Abstract
Background
Clinical risk factors for nephrotoxicity in Staphylococcus aureus bacteraemia remain largely undetermined, despite its common occurrence and clinical significance. In an international, multicentre, prospective clinical trial (CAMERA2), which compared standard therapy (vancomycin monotherapy) to combination therapy (adding an anti-staphylococcal beta-lactam) for methicillin-resistant S. aureus bacteraemia, significantly more people in the combination therapy arm experienced acute kidney injury compared with those in the monotherapy arm (23% vs 6%).
Objective
The aim of this post hoc analysis was to explore in greater depth the risk factors for acute kidney injury from the CAMERA2 trial.
Methods
Among participants of the CAMERA2 trial, demographic-related, infection-related and treatment-related risk factors were assessed for their relationship with acute kidney injury by univariable and multivariable logistic regression. Acute kidney injury was defined by a modified-KDIGO (Kidney Disease: Improving Global Outcomes) criteria (not including urinary output).
Results
Of the 266 participants included, age (p = 0.04), randomisation to combination therapy (p = 0.002), vancomycin area under the concentration–time curve (p = 0.03) and receipt of (flu)cloxacillin as the companion beta-lactam (p < 0.001) were significantly associated with acute kidney injury. On a multivariable analysis, concurrent use of (flu)cloxacillin increased the risk of acute kidney injury over four times compared with the use of cefazolin or no beta-lactam. The association of vancomycin area under the concentration–time curve with acute kidney injury also persisted in the multivariable model.
Conclusions
For participants receiving vancomycin for S. aureus bacteraemia, use of (flu)cloxacillin and increased vancomycin area under the concentration–time curve were risk factors for acute kidney injury. These represent potentially modifiable risk factors for nephrotoxicity and highlight the importance of avoiding the use of concurrent nephrotoxins.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
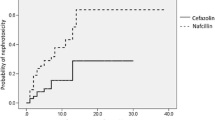
A significantly increased risk of acute kidney injury with concurrent vancomycin and (flu)cloxacillin use was found, compared with vancomycin with cefazolin or vancomycin alone. |
A higher vancomycin area under the concentration–time curve was also related to acute kidney injury. |
1 Introduction
Staphylococcus aureus bacteraemia remains a serious human infection, resulting in significant morbidity and mortality [1,2,3]. In Australia in 2017, 30-day mortality related to episodes of methicillin-resistant S. aureus (MRSA) infection was 18.9% [4]. Overall unadjusted in-hospital mortality in the USA between 2012 and 2017 was 18%, within the range of case fatality reports from other high-income countries between 15 and 23% [5,6,7]. Additionally, up to 40% of patients with MRSA bacteraemia experience acute kidney injury (AKI) [8]. This is a higher rate of AKI than seen in retrospective reviews for other organisms; AKI occurred in 29.5% of patients with carbapenem non-susceptible Gram-negative bacteraemia, and 36% of patients with melioidosis [9, 10]. Acute kidney injury has been associated with longer term sequelae for patients such as chronic kidney disease, kidney failure, fractures and hypertension [11,12,13].
Risk factors of nephrotoxicity in S. aureus bacteraemia remain poorly characterised [3, 14]. Vancomycin is a commonly used drug for the treatment of MRSA bacteraemia and a known nephrotoxin. Reported rates of vancomycin-induced nephrotoxicity vary between 5 and 43%, based on the diagnostic criteria and the presence of risk factors [15]. In clinical practice, clinical covariates that increase the risk of AKI for patients with MRSA bacteraemia need to be clearly identified, especially those that are modifiable.
The CAMERA2 trial was an international, multicentre, prospective clinical trial comparing standard MRSA therapy (monotherapy with vancomycin or daptomycin) to combination therapy (the addition of an anti-staphylococcal beta-lactam to standard therapy) for MRSA bacteraemia [16]. The rationale for combination therapy in CAMERA2 was derived from laboratory, animal and human studies, suggesting that combination therapy could be beneficial for the treatment of MRSA bacteraemia [17]. The CAMERA2 study was stopped early after review by the Data Safety and Monitoring Board, who identified an increased rate of AKI in the combination arm; modified RIFLE-defined AKI (any stage) occurred in 34/145 (23%) of patients in the combination therapy arm and 9/145 (6%) of patients in the standard therapy arm. Benefit in the primary outcome was not seen with combination therapy. Here, we provide a detailed post-hoc assessment of the clinical risk factors for AKI in the CAMERA2 trial participants.
2 Patients and Methods
2.1 Study Design and Population
This is a post hoc analysis of the prospective, multicentre, open-label, randomised CAMERA2 clinical trial (ClinicalTrials.gov Identifier: NCT02365493). The CAMERA2 trial enrolled 356 hospitalised adult patients with MRSA bacteraemia across 27 hospitals between August 2015 and July 2018. Full trial methodology and patient recruitment have been previously described [16, 18]. Institutional ethics approval was obtained at each study site, and written informed consent was obtained from each participant or surrogate decision maker.
