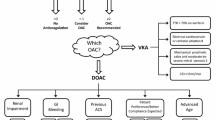
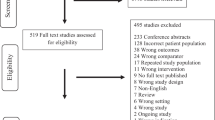
Abstract
Direct oral anticoagulants are now recommended by major guidelines as first-choice agents for both stroke prevention in non-valvular atrial fibrillation and treatment/prevention of venous thromboembolism in non-cancer patients. Although there are no published head-to-head trials comparing different direct oral anticoagulants, a growing body of evidence from indirect comparisons and observational studies is suggesting that each direct oral anticoagulant may have a specific risk profile. This review aims to (1) synthesize and critically assess the latest evidence in comparative effectiveness and safety research in the aforementioned consolidated therapeutic uses, by performing an overview of systematic reviews and (2) highlight current challenges, namely underexplored areas, where research should be directed, also considering ongoing unpublished studies. The evidence gathered so far on the risk–benefit profile of direct oral anticoagulants is appraised in the light of existing guidelines to discuss whether further implementation should be proposed.

Similar content being viewed by others
References
Huisman MV, Rothman KJ, Paquette M, et al. The changing landscape for stroke prevention in AF: findings from the GLORIA-AF Registry Phase 2. J Am Coll Cardiol. 2017;69(7):777–85.
Raschi E, Bianchin M, Ageno W, et al. Risk-benefit profile of direct-acting oral anticoagulants in established therapeutic indications: an overview of systematic reviews and observational studies. Drug Saf. 2016;39(12):1175–87.
Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST Guideline and Expert Panel Report. Chest. 2016;149(2):315–52.
Lip GYH, Banerjee A, Boriani G, et al. Antithrombotic therapy for atrial fibrillation: CHEST Guideline and Expert Panel Report. Chest. 2018;154(5):1121–201.
January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrialfFibrillation. Circulation. 2019;140(2):e125–51.
Aronow WS, Shamliyan TA. Comparative effectiveness and safety of rivaroxaban in adults with nonvalvular atrial fibrillation. Am J Ther. 2018. https://doi.org/10.1097/mjt.0000000000000890(Epub ahead of print).
Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
Almutairi AR, Zhou L, Gellad WF, et al. Effectiveness and safety of non-vitamin K antagonist oral anticoagulants for atrial fibrillation and venous thromboembolism: a systematic review and meta-analyses. Clin Ther. 2017;39(7):1456–78.
Bai Y, Guo SD, Deng H, et al. Effectiveness and safety of oral anticoagulants in older patients with atrial fibrillation: a systematic review and meta-regression analysis. Age Ageing. 2018;47(1):9–17.
Cohen AT, Hamilton M, Bird A, et al. Comparison of the non-VKA oral anticoagulants apixaban, dabigatran, and rivaroxaban in the extended treatment and prevention of venous thromboembolism: systematic review and network meta-analysis. PLoS One. 2016;11:e0160064.
Cohen AT, Hill NR, Luo X, et al. A systematic review of network meta-analyses among patients with nonvalvular atrial fibrillation: a comparison of efficacy and safety following treatment with direct oral anticoagulants. Int J Cardiol. 2018;269:174–81.
López-López JA, Sterne JAC, Thom HHZ, et al. Oral anticoagulants for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis, and cost effectiveness analysis. BMJ. 2017;359:j5058.
Sterne JA, Bodalia PN, Bryden PA, et al. Oral anticoagulants for primary prevention, treatment and secondary prevention of venous thromboembolic disease, and for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis and cost-effectiveness analysis. Health Technol Assess. 2017;21(9):1–386.
Deitelzweig S, Farmer C, Luo X, et al. Comparison of major bleeding risk in patients with non-valvular atrial fibrillation receiving direct oral anticoagulants in the real-world setting: a network meta-analysis. Curr Med Res Opin. 2018;34(3):487–98.
Deitelzweig S, Farmer C, Luo X, et al. Risk of major bleeding in patients with non-valvular atrial fibrillation treated with oral anticoagulants: a systematic review of real-world observational studies. Curr Med Res Opin. 2017;33(9):1583–94.
