity to GLP-1R. Although lixisenatide is administered once a day, it is still considered a short-acting GLP-1RA [18].
Some of the molecules that are considered to be long-acting GLP-1RAs are briefly described here and in Table 1. Long-acting exenatide is formulated in microspheres of poly-(d,l-lactide-co-glycolide) for once-weekly administration [19]. Liraglutide is another long-acting GLP-1RA that is administered once daily. It is identical to the native GLP-1, except for the replacement of lysine with arginine at position 34 and the attachment of a palmitic acid to lysine at position 26. These modifications result in increased self-association, binding to albumin, and a longer half-life [20]. Dulaglutide is a once-weekly GLP-1RA consisting of the fusion of two identical sequences (the N-terminal portion of the native GLP-1) covalently bound by a peptide linker to the Fc component of a modified human immunoglobulin G4 heavy chain [21]. Albiglutide is also a once-weekly GLP-1RA consisting of two copies of a 30 amino acid sequence of modified human GLP-1 (replacement of alanine with glycine at position 8) fused with human albumin [22]. The most relevant metabolic effects expected from the GLP-1RA are summarized in Table 2.
3 GLP-1 Receptor Agonists (GLP-1RAs) in the Central Nervous System and the Energy Balance in Rodents
GLP-1 is synthesized in NTS neurons that project to GLP-1R-expressing regions, such as the paraventricular nucleus (PVN) and ARC [23]; in the latter, proopiomelanocortin (POMC) neurons are present and it is believed that endogenous GLP-1 induces satiety, affecting both anorexigenic and orexigenic signaling pathways. Activation of GLP-1R in the PVN stimulates the release of oxytocin, which exerts anorexigenic effects [13]. There are also NTS neurons projecting to the ventral tegmental area, which is a reward center. Endogenous GLP-1 acts in GLP-1Rs located in this region and reduces the intake of highly palatable foods by suppressing mesolimbic dopamine signaling, which controls the pleasure-directed acquisition of food [24].
The GLP-1 derived from intestinal L cells may also communicate with the CNS through GLP-1Rs located in fibers innervating the portal vein or in the vagus nerve. However, the contributions of the peripheral nervous system and the CNS in the mediation of the anorexic effects of the GLP-1RAs is still not completely understood [23]. Experimental studies suggest that liraglutide exerts its anorexigenic but not hypoglycemic effects through CNS receptors, rather than through vagus nerve receptors [25]. Secher et al. [26] showed that liraglutide-dependent weight loss relies on its binding to GLP-1Rs located mainly in POMC/CART (cocaine- and amphetamine-regulated transcript) anorexigenic neurons in the ARC, but an indirect inhibitory effect of agouti-related peptide (AgRP) orexigenic neurons via γ-aminobutyric acid (GABA)-ergic signaling was also observed.
In addition to the studies demonstrating the central effects of GLP-1 and its analogs on satiety and reduction of energy intake, there are studies showing other central effects of GLP-1 involving BAT. BAT regulates energy expenditure through a process known as adaptive thermogenesis, in which uncoupling protein-1 (UCP1, a BAT marker) uncouples mitochondria respiration to generate heat instead of adenosine triphosphate [7].
Intracerebroventricular (ICV) infusion of native GLP-1 promotes not only a reduction in food intake and body weight, but also an increase in BAT thermogenesis induced by increased sympathetic nervous system (SNS) activity [6]. In addition, the ICV infusion of exendin-4 increases CNS activity, promotes BAT activation and white adipose tissue browning, and increases BAT glucose and triglyceride uptake [27]. Peripheral exendin-4 administration also increases energy expenditure and BAT thermogenesis [28]. Similarly, the ICV infusion of liraglutide stimulates BAT thermogenesis and white adipose tissue browning. These effects depend on the reduction of 5′ adenosine monophosphate-activated protein kinase (AMPK) activity in the VMN [29]. Together, these results suggest that GLP-1R signaling contributes to BAT thermogenic capacity. However, the increase in BAT recruitment apparently does not induce weight loss during long-term subcutaneous treatment with liraglutide in mice (Fig. 1) [30].
In addition to the expression in areas related to satiety, GLP-1R is present in several other areas in the CNS. Glial cells only express GLP-1R when activated, in response to inflammation [31]. Farr et al. [4] evaluated the distribution of GLP-1R in human brains and demonstrated its presence in all neurons of the parietal cortex, in the ARC, PVN, VMN, area postrema, dorsal motor nucleus of the vagus in the medulla oblongata, and in the NTS. They also confirmed the lack of expression of GLP-1R in glial cells [4].
