Abstract
Background and Objectives
While several studies have examined the pharmacokinetics of tobramycin in patients with cystic fibrosis (CF), there is no consensus on whether they differ in patients with and without CF. The objectives of this study were to identify covariates which explain pharmacokinetic variability and to examine whether having the disease CF in itself alters these relationships and drug dose requirements.
Methods
To investigate this issue, a population pharmacokinetic meta-analysis of data from eight centres was undertaken. NONMEM® 7.2 was used to analyse the data, which comprised 4,514 concentration–time measurements from 465 adults and children with CF and 1,095 concentration–time measurements from 267 adults and children without CF.
Results
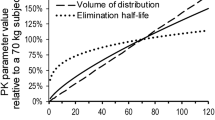
Tobramycin disposition was well described by a two-compartment model with first-order elimination. Patient age, fat-free mass, serum creatinine concentration and sex were identified as significant covariates in the final model. Fat-free mass was superior to total bodyweight as a descriptor of clearance, volume of distribution of the central and peripheral compartments and inter-compartmental clearance. CF as an independent disease-specific factor had no significant influence on the pharmacokinetics of tobramycin at any stage during covariate model building. An optimal dose of 11 mg/kg every 24 h was defined for CF patients using a utility function approach.
Conclusion
The pharmacokinetics of tobramycin do not differ significantly in CF patients compared with patients without CF when subject age, fat-free mass, sex and renal function are taken into consideration. Variations in tobramycin dosing between CF and non-CF patients should therefore reflect target concentrations or exposures based on differences in expected pathogen sensitivity and not the presence of CF.




Similar content being viewed by others
References
Levy J, Smith AL, Koup JR, Williams-Warren J, Ramsey B. Disposition of tobramycin in patients with cystic fibrosis: a prospective controlled study. J Pediatr. 1984;105(1):117–24.
Leeder JS, Spino M, Isles AF, Tesoro AM, Gold R, MacLeod SM. Ceftazidime disposition in acute and stable cystic fibrosis. Clin Pharmacol Ther. 1984;36(3):355–62.
Kearns GL, Hilman BC, Wilson JT. Dosing implications of altered gentamicin disposition in patients with cystic fibrosis. J Pediatr. 1982;100(2):312–8.
Bulitta JB, Duffull SB, Kinzig-Schippers M, Holzgrabe U, Stephan U, Drusano GL, et al. Population pharmacokinetics and pharmacodynamics of piperacillin: systematic comparison of cystic fibrosis patients and healthy volunteers. Antimicrob Agents Chemother. 2007;51(7):2497–507.
Bulitta J, Duffull SB, Kinzig-Schnippers M, Holzgrabe U, Stephan U, Soergel F, editors. Cystic fibrosis patients are pharmacokinetically comparable to healthy volunteers [abstract no. 1923 (A-12)]. Interscience Conference on Antimicrobial Agents and Chemotherapy; 16–19 Dec 2005; Washington, DC.
Touw DJ, Vinks AA, Heijerman HG, Hermans J, Bakker W. Suggestions for the optimization of the initial tobramycin dose in adolescent and adult patients with cystic fibrosis. Ther Drug Monit. 1994;16(2):125–31.
Bulitta JB, Duffull SB, Kinzig-Schippers M, Holzgrabe U, Stephan U, Drusano GL, et al. Systematic comparison of the population pharmacokinetics and pharmacodynamics of piperacillin in cystic fibrosis patients and healthy volunteers. Antimicrob Agents Chemother. 2007;51(7):2497–507.
Touw DJ, Knox AJ, Smyth A. Population pharmacokinetics of tobramycin administered thrice daily and once daily in children and adults with cystic fibrosis. J Cyst Fibros. 2007;6(5):327–33.
Hennig S, Norris R, Kirkpatrick CM. Target concentration intervention is needed for tobramycin dosing in paediatric patients with cystic fibrosis: a population pharmacokinetic study. Br J Clin Pharmacol. 2008;65(4):502–10.
Massie J, Cranswick N. Pharmacokinetic profile of once daily intravenous tobramycin in children with cystic fibrosis. J Paediatr Child Health. 2006;42(10):601–5.
Campbell D, Thomson AH, Stack B. Population pharmacokinetics of aminoglycoside antibiotics in patients with cystic fibrosis. Ther Drug Monit. 1999;21(3):281–8.
Lam W, Tjon J, Seto W, Dekker A, Wong C, Atenafu E, et al. Pharmacokinetic modelling of a once-daily dosing regimen for intravenous tobramycin in paediatric cystic fibrosis patients. J Antimicrob Chemother. 2007;59(6):1135–40.
Bracco D, Landry C, Dubois MJ, Eggimann P. Pharmacokinetic variability of extended interval tobramycin in burn patients. Burns. 2008;34(6):791–6.
de Hoog M, Schoemaker RC, van den Anker JN, Vinks AA. NONMEM and NPEM2 population modeling: a comparison using tobramycin data in neonates. Ther Drug Monit. 2002;24(3):359–65.