Bai Y, Deng H, Shantsila A, et al. Rivaroxaban versus dabigatran or warfarin in real-world studies of stroke prevention in atrial fibrillation: systematic review and meta-analysis. Stroke. 2017;48(4):970–6.
Bai Y, Shi XB, Ma CS, et al. Meta-analysis of effectiveness and safety of oral anticoagulants in atrial fibrillation with focus on apixaban. Am J Cardiol. 2017;120(9):1689–95.
Bundhun PK, Soogund MZ, Teeluck AR, et al. Bleeding outcomes associated with rivaroxaban and dabigatran in patients treated for atrial fibrillation: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2017;17:15.
Proietti M, Romanazzi I, Romiti GF, Farcomeni A, Lip GYH. Real-world use of apixaban for stroke prevention in atrial fibrillation: a systematic review and meta-analysis. Stroke. 2018;49(1):98–106.
Douros A, Durand M, Doyle CM, et al. Comparative effectiveness and safety of direct oral anticoagulants in patients with atrial fibrillation: a systematic review and meta-analysis of observational studies. Drug Saf. 2019;42(10):1135–48.
Li G, Lip GYH, Holbrook A, et al. Direct comparative effectiveness and safety between non-vitamin K antagonist oral anticoagulants for stroke prevention in nonvalvular atrial fibrillation: a systematic review and meta-analysis of observational studies. Eur J Epidemiol. 2019;34(2):173–90.
Sun Z, Liu Y, Zhang Y, et al. Differences in safety and efficacy of oral anticoagulants in patients with non-valvular atrial fibrillation: a Bayesian analysis. Int J Clin Pract. 2019;73(4):e13308.
Cohen AT, Berger SE, Milenkovic D, et al. Anticoagulant selection for patients with VTE: evidence from a systematic literature review of network meta-analyses. Pharmacol Res. 2019;143:166–77.
Frost C, Song Y, Barrett YC, et al. A randomized direct comparison of the pharmacokinetics and pharmacodynamics of apixaban and rivaroxaban. Clin Pharmacol. 2014;6:179–87.
Ageno W, Beyer-Westendorf J, Rubboli A. Once- versus twice-daily direct oral anticoagulants in non-valvular atrial fibrillation. Expert Opin Pharmacother. 2017;18(13):1325–32.
Suissa S, Moodie EE, Dell’Aniello S. Prevalent new-user cohort designs for comparative drug effect studies by time-conditional propensity scores. Pharmacoepidemiol Drug Saf. 2017;26(4):459–68.
Schisterman EF, Cole SR, Platt RW. Overadjustment bias and unnecessary adjustment in epidemiologic studies. Epidemiology. 2009;20(4):488–95.
Carrier M, Le GG, Wells PS, et al. Systematic review: case-fatality rates of recurrent venous thromboembolism and major bleeding events among patients treated for venous thromboembolism. Ann Intern Med. 2010;152(9):578–89.
Barbui C, Addis A, Amato L, et al. Can systematic reviews contribute to regulatory decisions? Eur J Clin Pharmacol. 2017;73(4):507–9.
Prada-Ramallal G, Takkouche B, Figueiras A. Summarising the evidence for drug safety: a methodological discussion of different meta-analysis approaches. Drug Saf. 2017;40(7):547–58.
Doundoulakis I, Antza C, Postolidou-Kiouti F, et al. Overview of systematic reviews of non-vitamin K oral anticoagulants in atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2018;11(12):e004769.
Tritschler T, Castellucci LA. It’s time for head-to-head trials with direct oral anticoagulants. Thromb Res. 2019;180:64–9.
Chan YH, Kuo CT, Yeh YH, et al. Thromboembolic, bleeding, and mortality risks of rivaroxaban and dabigatran in Asians with nonvalvular atrial fibrillation. J Am Coll Cardiol. 2016;68(13):1389–401.
Chan YH, Lee HF, See LC, et al. Effectiveness and safety of four direct oral anticoagulants in Asian patients with nonvalvular atrial fibrillation. Chest. 2019;156(3):529–43.