The identification of GLP-1R in other regions of the CNS provided a further advance in the understanding of the role played by GLP-1/serotonin cross-talk in the regulation of homeostatic control of body mass. Anderberg et al. [32] demonstrated that long-term stimulation with GLP-1RAs promoted an increase in the expression of serotonergic receptors in the hypothalamus. In addition, they showed that the 5-hydroxytryptamine 2A (5-HT2A) receptor is crucial for GLP-1RA-induced weight loss after exendin-4 ICV injection, such as peripheral injection of liraglutide in rats. The authors identified that the dorsal raphe nucleus (DR) has serotonergic neurons that secrete serotonin to hypothalamic nuclei and that GLP-1R activation promotes the activity of DR serotonin neurons. Thus, they provided evidence that serotonin is crucial for controlling feeding and weight loss induced by GLP-1R activation [32].
The first studies evaluating the effects of GLP-1 and exendin-4 in neuronal cells demonstrated their ability to promote neurite outgrowth (a similar effect to that observed with the neurotrophic nerve growth factor) and to protect cultured neurons against glutamate-induced apoptosis [33]. Such reports inspired further studies to evaluate the effects of GLP-1RAs in animal models of degenerative neurological diseases.
Parkinson disease is characterized by the degeneration of nigrostriatal dopamine-producing neurons. Intraperitoneal exendin-4 was shown to interrupt and even revert the progression of drug-induced nigrostriatal lesions [34]. In a rodent model of stroke, exendin-4 interrupted microglia infiltration and increased the stem cell proliferation elicited by middle cerebral artery transient occlusion [35].
The ability of exendin-4 and liraglutide administered subcutaneously to promote proliferation of progenitor cells located in the dentate gyrus in the hippocampus has been tested. Both GLP-1RAs further increased progenitor cells proliferation, suggesting that they may be promising drugs for the treatment of neurodegenerative diseases [36]. Lixisenatide administered intraperitoneally has also been shown to increase neurogenesis in the dentate gyrus [37].
Liraglutide was evaluated in a mouse model of Alzheimer disease in an intermediate stage of disease progression; it improved learning and memory and decreased amyloid deposition and chronic inflammation [38]. In another study conducted in mice with Alzheimer disease in the final stage, intraperitoneal liraglutide improved spatial memory, reduced amyloid deposition and inflammation, and increased dentate gyrus neurogenesis, suggesting that, besides exerting preventive effects, this GLP-1RA may reverse some of the key pathological findings of Alzheimer disease. Currently, liraglutide is being evaluated in humans with mild cognitive impairment [39].
A recently published study explored the neuroprotective effects of subcutaneous liraglutide in the ARC neuronal damage (gliosis and upregulation of the pro-apoptotic gene Bax) induced by a high-fat diet (HFD). In addition to promoting weight loss, activating POMC anorexigenic neurons, and increasing leptin sensitivity, liraglutide reduced gliosis and increased expression of the anti-apoptotic gene Bcl2. These effects could be attributed to treatment with liraglutide and not to weight loss, since they were not observed in the group of animals that lost weight without receiving the GLP-1RA [40]. Similar results of gliosis reduction have been shown after subcutaneous administration of exendin-4 to mice on HFDs [41]. These results demonstrate the potential of GLP-1RAs not only to promote weight loss and metabolic improvement but also to act directly on the hypothalamic inflammation.
4 Clinical Effects in Humans
4.1 Peripheral Effects
In 1998, Flint et al. [42] demonstrated in healthy volunteers that infusion of native GLP-1 during breakfast increased satiety and fullness and reduced the caloric intake in the next meal by 12%. The clinical trials conducted with GLP-1RAs in T2DM patients have demonstrated these beneficial effects. A meta-analysis of 21 clinical studies showed a weighted mean difference in body weight of –2.9 kg (95% confidence interval [CI] –3.6 to –2.2) with the maximum dose of each one of the GLP-1RAs [43]. These positive effects on weight loss together with the experimental evidence suggesting that liraglutide exerts its effects directly in the CNS raise the question of whether GLP-1RAs of higher molecular weights, such as dulaglutide and albiglutide, would be as effective as liraglutide in promoting weight loss [23].
A phase III, randomized, double-blind, placebo-controlled trial, which was open-label for the comparator liraglutide group, evaluated dulaglutide monotherapy (0.75 mg) versus placebo versus once-daily liraglutide in individuals with T2DM. Despite the positive results regarding glycemic control, in which dulaglutide was superior to placebo and non-inferior to liraglutide, there was no significant weight loss in any group after 26 weeks. This could be explained by the leanness of the Japanese population at the baseline, by the anabolic effect of improved β cell function, or even because of the low dose employed in this trial [44].