To focus on AKI, we excluded patients who were undergoing haemodialysis or peritoneal dialysis at enrolment, missing a baseline serum creatinine concentration, or those with two or more missing creatinine measurements after baseline (creatinine was measured on days 2, 5 and 7). We also excluded patients incorrectly randomised, those lost to follow-up (e.g. self-discharge before 7 days) or who did not receive vancomycin, as described in Fig. 1. In the CAMERA2 trial, vancomycin was dosed in accordance with Australian guidelines via an intermittent infusion of 15–20 mg/kg 12 hourly (adjusted for renal function), preceded by a loading dose of 20–35 mg/kg (if considered appropriate by the treating clinician) or the Infectious Diseases Society of America guidelines with subsequent adjustment to maintain trough concentrations at 15–20 mg/L [18].
2.2 Data Collection
Demographic and patient factors were collected at trial entry. Acute kidney injury risk factors identified from the published literature and available from the trial database included the following patient factors: age, sex, weight, previous hospitalisation for ≥ 48 h in the past 90 days, chronic kidney disease, diabetes mellitus, liver disease, congestive cardiac failure, myocardial infarction, Charlson Comorbidity Index [19], baseline creatinine (µmol/L) and baseline C-reactive protein (mg/L). Baseline creatinine was defined as the highest creatinine measurement in the 24 h preceding randomisation. Infection-related factors of interest included place of acquisition, hypotension (systolic blood pressure < 90 mmHg or receipt of inotropes at the time of enrolment), source of infection, final diagnosis of endocarditis, Pitt bacteraemia score [20] and SOFA score [21]. Treatment factors were collected for the duration of the trial and included β-lactam treatment received [none, (flu)cloxacillin only, cefazolin only]; allocated treatment group (standard therapy, combination therapy); any antibiotic in the 72 h preceding randomisation; any β-lactam in 72 h preceding randomisation; use of drugs that can affect renal function in the 48 h preceding randomisation or between study days 1 and 7 (radiocontrast dye, loop diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, aminoglycosides, amphotericin B, nonsteroidal anti-inflammatory drugs [NSAIDs] and calcineurin inhibitors); any non-study antibiotic between days 1 and 7; vancomycin trough concentration (ideally from study day 2; however, for 30 patients, a day 2 vancomycin trough concentration was not available, and a day 1 trough concentration was used), and the calculated vancomycin 24-h area under the concentration–time curve (AUC). The vancomycin AUC were calculated from vancomycin dose and trough concentrations using a nonparametric Bayesian pharmacokinetic model with a one-compartment clearance model. Vancomycin trough concentrations were assumed to be drawn 15 minutes prior to the last vancomycin dose. Further details of the AUC estimations, including goodness-of-fit results, have been previously published [22].
2.3 Outcome Measures
A modified RIFLE definition for AKI was a pre-specified CAMERA2 secondary trial outcome [23]. For this post hoc analysis, we classified kidney injury with the more contemporary KDIGO (Kidney Disease: Improving Global Outcomes) criteria and used a modified KDIGO (mKDIGO) as the primary outcome. As urinary output was not available, we defined the mKIDGO AKI as a ≥ 1.5-fold increase in serum creatinine from baseline at any time within the first 7 days; or an increase in serum creatinine of ≥ 0.3 mg/dL (≥ 26.5 µmol/L) within a 48-h period; or a new need for renal replacement therapy prior to day 90 [24]. Using KDIGO, stage 1 was defined as a serum creatinine level 1.5 to < 2.0 times baseline in the first 7 days or a ≥ 26.5 μmol/L increase from the baseline creatinine level in the first 48 h; stage 2 was a serum creatinine level 2.0 to < 3.0 times the baseline in the first 7 days; Stage 3 was a serum creatinine level ≥ 3.0 times the baseline in the first 7 days OR a ≥ 353.6-μmol/L increase from the baseline in the first 48 h OR new initiation of renal replacement therapy within 90 days (date of initiation of renal replacement therapy was not available). Fold change in creatinine was available for days 2, 5 and 7 and the highest value was used to stage AKI.
2.4 Statistical Analysis
Summary statistics describing the demographic, patient and treatment factors of interest are provided. Univariable logistic regression models were used to describe the association between each exposure and AKI. Unadjusted odds ratios (ORs) with corresponding 95% confidence intervals (CIs) and p values were reported for each factor. Likelihood ratio tests were used to assess the overall effect of categorical variables with more than two categories. Continuous exposures were assessed to ensure that the association between each exposure and log odds of AKI was approximately linear. Where this assumption was violated, categorical versions of these variables based on clinical knowledge were used instead.
Results from univariable analyses were used to guide variable selection for the subsequent multivariable analysis. Exposure variables where the associated p value from the univariable analysis was ≤ 0.1 were included in multivariable models unless collinearity between variables was likely—in this case, only one variable was included in the multivariable analysis. Choosing which variable to include in the setting of collinearity was based on the ability to provide clinically useful information. Additionally, the following relevant factors were included as forced covariates in the model: age, sex and baseline creatinine. Previously described methods of purposeful variable selection [25] were used to ensure the multivariable analyses were robust and included all important exposures and confounders. Adjusted ORs and 95% CIs for each of the risk factors included in the model were obtained from the multivariable analysis and reported.