Diener HC, Aisenberg J, Ansell J, et al. Choosing a particular oral anticoagulant and dose for stroke prevention in individual patients with non-valvular atrial fibrillation: part 1. Eur Heart J. 2017;38(12):852–9.
Diener HC, Aisenberg J, Ansell J, et al. Choosing a particular oral anticoagulant and dose for stroke prevention in individual patients with non-valvular atrial fibrillation: part 2. Eur Heart J. 2017;38(12):860–8.
Lip GY, Lane DA. Matching the NOAC to the patient: remember the modifiable bleeding risk factors. J Am Coll Cardiol. 2015;66(21):2282–4.
Shields AM, Lip GY. Choosing the right drug to fit the patient when selecting oral anticoagulation for stroke prevention in atrial fibrillation. J Intern Med. 2015;278(1):1–18.
Paravattil B, Elewa H. Approaches to direct oral anticoagulant selection in practice. J Cardiovasc Pharmacol Ther. 2018. https://doi.org/10.1177/1074248418793137(Epub ahead of print).
Mueller T, Varez-Madrazo S, Robertson C, et al. Comparative safety and effectiveness of direct oral anticoagulants in patients with atrial fibrillation in clinical practice in Scotland. Br J Clin Pharmacol. 2019;85(2):422–31.
Maura G, Billionnet C, Coste J, et al. Non-bleeding adverse events with the use of direct oral anticoagulants: a sequence symmetry analysis. Drug Saf. 2018;41(9):881–97.
Raschi E, Poluzzi E, Koci A, et al. Liver injury with novel oral anticoagulants: assessing post-marketing reports in the US Food and Drug Administration adverse event reporting system. Br J Clin Pharmacol. 2015;80(2):285–93.
Douros A, Azoulay L, Yin H, et al. Non-vitamin K antagonist oral anticoagulants and risk of serious liver injury. J Am Coll Cardiol. 2018;71(109):1105–13.
Alonso A, MacLehose RF, Chen LY, et al. Prospective study of oral anticoagulants and risk of liver injury in patients with atrial fibrillation. Heart. 2017;103(11):834–9.
Qamar A, Vaduganathan M, Greenberger NJ, et al. Oral anticoagulation in patients with liver disease. J Am Coll Cardiol. 2018;71(19):2162–75.
Lee SR, Lee HJ, Choi EK, et al. Direct oral anticoagulants in patients with atrial fibrillation and liver disease. J Am Coll Cardiol. 2019;73(25):3295–308.
Raschi E, Bianchin M, Ageno W, et al. Adverse events associated with the use of direct-acting oral anticoagulants in clinical practice: beyond bleeding complications. Pol Arch Med Wewn. 2016;126(7–8):552–61.
Wei AH, Gu ZC, Zhang C, et al. Increased risk of myocardial infarction with dabigatran etexilate: fact or fiction? A critical meta-analysis of over 580,000 patients from integrating randomized controlled trials and real-world studies. Int J Cardiol. 2018;267:1–7.
Lee CJ, Gerds TA, Carlson N, et al. Risk of myocardial infarction in anticoagulated patients with atrial fibrillation. J Am Coll Cardiol. 2018;72(1):17–26.
Anand SS, Bosch J, Eikelboom JW, et al. Rivaroxaban with or without aspirin in patients with stable peripheral or carotid artery disease: an international, randomised, double-blind, placebo-controlled trial. Lancet. 2018;391(10117):219–29.
Connolly SJ, Eikelboom JW, Bosch J, et al. Rivaroxaban with or without aspirin in patients with stable coronary artery disease: an international, randomised, double-blind, placebo-controlled trial. Lancet. 2018;391(10117):205–18.
Cosentino F, Grant PJ, Aboyans V, et al. ESC guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2019. https://doi.org/10.1093/eurheartj/ehz486(Epub ahead of print).
Capodanno D, Huber K, Mehran R, et al. Management of antithrombotic therapy in atrial fibrillation patients undergoing PCI: JACC state-of-the-art review. J Am Coll Cardiol. 2019;74(1):83–99.