Another phase III, randomized, open-label, non-inferiority, head-to-head trial, AWARD-6, evaluated dulaglutide (1.5 mg) versus liraglutide (1.8 mg) in patients with T2DM. The once-weekly dose of dulaglutide was non-inferior to once-daily liraglutide in relation to glycemic control. There was significant weight loss in both groups; however, the magnitude of the body mass reduction was significantly greater in the liraglutide group [45]. Similar results were seen in the non-inferiority HARMONY 7 trial, which compared liraglutide versus albiglutide [46]. Thus, two head-to-head trials demonstrated that liraglutide was superior for weight loss. However, the explanation for this is not clear. One possibility is that the difference in efficiency in weight loss is due to a higher uptake of liraglutide in the CNS. Another possibility is the existence of differences in the effect of high molecular weight versus low molecular weight peptides on GLP-1R signaling in the CNS [23]. Experimental evidence indicated that while peptides bound to albumin do not penetrate the CNS, peripheral activation of the GLP-1R system would be coupled to neuronal activation and central effects of gastric emptying and decreased food intake, independent of direct exposure on the CNS [47].
The effects of GLP-1 and its analogs on energy expenditure are still inconsistent [48]. An evaluation of T2DM patients using liraglutide for 4 weeks detected a trend towards an increased basal energy expenditure [49], but this effect was not confirmed in subsequent longer studies [50, 51].
A randomized, double-blind, placebo-controlled, crossover study investigated the effects of liraglutide 1.8 and 3.0 mg for 5 weeks in obese non-diabetic patients. One-hour gastric emptying was 13 and 23% slower than placebo, respectively. Liraglutide doses similarly increased postprandial satiety and fullness and reduced hunger and prospective food consumption [52]. Additional studies are needed to define the participation of increased energy expenditure on the weight loss induced by GLP-1RAs.
4.2 Brain Effects
Neuroimaging studies have been employed in attempts to explore the central mechanisms of action of GLP-1 and its analogs. Increasing GLP-1 in the postprandial period correlates with the increase in blood flow in brain areas related to satiety, such as the hypothalamus and areas of the prefrontal cortex [53]. De Silva et al. [54] employed functional magnetic resonance imaging (fMRI) and showed that GLP-1 infusion in fasting healthy volunteers of normal weight attenuated neuronal activity in areas related to reward processing and hedonic feeding to the same extent as a meal (Table 3).
fMRI was also used to evaluate the central effects of GLP-1RAs. van Bloemendaal et al. [55] demonstrated that obese subjects presented increased activation in appetite and reward-related brain areas in response to food images. This was attenuated by an intravenous injection of exenatide [55]. Farr et al. [4] treated 21 T2DM patients with increasing doses of liraglutide (up to 1.8 mg) for 17 days. In comparison to placebo, liraglutide reduced activation of the parietal cortex and of insula and putamen (areas involved in the reward system) in response to pictures of highly palatable foods [4].
The observation that liraglutide promotes relevant weight loss led to the approval of the daily dose of 3.0 mg for the treatment of obesity [16]. The efficacy of liraglutide 3.0 mg was evaluated in T2DM patients with a body mass index (BMI) ≥27 kg/m2 treated with diet and exercise alone or in combination with antidiabetic drugs. This was a randomized, double-blind study with three arms of treatment: placebo and liraglutide 1.8 and 3.0 mg. The primary endpoints were relative change in weight and the proportion of participants losing ≥5 or ≥10% of baseline weight at week 56. Weight loss was significantly greater with both doses of liraglutide than with placebo for all three primary endpoints. Weight loss was 6.0% (6.4 kg), 4.7% (5.0 kg), and 2.0% (2.2 kg) with liraglutide 3.0 mg, liraglutide 1.8 mg, and placebo, respectively. A weight loss ≥5% occurred in 54.3, 40.4, and 21.4% of patients, respectively, and a weight loss ≥10% occurred in 25.2, 15.9, and 6.7% of patients, respectively. The safety profile was similar to that described in other clinical trials [56].
The efficacy of liraglutide 3.0 mg as an adjunct to diet and exercise was also evaluated in 3731 non-diabetic patients presenting with a BMI ≥27 (in the presence of dyslipidemia or hypertension) or ≥30 kg/m2 treated for 56 weeks. The primary endpoints were the change in body weight and the proportions of patients losing at least 5% and ≥10% of their initial body weight. Weight loss was significantly greater with liraglutide than placebo for all primary endpoints. At week 56, the mean weight loss was 8.4 ± 7.3 kg in the liraglutide group and 2.8 ± 6.5 kg in the placebo group. Weight loss of at least 5% was observed in 63.2 and 27.1% of the patients, respectively, and weight loss ≥10% was seen in 33.1 and 10.6% of the patients, respectively. The most frequently reported adverse events with liraglutide were mild or moderate nausea and diarrhea, both of which were transitory [16].