Aarons L, Vozeh S, Wenk M, Weiss P, Follath F. Population pharmacokinetics of tobramycin. Br J Clin Pharmacol. 1989;28(3):305–14.
Standing JS, Greening EG, Holden V, Picton S, Young N, Chrystyn H, et al. Predicting paediatric tobramycin pharmacokinetics with five different methods [abstract]. Annual Meeting of the Population Approach Group in Europe; 18–20 Jun 2008; Marseille. http://www.page-meeting.org/?abstract=1401. Accessed 14 Jan 2013.
Beal S, Sheiner LB, Boeckmann A, Bauer RJ. NONMEM user’s guides (1989–2009). 7th ed. Ellicott City: Icon Development Solutions; 2009.
Lindbom L, Pihlgren P, Jonsson EN. PsN-Toolkit: a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput Methods Programs Biomed. 2005;79(3):241–57.
Jonsson EN, Karlsson MO. Xpose: an S-PLUS based population pharmacokinetic/pharmacodynamic model building aid for NONMEM. Comput Methods Programs Biomed. 1999;58(1):51–64.
Janmahasatian S, Duffull SB, Ash S, Ward LC, Byrne NM, Green B. Quantification of lean bodyweight. Clin Pharmacokinet. 2005;44(10):1051–65.
Hennig S, Karlsson MO. Which matrix is the most reliable to judge the inclusion of covariates: reduction of unexplained parameter variability, increase in explained parameter variability or change in OFV? [abstract no. 1301]. The 14th Annual meeting of the Population Approach Group Australia and New Zealand (PAGANZ); 6–8 Feb 2012; Melbourne. http://www.paganz.org/abstracts/which-matrix-is-the-most-reliable-to-judge-the-inclusion-of-covariates-reduction-of-unexplained-parameter-variability-increase-in-explained-parameter-variability-or-change-in-ofv/. Accessed 22 Jan 2013.
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16(1):31–41.
Schwartz GJ, Feld LG, Langford DJ. A simple estimate of glomerular filtration rate in full-term infants during the first year of life. J Pediatr. 1984;104(6):849–54.
Schwartz GJ, Gauthier B. A simple estimate of glomerular filtration rate in adolescent boys. J Pediatr. 1985;106(3):522–6.
Ceriotti F, Boyd JC, Klein G, Henny J, Queralto J, Kairisto V, et al. Reference intervals for serum creatinine concentrations: assessment of available data for global application. Clin Chem. 2008;54(3):559–66.
Junge W, Wilke B, Halabi A, Klein G. Determination of reference intervals for serum creatinine, creatinine excretion and creatinine clearance with an enzymatic and a modified Jaffe method. Clin Chim Acta. 2004;344(1–2):137–48.
Johansson AM, Hill N, Perisoglou M, Whelan J, Karlsson MO, Standing JF. A population pharmacokinetic/pharmacodynamic model of methotrexate and mucositis scores in osteosarcoma. Ther Drug Monit. 2011;33(6):711–8.
Karlsson MO, Holford NHG. A tutorial on visual predictive checks [abstract no. 1434]. Annual Meeting of the Population Approach Group in Europe; 18–20 Jun 2008; Marseille. http://www.page-meeting.org/?abstract=1434. Accessed 14 Jan 2013.
Bergstrand M, Hooker AC, Wallin JE, Karlsson MO. Prediction-corrected visual predictive checks for diagnosing nonlinear mixed-effects models. AAPS J. 2011;13(2):143–51.
Standing JF, Morris J, Germovsek E, Lutsar I, Cortina-Borja. Methods for optimising neonatal antimicrobial use: time- and concentration-dependent agents [abstract no. 2225]. Annual Meeting of the Population Approach Group in Europe; 7–10 Jun 2011; Athens. http://www.page-meeting.org/?abstract=2225. Accessed 14 Jan 2013.
Moore RD, Lietman PS, Smith CR. Clinical response to aminoglycoside therapy: importance of the ratio of peak concentration to minimal inhibitory concentration. J Infect Dis. 1987;155(1):93–9.
EUCAST - European Committee on Antimicrobial Susceptibility Testing. Antimicrobial wild type distributions of microorganisms. http://mic.eucast.org/Eucast2/SearchController/search.jsp?action=performSearch&BeginIndex=0&Micdif=mic&NumberIndex=50&Antib=-1&Specium=22. Accessed 17 Sep 2012.
Giuliano RA, Verpooten GA, Verbist L, Wedeen RP, De Broe ME. In vivo uptake kinetics of aminoglycosides in the kidney cortex of rats. J Pharmacol Exp Ther. 1986;236(2):470–5.
van Hest RM, Mathot RA, Pescovitz MD, Gordon R, Mamelok RD, van Gelder T. Explaining variability in mycophenolic acid exposure to optimize mycophenolate mofetil dosing: a population pharmacokinetic meta-analysis of mycophenolic acid in renal transplant recipients. J Am Soc Nephrol. 2006;17(3):871–80.