Nielsen PB, Skjoth F, Sogaard M, et al. Effectiveness and safety of reduced dose non-vitamin K antagonist oral anticoagulants and warfarin in patients with atrial fibrillation: propensity weighted nationwide cohort study. BMJ. 2017;356:j510.
Vinogradova Y, Coupland C, Hill T, et al. Risks and benefits of direct oral anticoagulants versus warfarin in a real world setting: cohort study in primary care. BMJ. 2018;362:k2505.
Maura G, Billionnet C, Drouin J, et al. Oral anticoagulation therapy use in patients with atrial fibrillation after the introduction of non-vitamin K antagonist oral anticoagulants: findings from the French healthcare databases, 2011-2016. BMJ Open. 2019;9:e026645.
Haastrup SB, Hellfritzsch M, Rasmussen L, et al. Use of non-vitamin K antagonist oral anticoagulants 2008-2016: a Danish nationwide cohort study. Basic Clin Pharmacol Toxicol. 2018;123(4):452–63.
Maura G, Pariente A, Alla F, et al. Adherence with direct oral anticoagulants in nonvalvular atrial fibrillation new users and associated factors: a French nationwide cohort study. Pharmacoepidemiol Drug Saf. 2017;26(11):1367–77.
Ray WA, Chung CP, Murray KT, et al. Association of oral anticoagulants and proton pump inhibitor cotherapy with hospitalization for upper gastrointestinal tract bleeding. JAMA. 2018;320(21):2221–30.
Tadrous M, Gagne JJ, Sturmer T, et al. Disease risk score as a confounder summary method: systematic review and recommendations. Pharmacoepidemiol Drug Saf. 2013;22(2):122–9.
Dawwas GK, Brown J, Dietrich E, et al. Effectiveness and safety of apixaban versus rivaroxaban for prevention of recurrent venous thromboembolism and adverse bleeding events in patients with venous thromboembolism: a retrospective population-based cohort analysis. Lancet Haematol. 2019;6(1):e20–8.
Ay C, Beyer-Westendorf J, Pabinger I. Treatment of cancer-associated venous thromboembolism in the age of direct oral anticoagulants. Ann Oncol. 2019. https://doi.org/10.1093/annonc/mdz111(Epub ahead of print).
Agnelli G. Direct oral anticoagulants for thromboprophylaxis in ambulatory patients with cancer. N Engl J Med. 2019;380(8):781–3.
Raschi E, Diemberger I, Cosmi B, et al. ESC position paper on cardiovascular toxicity of cancer treatments: challenges and expectations. Intern Emerg Med. 2018;13(1):1–9.
Raschi E, Bianchin M, Fantoni C, et al. Evolving cardiovascular uses of direct-acting oral anticoagulants: a paradigm shift on the horizon? Intern Emerg Med. 2017;12(7):923–34.
Raschi E, Bianchin M, De Ponti R, et al. Emerging therapeutic uses of direct-acting oral anticoagulants: an evidence-based perspective. Pharmacol Res. 2017;120:206–18.
Lobraico-Fernandez J, Baksh S, Nemec E. Elderly bleeding risk of direct oral anticoagulants in nonvalvular atrial fibrillation: a systematic review and meta-analysis of cohort studies. Drugs R D. 2019;19(3):235–45. https://doi.org/10.1007/s40268-019-0275-y.
Fawzy AM, Lip GYH. Pharmacokinetics and pharmacodynamics of oral anticoagulants used in atrial fibrillation. Expert Opin Drug Metab Toxicol. 2019;15(5):381–98.
Gelosa P, Castiglioni L, Tenconi M, et al. Pharmacokinetic drug interactions of the non-vitamin K antagonist oral anticoagulants (NOACs). Pharmacol Res. 2018;135:60–79.
Riess H, Prandoni P, Harder S, et al. Direct oral anticoagulants for the treatment of venous thromboembolism in cancer patients: potential for drug-drug interactions. Crit Rev Oncol Hematol. 2018;132:169–79.
Fitzgerald JL, Howes LG. Drug interactions of direct-acting oral anticoagulants. Drug Saf. 2016;39(9):841–5.
Vazquez SR. Drug-drug interactions in an era of multiple anticoagulants: a focus on clinically relevant drug interactions. Blood. 2018;132(21):2230–9.