5 Concluding Remarks
It is believed that GLP-1RAs promote weight loss mainly because of their inhibitory effect on food intake [42]. However, the numerous central effects described for native GLP-1 and for some of the GLP-1RAs in rodents and in humans encourage future clinical trials exploring additional mechanisms that could potentially underlie the beneficial effects observed with this drug class. Among the aspects that deserve special attention, we highlight three: (i) the increased thermogenesis by activation of BAT or browning of the white adipose tissue; (ii) the neuroprotective properties, including the ability to reduce hypothalamic inflammation triggered by HFD; and (iii) the effects on damaged neurons in neurodegenerative diseases. Considering that hypothalamic inflammation directly interferes in neural circuits controlling food intake and energy balance, its reversal could contribute to the restoration of the hypothalamic energy set point.
References
Bauer PV, Duca FA. Targeting the gastrointestinal tract to treat type 2 diabetes. J Endocrinol. 2016;230(3):R95–113.
Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368(9548):1696–705.
Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 2013;17(6):819–37.
Farr OM, Sofopoulos M, Tsoukas MA, Dincer F, Thakkar B, Sahin-Efe A, et al. GLP-1 receptors exist in the parietal cortex, hypothalamus and medulla of human brains and the GLP-1 analogue liraglutide alters brain activity related to highly desirable food cues in individuals with diabetes: a crossover, randomised, placebo-controlled trial. Diabetologia. 2016;59(5):954–65.
Heppner KM, Kirigiti M, Secher A, Paulsen SJ, Buckingham R, Pyke C, et al. Expression and distribution of glucagon-like peptide-1 receptor mRNA, protein and binding in the male nonhuman primate (Macaca mulatta) brain. Endocrinology. 2015;156(1):255–67.
Lockie SH, Heppner KM, Chaudhary N, Chabenne JR, Morgan DA, Veyrat-Durebex C, et al. Direct control of brown adipose tissue thermogenesis by central nervous system glucagon-like peptide-1 receptor signaling. Diabetes. 2012;61(11):2753–62.
Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84(1):277–359.
Xu F, Lin B, Zheng X, Chen Z, Cao H, Xu H, et al. GLP-1 receptor agonist promotes brown remodelling in mouse white adipose tissue through SIRT1. Diabetologia. 2016;59(5):1059–69.
Gautron L, Elmquist JK, Williams KW. Neural control of energy balance: translating circuits to therapies. Cell. 2015;161(1):133–45.
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med. 2009;360(15):1509–17.
Larsen PJ, Tang-Christensen M, Holst JJ, Orskov C. Distribution of glucagon-like peptide-1 and other preproglucagon-derived peptides in the rat hypothalamus and brainstem. Neuroscience. 1997;77(1):257–70.
Roberge JN, Brubaker PL. Regulation of intestinal proglucagon-derived peptide secretion by glucose-dependent insulinotropic peptide in a novel enteroendocrine loop. Endocrinology. 1993;133(1):233–40.
Dailey MJ, Moran TH. Glucagon-like peptide 1 and appetite. Trends in endocrinology and metabolism: TEM. 2013;24(2):85–91 (PubMed PMID: 23332584. Pubmed Central PMCID: 3594872).
Tomas E, Habener JF. Insulin-like actions of glucagon-like peptide-1: a dual receptor hypothesis. Trends Endocrinol Metab. 2010;21(2):59–67.
Mayo KE, Miller LJ, Bataille D, Dalle S, Goke B, Thorens B, et al. International Union of Pharmacology. XXXV. The glucagon receptor family. Pharmacol Rev. 2003;55(1):167–94.
Pi-Sunyer X, Astrup A, Fujioka K, Greenway F, Halpern A, Krempf M, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;373(1):11–22.
Verspohl EJ. Novel therapeutics for type 2 diabetes: incretin hormone mimetics (glucagon-like peptide-1 receptor agonists) and dipeptidyl peptidase-4 inhibitors. Pharmacol Ther. 2009;124(1):113–38.
Werner U, Haschke G, Herling AW, Kramer W. Pharmacological profile of lixisenatide: a new GLP-1 receptor agonist for the treatment of type 2 diabetes. Regul Pept. 2010;164(2–3):58–64.
Minze MG, Klein MS, Jernigan MJ, Wise SL, Fruge K. Once-weekly exenatide: an extended-duration glucagon-like peptide agonist for the treatment of type 2 diabetes mellitus. Pharmacotherapy. 2013;33(6):627–38.
Knudsen LB, Nielsen PF, Huusfeldt PO, Johansen NL, Madsen K, Pedersen FZ, et al. Potent derivatives of glucagon-like peptide-1 with pharmacokinetic properties suitable for once daily administration. J Med Chem. 2000;43(9):1664–9.
Glaesner W, Vick AM, Millican R, Ellis B, Tschang SH, Tian Y, et al. Engineering and characterization of the long-acting glucagon-like peptide-1 analogue LY2189265, an Fc fusion protein. Diabetes Metab Res Rev. 2010;26(4):287–96.