During analysis of baseline creatinine, association between AKI and baseline creatinine was nonlinear. To incorporate the nonlinearity of this association into the model, baseline creatinine was categorised into levels of < 110 µmol/L, 111–230 µmol/L and above 231 µmol/L. These were decided a priori, to reflect the estimated glomerular filtration rate of above 60 mL/min, 30–60 mL/min and below 30 mL/min.
In this analysis, drugs that can affect renal function were assessed in two groups to differentiate between drugs considered true nephrotoxins (NSAIDs and aminoglycosides) and a larger group of potential nephrotoxins (referred to as drugs affecting kidney function and includes radiocontrast dye, loop diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, NSAIDs, aminoglycosides and calcineurin inhibitors) that are either known nephrotoxins or can cause nephrotoxicity in certain clinical scenarios. Timing of administration of these drugs was divided into two timeframes, the 48 h before study enrolment, and the first 7 days after enrolment. Statistical analyses were conducted using Stata (Stata Statistical Software: Release 16, 2019; StataCorp LLC, College Station, TX, USA).
3 Results
There were 356 participants in the full CAMERA2 dataset. Of these, 90 were excluded from further analysis (Fig. 1). Of the remaining 266 included, 46 participants (17.3%) experienced AKI. A breakdown of the stage of AKI experienced is shown in Table 1. Of the seven patients who required renal replacement therapy at any time in the CAMERA2 follow-up period, two remained on renal replacement therapy at day 90.
Participant characteristics are presented in Table 2. The median age of the cohort was 64 years; age was higher in those who did not experience AKI (66 years) compared with those who did experience AKI (56 years) [p = 0.039, OR 0.98 (0.97–1.00)]. No other patient factors were significantly different between the AKI and no AKI groups. Diabetes was present in 44% of patients (n = 117). Receipt of any antibiotic, or a beta-lactam specifically, in the 72 h– before enrolment, or prescription of a non-study antibiotic between study days 1–7 was not significantly different between those who did and did not experience AKI (data not shown). Chronic kidney disease was documented as a comorbidity in 22% of patients (n = 59), though a baseline creatinine level above 110 µmol/L was present in 37% (n = 99).
Infection-related and treatment-related factors and their relationship to mKDIGO-defined AKI are shown in Table 3. Place of acquisition of infection and source of infection were not risk factors for AKI. Randomisation to combination therapy was significantly associated with AKI (p = 0.002), as was receipt of (flu)cloxacillin as the companion beta-lactam (p < 0.001). Calculated day 2 median vancomycin AUC was higher in patients who developed AKI (461 mg × h/L) compared with those who did not (400 mg × h/L, p = 0.03). Vancomycin trough concentration at day 2 was not significantly different between the patients who did and did not experience AKI.
Despite over 60% of patients receiving concurrent administration of a drug that can affect kidney function (radiocontrast dye, loop diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, NSAIDS, aminoglycosides and calcineurin inhibitors) both in the 48 h before study enrolment and during the first 7 study days, there was no statistical association with the development of AKI (see Table 4).
Based on results from the univariable analysis, age, choice of companion beta-lactam and vancomycin AUC met criteria for inclusion in the multivariable model (p ≥ 0.1). Randomisation to combination therapy was not included because of collinearity with the beta-lactam treatment received. Sex and baseline creatinine were also included as clinically significant variables from the published literature. The results of the multivariable analysis are shown in Table 5.
The multivariable analysis identified concurrent use of (flu)cloxacillin as a risk factor for AKI (p < 0.001), increasing the risk of AKI significantly compared with the use of cefazolin or no beta-lactam (OR 4.50; 95% CI 2.09–9.70). Increased vancomycin AUC was also associated with AKI (OR 1.10 for a 50 mg × h/L increase in vancomycin AUC, 95% CI 1.01–1.21; p = 0.04).
4 Discussion
In this post hoc analysis of the CAMERA2 trial, use of (flu)cloxacillin in combination with anti-MRSA therapy (predominantly vancomycin) was significantly associated with AKI in a cohort of patients with MRSA bacteraemia, increasing the odds of AKI over four-fold. Use of cefazolin as the companion beta-lactam was not associated with an increased risk of AKI compared with no beta-lactam. These data suggest a drug interaction may be occurring between flucloxacillin and vancomycin, although we were unable to statistically show this without a beta-lactam monotherapy group. Increased vancomycin AUC was also associated with AKI. Although a detailed analysis of the association between vancomycin exposure and AKI in CAMERA2 has been undertaken [22], this analysis had a broader scope, including other relevant risk factors for AKI with markers of vancomycin exposure.
Clinical risk factors related to AKI in patients with MRSA bacteraemia are poorly characterised. The clinical utility of identifying modifiable risk factors for AKI is significant and has an important role in kidney stewardship. In a study of 335 patients with MRSA bacteraemia in Korea, 135 patients developed AKI, and risk factors for nephrotoxicity included male sex, pre-existing kidney disease, intra-abdominal or central venous catheter infection, and higher Pitt bacteraemia scores [8]. We did not find a correlation between sex, pre-existing kidney disease, source of infection, or Pitt bacteraemia scores and AKI in our analysis.