Houk BE, Bello CL, Kang D, Amantea M. A population pharmacokinetic meta-analysis of sunitinib malate (SU11248) and its primary metabolite (SU12662) in healthy volunteers and oncology patients. Clin Cancer Res. 2009;15(7):2497–506.
Touw DJ, Vinks AA, Neef C. Pharmacokinetic modelling of intravenous tobramycin in adolescent and adult patients with cystic fibrosis using the nonparametric expectation maximization (NPEM) algorithm. Pharm World Sci. 1997;19(3):142–51.
Alghanem S, Paterson I, Thomson AH. Development of a tobramycin dosage adjustment nomogram for patients with cystic fibrosis [abstract no. 2077]. Annual Meeting of the Population Approach Group in Europe; 7–10 Jun 2011; Athens. http://www.page-meeting.org/?abstract=2077. Accessed 15 Jan 2012.
Conil JM, Georges B, Ruiz S, Rival T, Seguin T, Cougot P, et al. Tobramycin disposition in ICU patients receiving a once daily regimen: population approach and dosage simulations. Br J Clin Pharmacol. 2011;71(1):61–71.
Hermida J, Tutor JC. Serum cystatin C for the prediction of glomerular filtration rate with regard to the dose adjustment of amikacin, gentamicin, tobramycin, and vancomycin. Ther Drug Monit. 2006;28(3):326–31.
Bertenshaw C, Watson AR, Lewis S, Smyth A. Survey of acute renal failure in patients with cystic fibrosis in the UK. Thorax. 2007;62(6):541–5.
Smyth A, Lewis S, Bertenshaw C, Choonara I, McGaw J, Watson A. Case-control study of acute renal failure in patients with cystic fibrosis in the UK. Thorax. 2008;63(6):532–5.
Smyth A, Tan KH, Hyman-Taylor P, Mulheran M, Lewis S, Stableforth D, et al. Once versus three-times daily regimens of tobramycin treatment for pulmonary exacerbations of cystic fibrosis–the TOPIC study: a randomised controlled trial. Lancet. 2005;365(9459):573–8.
Rhodin MM, Anderson BJ, Peters AM, Coulthard MG, Wilkins B, Cole M, et al. Human renal function maturation: a quantitative description using weight and postmenstrual age. Pediatr Nephrol. 2009;24(1):67–76.
Viberg A, Cars O, Karlsson MO, Jonsson S. Estimation of cefuroxime dosage using pharmacodynamic targets, MIC distributions, and minimization of a risk function. J Clin Pharmacol. 2008;48(11):1270–81.
Acknowledgments
We would like to acknowledge Professor Sander Vinks from the School of Medicine at the University of Cincinnati, and the Division of Clinical Pharmacology and Paediatric Pharmacology Research Unit at the Cincinnati Children’s Hospital Medical Centre, Cincinnati, OH, USA, as well as Associate Professor Noel Cranswick and Associate Professor John Massie from Departments of Respiratory Medicine and Clinical Pharmacology, University of Melbourne and the Murdoch Children’s Research Institute and the Royal Children’s Hospital, Melbourne, VIC, Australia, for the contribution of data from their previous studies in patients with cystic fibrosis. Furthermore, we would like to acknowledge the use of a dataset provided by Professor Leon Aarons (laarons@fs1.pa.man.ac.uk), which was downloaded from the website of the Resource Facility for Population Kinetics (http://depts.washington.edu/rfpk/service/datasets/index.html) [funding source: NIH/NIBIB grant P41-EB01975] and published previously in an original publication (Aarons L, Vozeh S, Wenk M, Weiss P, Follath F. Population pharmacokinetics of tobramycin. Br J Clin Pharmacol. 1989 Sep;28(3):305–14), and Victoria Holden and colleagues at St James’s Hospital in Leeds, UK, for contributing data on children with febrile neutropenia. We also acknowledge support from the following individuals who helped with the data collection and creation of the database: Iona Paterson and Allan Smith, Cystic Fibrosis Unit Pharmacy Department, Gartnavel General Hospital, Glasgow, UK, Sonya Stacey, Royal Children’s Hospital, Brisbane, QLD, Australia. We would like to thank the High Performance Computing Support Group at the University of Queensland (http://www.hpcu.uq.edu.au/hpc/content/view/181/5/) for the support with the NONMEM® installation and the use of the computer facilities.
Author contributions
Dr Hennig had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the pharmacometric data analysis. Authors Hennig, Standing, Staatz and Thomson contributed to study concept and design, acquisition of data, analysis and interpretation of data, drafting of the manuscript and the critical revision of the manuscript for important intellectual content equally.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hennig, S., Standing, J.F., Staatz, C.E. et al. Population Pharmacokinetics of Tobramycin in Patients With and Without Cystic Fibrosis. Clin Pharmacokinet 52, 289–301 (2013). https://doi.org/10.1007/s40262-013-0036-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-013-0036-y