Blair HA, Keating GM. Albiglutide: a review of its use in patients with type 2 diabetes mellitus. Drugs. 2015;75(6):651–63.
Baggio LL, Drucker DJ. Glucagon-like peptide-1 receptors in the brain: controlling food intake and body weight. J Clin Invest. 2014;124(10):4223–6.
Wang XF, Liu JJ, Xia J, Liu J, Mirabella V, Pang ZP. Endogenous glucagon-like peptide-1 suppresses high-fat food intake by reducing synaptic drive onto mesolimbic dopamine neurons. Cell Rep. 2015;12(5):726–33.
Sisley S, Gutierrez-Aguilar R, Scott M, D’Alessio DA, Sandoval DA, Seeley RJ. Neuronal GLP1R mediates liraglutide’s anorectic but not glucose-lowering effect. J Clin Invest. 2014;124(6):2456–63.
Secher A, Jelsing J, Baquero AF, Hecksher-Sorensen J, Cowley MA, Dalboge LS, et al. The arcuate nucleus mediates GLP-1 receptor agonist liraglutide-dependent weight loss. J Clin Invest. 2014;124(10):4473–88.
Kooijman S, Wang Y, Parlevliet ET, Boon MR, Edelschaap D, Snaterse G, et al. Central GLP-1 receptor signalling accelerates plasma clearance of triacylglycerol and glucose by activating brown adipose tissue in mice. Diabetologia. 2015;58(11):2637–46.
Wei Q, Li L, Chen JA, Wang SH, Sun ZL. Exendin-4 improves thermogenic capacity by regulating fat metabolism on brown adipose tissue in mice with diet-induced obesity. Ann Clin Lab Sci. 2015;45(2):158–65.
Beiroa D, Imbernon M, Gallego R, Senra A, Herranz D, Villarroya F, et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes. 2014;63(10):3346–58.
Heppner KM, Marks S, Holland J, Ottaway N, Smiley D, Dimarchi R, et al. Contribution of brown adipose tissue activity to the control of energy balance by GLP-1 receptor signalling in mice. Diabetologia. 2015;58(9):2124–32.
Holscher C. Potential role of glucagon-like peptide-1 (GLP-1) in neuroprotection. CNS Drugs. 2012;26(10):871–82.
Anderberg RH, Richard JE, Eerola K, Ferreras LL, Nordbeck EB, Hansson C, et al. Glucagon-like peptide-1 and its analogues act in the dorsal raphe and modulate central serotonin to reduce appetite and body weight. Diabetes. 2017. doi:10.2337/db16-0755 (Epub 2017 Jan 5).
Perry T, Lahiri DK, Chen D, Zhou J, Shaw KT, Egan JM, et al. A novel neurotrophic property of glucagon-like peptide 1: a promoter of nerve growth factor-mediated differentiation in PC12 cells. J Pharmacol Exp Ther. 2002;300(3):958–66.
Harkavyi A, Abuirmeileh A, Lever R, Kingsbury AE, Biggs CS, Whitton PS. Glucagon-like peptide 1 receptor stimulation reverses key deficits in distinct rodent models of Parkinson’s disease. J Neuroinflamm. 2008;21(5):19.
Darsalia V, Mansouri S, Ortsater H, Olverling A, Nozadze N, Kappe C, et al. Glucagon-like peptide-1 receptor activation reduces ischaemic brain damage following stroke in Type 2 diabetic rats. Clin Sci (Lond). 2012;122(10):473–83.
Hamilton A, Patterson S, Porter D, Gault VA, Holscher C. Novel GLP-1 mimetics developed to treat type 2 diabetes promote progenitor cell proliferation in the brain. J Neurosci Res. 2011;89(4):481–9.
Hunter K, Holscher C. Drugs developed to treat diabetes, liraglutide and lixisenatide, cross the blood brain barrier and enhance neurogenesis. BMC Neurosci. 2012;23(13):33.
McClean PL, Parthsarathy V, Faivre E, Holscher C. The diabetes drug liraglutide prevents degenerative processes in a mouse model of Alzheimer’s disease. J Neurosci. 2011;31(17):6587–94.
McClean PL, Holscher C. Liraglutide can reverse memory impairment, synaptic loss and reduce plaque load in aged APP/PS1 mice, a model of Alzheimer’s disease. Neuropharmacology. 2014;76(Pt A):57–67.
Barreto-Vianna AR, Aguila MB, Mandarim-de-Lacerda CA. Effects of liraglutide in hypothalamic arcuate nucleus of obese mice. Obesity. 2016;24(3):626–33.
Gao Y, Ottaway N, Schriever SC, Legutko B, Garcia-Caceres C, de la Fuente E, et al. Hormones and diet, but not body weight, control hypothalamic microglial activity. Glia. 2014;62(1):17–25.