In this analysis, mKDIGO was used to define AKI, as the KDIGO criteria was included for AKI diagnosis in the 2020 consensus guideline on vancomycin-induced kidney injury [26]. Using KDIGO, which includes an absolute creatinine change in diagnosis, provides the advantage of improving AKI diagnosis across a variety of baseline creatinine levels [27]. Although the relatively small changes in creatinine needed to diagnose stage 1 AKI can relate to either mild fluctuations in kidney function or true AKI, evidence suggests even these small changes in creatinine can affect patient outcomes [28, 29]. Similarly, creatinine elevations in patients on vancomycin have a significant impact on outcomes. In a retrospective review of 128,993 adult patients, a linear increase in in-hospital mortality was found across categories of creatinine increase, with ORs for mortality that ranged from 1.60 (95% CI 1.47–1.75) for a serum creatinine increase of > 0–10% to 13.66 for a serum creatinine increase of > 200% [30]. The longer term implications of vancomycin-induced kidney injury remain undetermined.
All patients in this analysis received vancomycin, a known nephrotoxin. Many risk factors for vancomycin-induced nephrotoxicity have been proposed, including a trough concentration above 15 mg/L, duration of therapy (longer than 7 days), greater patient weight (over 100 kg), pre-existing kidney disease or a previous episode of AKI, concomitant use of nephrotoxins and a longer duration of admission in an intensive care unit [15]. A prospective observational study of vancomycin-induced nephrotoxicity identified baseline creatinine clearance, malignancy, previous AKI and admission to the intensive care unit as risk factors for AKI, and that AKI was significantly associated with mortality (19 vs 5%, p < 0.05) [31]. A 2012 literature review similarly identified age, longer duration of therapy, concomitant use of nephrotoxic agents, high trough concentrations of vancomycin and critical illness, or kidney impairment at baseline as AKI risk factors [32]. Because of the detailed data collection from the primary CAMERA2 study, we were able to assess for the majority of these risk factors. In the univariable analysis, we included both trough vancomycin concentration and vancomycin AUC as markers of vancomycin exposure, with discordant results. Vancomycin AUC was associated with the development of AKI, where trough concentration was not. This supports human and animal data showing a relationship between vancomycin AUC and nephrotoxicity [33,34,35]. Although the vancomycin AUC was associated with AKI, the result was borderline with a large overlap in the AUCs obtained between the AKI and no AKI groups. Patients treated with daptomycin monotherapy are likely to be systematically different from those treated with vancomycin and were excluded from the analysis.
The biological mechanisms for (flu)cloxacillin causing AKI in patients with MRSA bacteraemia receiving concurrent vancomycin remain unknown. Penicillin and cephalosporin antibiotics are considered a rare cause of nephrotoxicity, primarily mediated by acute interstitial nephritis (AIN) related to hypersensitivity [36,37,38,39]. However, piperacillin (with tazobactam) has been reported to increase the rate of nephrotoxicity when combined with vancomycin, particularly compared with vancomycin with cefepime or meropenem [40,41,42]. For piperacillin, the most popular hypothesis for this effect is a combination of AIN caused by the penicillin and direct cellular toxicity from vancomycin [43]. Very limited data from kidney biopsies of patients on vancomycin and piperacillin and tazobactam show AIN, acute tubular necrosis or both [44]. There are flaws in interpreting an elevated creatinine as synonymous with kidney injury, the combination of piperacillin (with tazobactam) and vancomycin could be causing a hypercreatinineamia without kidney injury (i.e. pseudonephrotoxicity) related to the blockade of secretory pathways or reabsorption [45]. Additionally, some beta-lactams (e.g. flucloxacillin) affect organic anion cotransporter 3, which facilitates uptake into proximal tubular cells, where vancomycin may also be present in sufficient concentrations to cause damage [46, 47]. Interaction with this transporter was most pronounced with nafcillin and was related to drug lipophilicity. This theory also accounts for why hydrophilic beta-lactams (i.e. aminopenicillins and cephalosporins) may be safer in combination with vancomycin. The risk of AKI may also be increased when vancomycin is used with penicillins because of physical incompatibilities causing precipitation that could accumulate in kidney tubules; however, as the flucloxacillin was administered separately to the vancomycin, this is less likely [48].
In our analysis, the patient numbers were too small to separate flucloxacillin and cloxacillin, thus it remains unclear if the risk of AKI applies to both drugs equally. The rates of AKI were similar for flucloxacillin (25/90 [28%]) and cloxacillin (5/21 [24%]) [16]. Therefore, we expected these drugs to perform similarly, and to avoid overfitting with the small sample size, we did not separate them in the multivariable analysis.
The risk of AKI with flucloxacillin may be underappreciated in clinical practice. In an analysis of patients with AIN confirmed by kidney biopsy, flucloxacillin was the most common beta-lactam implicated [49]. Even with only 24 hours of flucloxacillin for surgical prophylaxis, the rate of AKI was 8.5% when used as monotherapy, with the rate of AKI increasing when flucloxacillin was used in combination with gentamicin [50, 51].