Mouly S, Lloret-Linares C, Sellier PO, et al. Is the clinical relevance of drug-food and drug-herb interactions limited to grapefruit juice and Saint-John’s wort? Pharmacol Res. 2017;118:82–92.
Di Minno A, Frigerio B, Spadarella G, et al. Old and new oral anticoagulants: food, herbal medicines and drug interactions. Blood Rev. 2017;31(4):193–203.
Steffel J, Verhamme P, Potpara TS, et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur Heart J. 2018;39(16):1330–93.
Chang SH, Chou IJ, Yeh YH, et al. Association between use of non-vitamin K oral anticoagulants with and without concurrent medications and risk of major bleeding in nonvalvular atrial fibrillation. JAMA. 2017;318(13):1250–9.
Antoniou T, Macdonald EM, Yao Z, et al. Association between statin use and ischemic stroke or major hemorrhage in patients taking dabigatran for atrial fibrillation. CMAJ. 2017;189(1):E4–10.
Ho BL, Lin YJ, Lin SF, et al. Statins and the risk of bleeding in patients taking dabigatran. Acta Neurol Scand. 2019;139(5):455–61.
Undas A, Brummel-Ziedins KE, Mann KG. Anticoagulant effects of statins and their clinical implications. Thromb Haemost. 2014;111(3):392–400.
Antonazzo IC, Poluzzi E, Forcesi E, et al. Myopathy with DPP-4 inhibitors and statins in the real world: investigating the likelihood of drug-drug interactions through the FDA adverse event reporting system. Acta Diabetol. 2019. https://doi.org/10.1007/s00592-019-01378-7(Epub ahead of print).
Ibanez L, Sabate M, Vidal X, et al. Incidence of direct oral anticoagulant use in patients with non-valvular atrial fibrillation and characteristics of users in six European countries (2008-2015): a cross-national drug utilization study. Br J Clin Pharmacol. 2019. https://doi.org/10.1111/bcp.14071(Epub ahead of print).
Aronow WS, Shamliyan TA. Comparative clinical outcomes of edoxaban in adults with nonvalvular atrial fibrillation. Am J Ther. 2018. https://doi.org/10.1097/mjt.0000000000000848(Epub ahead of print).
McGettigan P, Alonso Olmo C, Plueschke K, et al. Patient registries: an underused resource for medicines evaluation: operational proposals for increasing the use of patient registries in regulatory assessments. Drug Saf. 2019. https://doi.org/10.1007/s40264-019-00859-6(Epub ahead of print).
Kirchhof P, Radaideh G, Kim YH, et al. Global prospective safety analysis of rivaroxaban. J Am Coll Cardiol. 2018;72(2):141–53.
Poli D, Antonucci E, Ageno W, et al. Oral anticoagulation in very elderly patients with atrial fibrillation: results from the prospective multicenter START2-REGISTER study. PLoS One. 2019;14(5):e0216831.
Author information
Authors and Affiliations
Contributions
ER conceived the idea and the overall content of this review, performed the literature search and data analysis, and drafted and critically revised the work. MB provided a substantial contribution to data analysis. All authors critically revised the content and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Funding
Emanuel Raschi, Matteo Bianchin, Milo Gatti, and Fabrizio De Ponti at the University of Bologna are supported by institutional research funds (Ricerca Fondamentale Orientata). No sources of funding were received for the preparation of this article.
Conflict of interest
Emanuel Raschi, Matteo Bianchin, Milo Gatti, and Fabrizio De Ponti have no conflicts of interest that are directly relevant to the content of this article. Alessandro Squizzato received fees for lectures and advisory board meetings from Daiichi Sankyo, Pfizer, Bristol Myers Squibb, Bayer, Boehringer Ingelheim, Sanofi, and Techdow.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Raschi, E., Bianchin, M., Gatti, M. et al. Comparative Effectiveness and Safety of Direct Oral Anticoagulants: Overview of Systematic Reviews. Drug Saf 42, 1409–1422 (2019). https://doi.org/10.1007/s40264-019-00866-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40264-019-00866-7