Flint A, Raben A, Astrup A, Holst JJ. Glucagon-like peptide 1 promotes satiety and suppresses energy intake in humans. J Clin Invest. 1998;101(3):515–20.
Vilsboll T, Christensen M, Junker AE, Knop FK, Gluud LL. Effects of glucagon-like peptide-1 receptor agonists on weight loss: systematic review and meta-analyses of randomised controlled trials. BMJ. 2012;10(344):d7771.
Miyagawa J, Odawara M, Takamura T, Iwamoto N, Takita Y, Imaoka T. Once-weekly glucagon-like peptide-1 receptor agonist dulaglutide is non-inferior to once-daily liraglutide and superior to placebo in Japanese patients with type 2 diabetes: a 26-week randomized phase III study. Diabetes Obes Metab. 2015;17(10):974–83.
Dungan KM, Povedano ST, Forst T, Gonzalez JG, Atisso C, Sealls W, et al. Once-weekly dulaglutide versus once-daily liraglutide in metformin-treated patients with type 2 diabetes (AWARD-6): a randomised, open-label, phase 3, non-inferiority trial. Lancet. 2014;384(9951):1349–57.
Pratley RE, Nauck MA, Barnett AH, Feinglos MN, Ovalle F, Harman-Boehm I, et al. Once-weekly albiglutide versus once-daily liraglutide in patients with type 2 diabetes inadequately controlled on oral drugs (HARMONY 7): a randomised, open-label, multicentre, non-inferiority phase 3 study. Lancet Diabetes Endocrinol. 2014;2(4):289–97.
Baggio LL, Huang Q, Brown TJ, Drucker DJ. A recombinant human glucagon-like peptide (GLP)-1-albumin protein (albugon) mimics peptidergic activation of GLP-1 receptor-dependent pathways coupled with satiety, gastrointestinal motility, and glucose homeostasis. Diabetes. 2004;53(9):2492–500.
van Bloemendaal L, Ten Kulve JS, la Fleur SE, Ijzerman RG, Diamant M. Effects of glucagon-like peptide 1 on appetite and body weight: focus on the CNS. J Endocrinol. 2014;221(1):T1–16.
Horowitz M, Flint A, Jones KL, Hindsberger C, Rasmussen MF, Kapitza C, et al. Effect of the once-daily human GLP-1 analogue liraglutide on appetite, energy intake, energy expenditure and gastric emptying in type 2 diabetes. Diabetes Res Clin Pract. 2012;97(2):258–66.
Harder H, Nielsen L, Tu DT, Astrup A. The effect of liraglutide, a long-acting glucagon-like peptide 1 derivative, on glycemic control, body composition, and 24-h energy expenditure in patients with type 2 diabetes. Diabetes Care. 2004;27(8):1915–21.
Bradley DP, Kulstad R, Racine N, Shenker Y, Meredith M, Schoeller DA. Alterations in energy balance following exenatide administration. Appl Physiol Nutr Metab. 2012;37(5):893–9.
van Can J, Sloth B, Jensen CB, Flint A, Blaak EE, Saris WH. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int J Obes (Lond). 2014;38(6):784–93.
Pannacciulli N, Le DS, Salbe AD, Chen K, Reiman EM, Tataranni PA, et al. Postprandial glucagon-like peptide-1 (GLP-1) response is positively associated with changes in neuronal activity of brain areas implicated in satiety and food intake regulation in humans. Neuroimage. 2007;35(2):511–7.
De Silva A, Salem V, Long CJ, Makwana A, Newbould RD, Rabiner EA, et al. The gut hormones PYY 3-36 and GLP-1 7-36 amide reduce food intake and modulate brain activity in appetite centers in humans. Cell Metab. 2011;14(5):700–6.
van Bloemendaal L, IJzerman RG, Ten Kulve JS, Barkhof F, Konrad RJ, Drent ML, et al. GLP-1 receptor activation modulates appetite- and reward-related brain areas in humans. Diabetes. 2014;63(12):4186–96.
Davies MJ, Bergenstal R, Bode B, Kushner RF, Lewin A, Skjoth TV, et al. Efficacy of liraglutide for weight loss among patients with type 2 diabetes: the SCALE Diabetes Randomized Clinical Trial. JAMA. 2015;314(7):687–99.
Byetta® (exenatide) injection - full prescribing information. 2005. http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/021773s9s11s18s22s25lbl.pdf. Accessed 13 Feb 2017.
FDA. Adlyxin (lixisenatide) injection. 2016. http://www.accessdata.fda.gov/drugsatfda_docs/label/2016/208471Orig1s000lbl.pdf. Accessed 13 Feb 2017.
FDA. Victoza injection. 2010. https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/022341lbl.pdf. Accessed 13 Feb 2017.
FDA. Exenatide extended-release for injectable suspension. 2014. http://www.accessdata.fda.gov/drugsatfda_docs/nda/2014/022200Orig1s008.pdf. Accessed 13 Feb 2017.