This analysis has limitations, the CAMERA2 study and data collection were not designed to identify clinical predictors of AKI. Concomitant drugs were included in the model without consideration of dose or duration of therapy, limiting our ability to truly understand the effects of these drugs on the development of AKI. We included one group of ‘true nephrotoxins’ separately to a larger group of these nephrotoxins with drugs that can affect kidney function (though are not nephrotoxins) or have been shown in other research to be associated with vancomycin-induced nephrotoxicity, despite not generally considered nephrotoxic when used in isolation (e.g. loop diuretics, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers) [15, 52]. Vancomycin-induced kidney injury in the monotherapy arm was low at 6%. Thus, only a small cohort of patients were included who experienced AKI, and the analysis may be underpowered to detect significant predictors of AKI (e.g. the traditional risk factors associated with vancomycin-induced nephrotoxicity). An analysis combining the results of CAMERA2 and PROVIDE (another prospective study that focused on the relationship between vancomycin exposure and kidney injury [53]) demonstrated that exposure toxicity relationships were highly similar between the two prospective trials, with CAMERA2 demonstrating a slightly lower rate of AKI [54]. Another important limitation is a lack of clinical information about the presumed aetiology of the AKI; kidney biopsies were not performed, and urinary characteristics (i.e. presence of eosinophils) were not available. Hospital-specific factors were not considered; however, treatment was largely standardised by virtue of enrolment in the clinical trial.
5 Conclusions
In this post hoc analysis of patients in the CAMERA2 trial who received vancomycin, the use of (flu)cloxacillin and vancomycin AUC were risk factors for AKI, though a definite biological mechanism for this effect remains undefined. Combination therapy represents a modifiable risk factor for nephrotoxicity, with significant implications for recommending antimicrobial therapy for MRSA bacteraemia.
Change history
19 November 2022
A Correction to this paper has been published: https://doi.org/10.1007/s40261-022-01224-9
References
Shorr AF, Tabak YP, Killian AD, Gupta V, Liu LZ, Kollef MH. Healthcare-associated bloodstream infection: a distinct entity? Insights from a large US database. Crit Care Med. 2006;34(10):2588–95. https://doi.org/10.1097/01.CCM.0000239121.09533.09.
Thwaites GE, Edgeworth JD, Gkrania-Klotsas E, Kirby A, Tilley R, Torok ME, et al. Clinical management of Staphylococcus aureus bacteraemia. Lancet Infect Dis. 2011;11(3):208–22. https://doi.org/10.1016/S1473-3099(10)70285-1.
van Hal SJ, Jensen SO, Vaska VL, Espedido BA, Paterson DL, Gosbell IB. Predictors of mortality in Staphylococcus aureus bacteremia. Clin Microbiol Rev. 2012;25(2):362–86. https://doi.org/10.1128/CMR.05022-11.
Australian Commission on Safety and Quality in Health Care (ACSQHC). AURA 2019: third Australian report on antimicrobial use and resistance in human health. Sydney: ACSQHC; 2019.
Kourtis AP, Hatfield K, Baggs J, Mu Y, See I, Epson E, et al. Vital signs: epidemiology and recent trends in methicillin-resistant and in methicillin-susceptible Staphylococcus aureus bloodstream infections: United States. MMWR Morb Mortal Wkly Rep. 2019;68(9):214–9. https://doi.org/10.15585/mmwr.mm6809e1.
Tong SY, Davis JS, Eichenberger E, Holland TL, Fowler VG Jr. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev. 2015;28(3):603–61. https://doi.org/10.1128/CMR.00134-14.
Verway M, Brown KA, Marchand-Austin A, Diong C, Lee S, Langford B, et al. Prevalence and mortality associated with bloodstream organisms: a population-wide retrospective cohort study. J Clin Microbiol. 2022. https://doi.org/10.1128/jcm.02429-21.
Joo EJ, Peck KR, Ha YE, Kim YS, Song YG, Lee SS, et al. Impact of acute kidney injury on mortality and medical costs in patients with meticillin-resistant Staphylococcus aureus bacteraemia: a retrospective, multicentre observational study. J Hosp Infect. 2013;83(4):300–6. https://doi.org/10.1016/j.jhin.2012.12.008.
Prabhu RA, Shaw T, Rao IR, Kalwaje Eshwara V, Nagaraju SP, Shenoy SV, et al. Acute kidney injury and its outcomes in melioidosis. J Nephrol. 2021;34(6):1941–8. https://doi.org/10.1007/s40620-021-00970-x.
Papadimitriou-Olivgeris M, Assimakopoulos SF, Kolonitsiou F, Solomou A, Vamvakopoulou S, Spyropoulou A, et al. Risk factors for acute kidney injury in critically ill patients with bacteraemia by carbapenem non-susceptible Gram negative bacteria. Infez Med. 2019;27(4):380–92.