FDA. Tanzeum (albiglutide) for injection. 2014. https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/125431s000lbl.pdf. Accessed 13 Feb 2017.
FDA. Trulicity (dulaglutide) injection. 2014. http://www.accessdata.fda.gov/drugsatfda_docs/appletter/2015/125469Orig1s004ltr.pdf. Accessed 13 Feb 2017.
Svane MS, Bojsen-Moller KN, Nielsen S, Jorgensen NB, Dirksen C, Bendtsen F, et al. Effects of endogenous GLP-1 and GIP on glucose tolerance after Roux-en-Y gastric bypass surgery. Am J Physiol Endocrinol Metab. 2016;310(7):E505–14.
Retnakaran R, Kramer CK, Choi H, Swaminathan B, Zinman B. Liraglutide and the preservation of pancreatic beta-cell function in early type 2 diabetes: the LIBRA trial. Diabetes Care. 2014;37(12):3270–8.
Lin CH, Hsieh SH, Sun JH, Tsai JS, Huang YY. Glucose variability and beta-cell response by GLP-1 analogue added-on CSII for patients with poorly controlled type 2 diabetes. Sci Rep. 2015;26(5):16968.
van Raalte DH, Bunck MC, Smits MM, Hoekstra T, Corner A, Diamant M, et al. Exenatide improves beta-cell function up to 3 years of treatment in patients with type 2 diabetes: a randomised controlled trial. Eur J Endocrinol. 2016;175(4):345–52.
Brill AL, Wisinski JA, Cadena MT, Thompson MF, Fenske RJ, Brar HK, et al. Synergy between Gαz deficiency and GLP-1 analog treatment in preserving functional β-cell mass in experimental diabetes. Mol Endocrinol. 2016;30(5):543–56.
Green AD, Vasu S, Moffett RC, Flatt PR. Co-culture of clonal beta cells with GLP-1 and glucagon-secreting cell line impacts on beta cell insulin secretion, proliferation and susceptibility to cytotoxins. Biochimie. 2016;125:119–25.
Abe H, Uchida T, Hara A, Mizukami H, Komiya K, Koike M, et al. Exendin-4 improves beta-cell function in autophagy-deficient beta-cells. Endocrinology. 2013;154(12):4512–24.
Lim SW, Jin L, Jin J, Yang CW. Effect of exendin-4 on autophagy clearance in beta cell of rats with tacrolimus-induced diabetes mellitus. Sci Rep. 2016;20(6):29921.
Kim MH, Kim EH, Jung HS, Yang D, Park EY, Jun HS. EX4 stabilizes and activates Nrf2 via PKCdelta, contributing to the prevention of oxidative stress-induced pancreatic beta cell damage. Toxicol Appl Pharmacol. 2016;07(315):60–9.
Drucker DJ. The biology of incretin hormones. Cell Metab. 2006;3(3):153–65.
Drucker DJ. The cardiovascular biology of glucagon-like peptide-1. Cell Metab. 2016;24(1):15–30.
Wang B, Zhong J, Lin H, Zhao Z, Yan Z, He H, et al. Blood pressure-lowering effects of GLP-1 receptor agonists exenatide and liraglutide: a meta-analysis of clinical trials. Diabetes Obes Metab. 2013;15(8):737–49.
Nauck MA, Kemmeries G, Holst JJ, Meier JJ. Rapid tachyphylaxis of the glucagon-like peptide 1-induced deceleration of gastric emptying in humans. Diabetes. 2011;60(5):1561–5.
He W, Yu S, Wang L, He M, Cao X, Li Y, et al. Exendin-4 inhibits growth and augments apoptosis of ovarian cancer cells. Mol Cell Endocrinol. 2016;15(436):240–9.
Gutzwiller JP, Tschopp S, Bock A, Zehnder CE, Huber AR, Kreyenbuehl M, et al. Glucagon-like peptide 1 induces natriuresis in healthy subjects and in insulin-resistant obese men. J Clin Endocrinol Metab. 2004;89(6):3055–61.
Zhao X, Liu G, Shen H, Gao B, Li X, Fu J, et al. Liraglutide inhibits autophagy and apoptosis induced by high glucose through GLP-1R in renal tubular epithelial cells. Int J Mol Med. 2015;35(3):684–92.
Valdecantos MP, Pardo V, Ruiz L, Castro-Sanchez L, Lanzon B, Fernandez-Millan E, et al. A novel glucagon-like peptide 1/glucagon receptor dual agonist improves steatohepatitis and liver regeneration in mice. Hepatology. 2016. doi:10.1002/hep.28962 (Epub 2016 Nov 23).