Hall RG 2nd, Giuliano CA, Haase KK, Hazlewood KA, Frei CR, Forcade NA, et al. Empiric guideline-recommended weight-based vancomycin dosing and mortality in methicillin-resistant Staphylococcus aureus bacteremia: a retrospective cohort study. BMC Infect Dis. 2012;12:104. https://doi.org/10.1111/jcpt.12203.
Noble RA, Lucas BJ, Selby NM. Long-term outcomes in patients with acute kidney injury. Clin J Am Soc Nephrol. 2020;15(3):423–9. https://doi.org/10.2215/CJN.10410919.
Blot SI, Vandewoude KH, Hoste EA, Colardyn FA. Outcome and attributable mortality in critically Ill patients with bacteremia involving methicillin-susceptible and methicillin-resistant Staphylococcus aureus. Arch Intern Med. 2002;162(19):2229–35. https://doi.org/10.1001/archinte.162.19.2229.
Yilmaz M, Elaldi N, Balkan II, Arslan F, Batirel AA, Bakici MZ, et al. Mortality predictors of Staphylococcus aureus bacteremia: a prospective multicenter study. Ann Clin Microbiol Antimicrob. 2016;15:7. https://doi.org/10.1186/s12941-016-0122-8.
van Hal SJ, Paterson DL, Lodise TP. Systematic review and meta-analysis of vancomycin-induced nephrotoxicity associated with dosing schedules that maintain troughs between 15 and 20 milligrams per liter. Antimicrob Agents Chemother. 2013;57(2):734–44. https://doi.org/10.1128/AAC.01568-12.
Tong SYC, Lye DC, Yahav D, Sud A, Robinson JO, Nelson J, et al. Effect of vancomycin or daptomycin with vs without an antistaphylococcal beta-lactam on mortality, bacteremia, relapse, or treatment failure in patients with MRSA bacteremia: a randomized clinical trial. JAMA. 2020;323(6):527–37. https://doi.org/10.1001/jama.2020.0103.
Davis JS, Sud A, O’Sullivan MVN, Robinson JO, Ferguson PE, Foo H, et al. Combination of vancomycin and beta-lactam therapy for methicillin-resistant Staphylococcus aureus bacteremia: a pilot multicenter randomized controlled trial. Clin Infect Dis. 2016;62(2):173–80. https://doi.org/10.1093/cid/civ808.
Tong SY, Nelson J, Paterson DL, Fowler VG Jr, Howden BP, Cheng AC, et al. CAMERA2—combination antibiotic therapy for methicillin-resistant Staphylococcus aureus infection: study protocol for a randomised controlled trial. Trials. 2016;17:170. https://doi.org/10.1093/cid/civ808.
Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–51. https://doi.org/10.1016/0895-4356(94)90129-5.
Roth JA, Tschudin-Sutter S, Dangel M, Frei R, Battegay M, Widmer AF. Value of the Pitt bacteraemia score to predict short-term mortality in Staphylococcus aureus bloodstream infection: a validation study. Swiss Med Wkly. 2017;147: w14482. https://doi.org/10.4414/smw.2017.14482.
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996;22(7):707–10. https://doi.org/10.1007/BF01709751.
Liu J, Tong SYC, Davis JS, Rhodes NJ, Scheetz MH, Group CS. Vancomycin exposure and acute kidney injury outcome: a snapshot from the CAMERA2 Study. Open Forum Infect Dis. 2020;7(12):ofaa538. https://doi.org/10.1093/ofid/ofaa538.
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative Workgroup. Acute renal failure: definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):204–12. https://doi.org/10.1186/cc2872.
Kidney Disease: Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2:1–138.
Hosmer DW, Lemeshow S, Sturdivant RX. Applied logistic regression. 3rd ed. Hoboken: Wiley; 2013.
Rybak MJ, Le J, Lodise TP, Levine DP, Bradley JS, Liu C, et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: a revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Clin Infect Dis. 2020;71(6):1361–4. https://doi.org/10.1093/ajhp/zxaa036.
Kellum JA, Lameire N, KDIGO AKI Guideline Work Group. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care. 2013;17(1):204. https://doi.org/10.1186/cc11454.
Thakar CV, Christianson A, Freyberg R, Almenoff P, Render ML. Incidence and outcomes of acute kidney injury in intensive care units: a Veterans Administration study. Crit Care Med. 2009;37(9):2552–8. https://doi.org/10.1097/CCM.0b013e3181a5906f.
Lassnigg A, Schmidlin D, Mouhieddine M, Bachmann LM, Druml W, Bauer P, et al. Minimal changes of serum creatinine predict prognosis in patients after cardiothoracic surgery: a prospective cohort study. J Am Soc Nephrol. 2004;15(6):1597–605. https://doi.org/10.1097/01.asn.0000130340.93930.dd.
Yu KC, Yamaga C, Vankeepuram L, Tabak YP. Relationships between creatinine increase and mortality rates in patients given vancomycin in 76 hospitals: the increasing role of infectious disease pharmacists. Am J Health Syst Pharm. 2021. https://doi.org/10.1093/ajhp/zxab247.
Minejima E, Choi J, Beringer P, Lou M, Tse E, Wong-Beringer A. Applying new diagnostic criteria for acute kidney injury to facilitate early identification of nephrotoxicity in vancomycin-treated patients. Antimicrob Agents Chemother. 2011;55(7):3278–83. https://doi.org/10.1128/AAC.00173-11.
Elyasi S, Khalili H, Dashti-Khavidaki S, Mohammadpour A. Vancomycin-induced nephrotoxicity: mechanism, incidence, risk factors and special populations: a literature review. Eur J Clin Pharmacol. 2012;68(9):1243–55. https://doi.org/10.1007/s00228-012-1259-9.
Aljefri DM, Avedissian SN, Rhodes NJ, Postelnick MJ, Nguyen K, Scheetz MH. Vancomycin area under the curve and acute kidney injury: a meta-analysis. Clin Infect Dis. 2019;69(11):1881–7. https://doi.org/10.1093/cid/ciz051.
Avedissian SN, Pais GM, O’Donnell JN, Lodise TP, Liu J, Prozialeck WC, et al. Twenty-four hour pharmacokinetic relationships for intravenous vancomycin and novel urinary biomarkers of acute kidney injury in a rat model. J Antimicrob Chemother. 2019;74(8):2326–34. https://doi.org/10.1093/jac/dkz167.
Rhodes NJ, Prozialeck WC, Lodise TP, Venkatesan N, O’Donnell JN, Pais G, et al. Evaluation of vancomycin exposures associated with elevations in novel urinary biomarkers of acute kidney injury in vancomycin-treated rats. Antimicrob Agents Chemother. 2016;60(10):5742–51. https://doi.org/10.1128/AAC.00591-16.
Xu B, Murray M. Flucloxacillin induced acute renal failure. Aust Fam Physician. 2008;37(12):1009–11.
Bakker SJ, Luik AJ, Leunissen KM. Flucloxacillin-induced acute interstitial nephritis. Nephrol Dial Transplant. 1995;10(4):579.
Linton AL, Clark WF, Driedger AA, Turnbull DI, Lindsay RM. Acute interstitial nephritis due to drugs: review of the literature with a report of nine cases. Ann Intern Med. 1980;93(5):735–41. https://doi.org/10.7326/0003-4819-93-5-735.
Moledina DG, Perazella MA. Drug-induced acute interstitialnNephritis. Clin J Am Soc Nephrol. 2017;12(12):2046–9. https://doi.org/10.2215/CJN.07630717.
Bellos I, Karageorgiou V, Pergialiotis V, Perrea DN. Acute kidney injury following the concurrent administration of antipseudomonal beta-lactams and vancomycin: a network meta-analysis. Clin Microbiol Infect. 2020;26(6):696–705. https://doi.org/10.1016/j.cmi.2020.03.019.
Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL. Vancomycin plus piperacillin-tazobactam and acute kidney injury in adults: a systematic review and meta-analysis. Crit Care Med. 2018;46(1):12–20. https://doi.org/10.1097/CCM.0000000000002769.
Mullins BP, Kramer CJ, Bartel BJ, Catlin JS, Gilder RE. Comparison of the nephrotoxicity of vancomycin in combination with cefepime, meropenem, or piperacillin/tazobactam: a prospective, multicenter study. Ann Pharmacother. 2018;52(7):639–44. https://doi.org/10.1177/1060028018757497.
Watkins RR, Deresinski S. Increasing evidence of the nephrotoxicity of piperacillin/tazobactam and vancomycin combination therapy: what is the clinician to do? Clin Infect Dis. 2017;65(12):2137–43. https://doi.org/10.1093/cid/cix675.
Blair M, Cote JM, Cotter A, Lynch B, Redahan L, Murray PT. Nephrotoxicity from vancomycin combined with piperacillin-tazobactam: a comprehensive review. Am J Nephrol. 2021;52(2):85–97. https://doi.org/10.1159/000513742.
Avedissian SN, Pais GM, Liu J, Rhodes NJ, Scheetz MH. Piperacillin-tazobactam added to vancomycin increases risk for acute kidney injury: fact or fiction? Clin Infect Dis. 2020;71(2):426–32. https://doi.org/10.1093/cid/ciz1189.
Rose W, Fantl M, Geriak M, Nizet V, Sakoulas G. Current paradigms of combination therapy in methicillin-resistant Staphylococcus aureus (MRSA) bacteremia: does it work, which combination, and for which patients? Clin Infect Dis. 2021;73(12):2353–60. https://doi.org/10.1093/cid/ciab452.
Wolman AT, Gionfriddo MR, Heindel GA, Mukhija P, Witkowski S, Bommareddy A, et al. Organic anion transporter 3 interacts selectively with lipophilic beta-lactam antibiotics. Drug Metab Dispos. 2013;41(4):791–800. https://doi.org/10.1124/dmd.112.049569.
Raverdy V, Ampe E, Hecq JD, Tulkens PM. Stability and compatibility of vancomycin for administration by continuous infusion. J Antimicrob Chemother. 2013;68(5):1179–82. https://doi.org/10.1093/jac/dks510.