Gastaldelli A, Gaggini M, Daniele G, Ciociaro D, Cersosimo E, Tripathy D, et al. Exenatide improves both hepatic and adipose tissue insulin resistance: a dynamic positron emission tomography study. Hepatology. 2016;64(6):2028–37.
Hadjiyanni I, Siminovitch KA, Danska JS, Drucker DJ. Glucagon-like peptide-1 receptor signalling selectively regulates murine lymphocyte proliferation and maintenance of peripheral regulatory T cells. Diabetologia. 2010;53(4):730–40.
Schlogl H, Kabisch S, Horstmann A, Lohmann G, Muller K, Lepsien J, et al. Exenatide-induced reduction in energy intake is associated with increase in hypothalamic connectivity. Diabetes Care. 2013;36(7):1933–40.
van Bloemendaal L, Veltman DJ, ten Kulve JS, Drent ML, Barkhof F, Diamant M, et al. Emotional eating is associated with increased brain responses to food-cues and reduced sensitivity to GLP-1 receptor activation. Obesity. 2015;23(10):2075–82.
van Bloemendaal L, Veltman DJ, Ten Kulve JS, Groot PF, Ruhe HG, Barkhof F, et al. Brain reward-system activation in response to anticipation and consumption of palatable food is altered by glucagon-like peptide-1 receptor activation in humans. Diabetes Obes Metab. 2015;17(9):878–86.
Athauda D, Foltynie T. The glucagon-like peptide 1 (GLP) receptor as a therapeutic target in Parkinson’s disease: mechanisms of action. Drug Discov Today. 2016;21(5):802–18.
ten Kulve JS, Veltman DJ, van Bloemendaal L, Barkhof F, Deacon CF, Holst JJ, et al. Endogenous GLP-1 mediates postprandial reductions in activation in central reward and satiety areas in patients with type 2 diabetes. Diabetologia. 2015;58(12):2688–98.
Daniele G, Iozzo P, Molina-Carrion M, Lancaster J, Ciociaro D, Cersosimo E, et al. Exenatide regulates cerebral glucose metabolism in brain areas associated with glucose homeostasis and reward system. Diabetes. 2015;64(10):3406–12.
Eldor R, Daniele G, Huerta C, Al-Atrash M, Adams J, DeFronzo R, et al. Discordance between central (brain) and pancreatic action of exenatide in lean and obese subjects. Diabetes Care. 2016;39(10):1804–10.
Aviles-Olmos I, Dickson J, Kefalopoulou Z, Djamshidian A, Ell P, Soderlund T, et al. Exenatide and the treatment of patients with Parkinson’s disease. J Clin Invest. 2013;123(6):2730–6.
Farr OM, Tsoukas MA, Triantafyllou G, Dincer F, Filippaios A, Ko BJ, et al. Short-term administration of the GLP-1 analog liraglutide decreases circulating leptin and increases GIP levels and these changes are associated with alterations in CNS responses to food cues: a randomized, placebo-controlled, crossover study. Metabolism. 2016;65(7):945–53.
Ten Kulve JS, Veltman DJ, van Bloemendaal L, Barkhof F, Drent ML, Diamant M, et al. Liraglutide reduces CNS activation in response to visual food cues only after short-term treatment in patients with type 2 diabetes. Diabetes Care. 2016;39(2):214–21.
Femminella GD, Edison P. Evaluation of neuroprotective effect of glucagon-like peptide 1 analogs using neuroimaging. Alzheimers Dement. 2014;10(1 Suppl):S55–61.
Authors’ contributions
BG, JCL, and LAV participated in the concept and drafting of the manuscript, and performed the critical review for intellectual content. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The authors wish to thank Daniel Soares Freire, MD, PhD for providing medical writing and Cristiane Mapurunga Aoqui Guenoub, MD, PhD for providing assistance with English-language editing, both on behalf of Springer Healthcare. This manuscript was prepared according to the International Society for Medical Publication Professionals’ Good Publication Practice for Communicating Company-Sponsored Medical Research: the GPP3 Guidelines. Funding to support the preparation of this manuscript was provided by Novo Nordisk Inc. The authors take full responsibility for the content and conclusions stated in this manuscript. Novo Nordisk did not influence the content of this publication.
Conflict of interest
BG reports receiving fees for serving on an advisory board from Novo Nordisk, AstraZeneca, and MSD; lecture fees from Novo Nordisk, AstraZeneca, and MSD; and grant support from Novo Nordisk and Boehringer Ingelheim. JCL and LAV declare that they have no competing interests that would influence the content of this review.
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s40265-017-0766-5.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Geloneze, B., de Lima-Júnior, J.C. & Velloso, L.A. Glucagon-Like Peptide-1 Receptor Agonists (GLP-1RAs) in the Brain–Adipocyte Axis. Drugs 77, 493–503 (2017). https://doi.org/10.1007/s40265-017-0706-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40265-017-0706-4