Lazarus B, Davies MRP, Trubiano JA, Pellicano R. Time to acute kidney injury in beta-lactam-induced acute interstitial nephritis. Kidney Int Rep. 2020;5(7):1068–70. https://doi.org/10.1016/j.ekir.2020.04.008.
Graham J, Borthwick E, Hill C, Blaney J, Gallagher N, Armstrong L, et al. Acute kidney injury following prophylactic flucloxacillin and gentamicin in primary hip and knee arthroplasty. Clin Kidney J. 2021;14(4):1114–9. https://doi.org/10.1093/ckj/sfaa059.
Challagundla SR, Knox D, Hawkins A, Hamilton D, Flynn RWV, Robertson S, et al. Renal impairment after high-dose flucloxacillin and single-dose gentamicin prophylaxis in patients undergoing elective hip and knee replacement. Nephrol Dial Transplant. 2013;28(3):612–9. https://doi.org/10.1093/ndt/gfs458.
Norton K, Ingram PR, Heath CH, Manning L. Risk factors for nephrotoxicity in patients receiving outpatient continuous infusions of vancomycin in an Australian tertiary hospital. J Antimicrob Chemother. 2014;69(3):805–8. https://doi.org/10.1093/jac/dkt402.
Lodise TP, Rosenkranz SL, Finnemeyer M, Evans S, Sims M, Zervos MJ, et al. The emperor’s new clothes: PRospective Observational evaluation of the association between initial VancomycIn exposure and failure rates among ADult HospitalizEd patients with methicillin-resistant Staphylococcus aureus bloodstream infections (PROVIDE). Clin Infect Dis. 2020;70(8):1536–45. https://doi.org/10.1093/cid/ciz460.
Scheetz MH, Pais GM, Lodise TP, Tong SYC, Davis JS, O’Donnell JN, et al. Of rats and men: a translational model to understand vancomycin pharmacokinetic/toxicodynamic relationships. Antimicrob Agents Chemother. 2021;65(10): e0106021. https://doi.org/10.1128/AAC.01060-21.
Acknowledgements
We thank the CAMERA2 Study Group for use of the data from the CAMERA2 trial. The CAMERA2 Study Group collaborating authors were: Nick Anagostou, David Andresen, Sophia Archuleta, Narin Bak, Mark Chatfield, Allen Cheng, Yael Dishon, Ravindra Dotel, Patricia Ferguson, Hong Foo, Vance Fowler, Niladri Ghosh, Timothy Gray, Stephen Guy, Natasha Holmes, Benjamin Howden, Shirin Kalimuddin, David Lye, Stephen McBride, Genevieve McKew, Jane Nelson, Matthew O’Sullivan, David Paterson, Mical Paul, David Price, Anna Ralph, Matthew Roberts, Owen Robinson, Ben Rogers, Naomi Runnegar, Simon Smith, Archana Sud, Steven Tong, Adrian Tramontana, Sebastian Van Hal, Genevieve Walls, Morgyn Warner, Dafna Yahav, Barnaby Young, Jane Davies, Alan Cass, Joshua Davis and Steven Tong.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The CAMERA2 trial was supported by Australian National Health & Medical Research Council Career Development Fellowships for Steven Y. C. Tong (1145033) and Joshua S. Davis (1160331).
Conflict of interest
Marc H. Scheetz has ongoing research contracts with Nevakar and SuperTrans Medical as well as having filed patent US10688195B2. He is supported in part by the National Institute of Allergy and Infectious Diseases under award number R21-AI149026. Jason A. Roberts acknowledge funding from the Australian National Health and Medical Research Council for a Centre of Research Excellence (APP2007007) and an Investigator Grant (APP2009736) as well as an Advancing Queensland Clinical Fellowship. Amy Legg, Niamh Meagher, Sandra A. Johnson, Matthew A. Roberts, Alan Cass, Jane Davies, Joshua S. Davis and Steven Y.C. Tong have no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
All procedures in this study were in accordance with the 1964 Declaration of Helsinki (and its amendments). Institutional ethics approval was obtained at each site as amendments to the CAMERA2 trial approval (principal site Hunter New England 2019/ETH00841).
Consent to participate
Written informed consent was obtained from each participant or surrogate decision maker for the CAMERA2 trial.
Consent for publication
Not applicable.
Availability of data and material
Deidentified data can be made available based on a reasonable request to the authors.
Code availability
Not available.
Author contributions
Conceptualisation ST, JoD, ST; statistical analysis: NM; draft manuscript: AL JoD, ST, manuscript review and advice: all authors.
Additional information
Disclaimer: The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
The original online version of this article was revised: There was an error in the affiliation details for authors Amy Legg, Alan Cass, Jane Davies and Joshua S. Davis and it has been corrected.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Legg, A., Meagher, N., Johnson, S.A. et al. Risk Factors for Nephrotoxicity in Methicillin-Resistant Staphylococcus aureus Bacteraemia: A Post Hoc Analysis of the CAMERA2 Trial. Clin Drug Investig 43, 23–33 (2023). https://doi.org/10.1007/s40261-022-01204-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-022-01204-z